[English] 日本語
 Yorodumi
Yorodumi- PDB-1enm: UDA TRISACCHARIDE COMPLEX. CRYSTAL STRUCTURE OF URTICA DIOICA AGG... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1enm | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | UDA TRISACCHARIDE COMPLEX. CRYSTAL STRUCTURE OF URTICA DIOICA AGGLUTININ, A SUPERANTIGEN PRESENTED BY MHC MOLECULES OF CLASS I AND CLASS II | ||||||||||||
 Components Components | AGGLUTININ ISOLECTIN I/AGGLUTININ ISOLECTIN V/ AGGLUTININ ISOLECTIN VI | ||||||||||||
 Keywords Keywords | SUGAR BINDING PROTEIN / LECTIN / HEVEIN DOMAIN / UDA / SUPERANTIGEN / SACCHARIDE BINDING | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationendochitinase activity / chitinase / chitin catabolic process / chitin binding / polysaccharide catabolic process / defense response to fungus / cell wall macromolecule catabolic process / carbohydrate binding / killing of cells of another organism / metal ion binding Similarity search - Function | ||||||||||||
| Biological species |  Urtica dioica (great nettle) Urtica dioica (great nettle) | ||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.9 Å MOLECULAR REPLACEMENT / Resolution: 1.9 Å | ||||||||||||
 Authors Authors | Saul, F.A. / Rovira, P. / Boulot, G. / Van Damme, E.J.M. / Peumans, W.J. / Truffa-Bachi, P. / Bentley, G.A. | ||||||||||||
 Citation Citation |  Journal: Structure Fold.Des. / Year: 2000 Journal: Structure Fold.Des. / Year: 2000Title: Crystal structure of Urtica dioica agglutinin, a superantigen presented by MHC molecules of class I and class II. Authors: Saul, F.A. / Rovira, P. / Boulot, G. / Van Damme, E.J.M. / Peumans, W.J. / Truffa-Bachi, P. / Bentley, G.A. #1:  Journal: Plant Mol.Biol. / Year: 1999 Journal: Plant Mol.Biol. / Year: 1999Title: Characterisation of Urtica dioica Agglutinin Isolectins and the Encoding Gene Family Authors: Does, M.P. / Ng, D.K. / Dekker, H.L. / Peumans, W.J. / Houterman, P.M. / Van Damme, E.J. / Cornelissen, B.J.C. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1enm.cif.gz 1enm.cif.gz | 34 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1enm.ent.gz pdb1enm.ent.gz | 21.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1enm.json.gz 1enm.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/en/1enm https://data.pdbj.org/pub/pdb/validation_reports/en/1enm ftp://data.pdbj.org/pub/pdb/validation_reports/en/1enm ftp://data.pdbj.org/pub/pdb/validation_reports/en/1enm | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1eisSC  1en2C S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 9350.269 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Details: PURIFIED FROM RHIZOMES / Source: (natural)  Urtica dioica (great nettle) / References: GenBank: 4138900, UniProt: P11218*PLUS Urtica dioica (great nettle) / References: GenBank: 4138900, UniProt: P11218*PLUS |
|---|---|
| #2: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2- ...2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose / triacetyl-beta-chitotriose |
| #3: Water | ChemComp-HOH / |
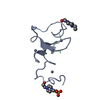
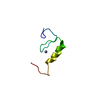
| Compound details | The structure comprises two hevein-like domains, each containing a distinct saccharide-binding site. ...The structure comprises two hevein-like domains, each containing a distinct saccharide-binding site. The two binding sites are located at opposite extremities of the molecule. The principal binding-site residues are SER 19, TRP 21, TRP 23, and TYR 30 on the first domain, and the homologous residues SER 65, HIS 67, TRP 69, and TYR 76 on the second domain. The crystallographic asymmetric unit contains one molecule of UDA and a single trisaccharide ligand. The ligand interacts simultaneously with the binding site on the N-terminal domain of one molecule and that of the C-terminal domain of a symmetry-related molecule. |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.72 Å3/Da / Density % sol: 54.81 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 290 K / Method: vapor diffusion, sitting drop / pH: 6 Details: PEG 6000, sodium acetate, sodium chloride, pH 6.0, VAPOR DIFFUSION, SITTING DROP, temperature 290.0K | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS PH range low: 6.5 / PH range high: 5.5 | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 298 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: LURE SYNCHROTRON / Site: LURE  / Beamline: DW32 / Wavelength: 0.96 / Beamline: DW32 / Wavelength: 0.96 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Oct 22, 1999 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.96 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→25 Å / Num. all: 8284 / Num. obs: 8284 / % possible obs: 96.9 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 3.9 % / Biso Wilson estimate: 30.8 Å2 / Rmerge(I) obs: 0.102 / Net I/σ(I): 8.8 |
| Reflection shell | Resolution: 1.9→1.97 Å / Redundancy: 3.9 % / Rmerge(I) obs: 0.473 / Num. unique all: 817 / % possible all: 98 |
| Reflection | *PLUS Num. obs: 8283 |
| Reflection shell | *PLUS % possible obs: 98 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1EIS Resolution: 1.9→25 Å / σ(F): 0 / σ(I): 0 / Stereochemistry target values: Engh & Huber Details: The structure was determined by molecular replacement methods based on the uncomplexed UDA structure (1EIS). A bulk solvent correction was applied.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.9→25 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: REFMAC / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS % reflection Rfree: 5 % / Rfactor Rfree: 0.239 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller








 PDBj
PDBj



