[English] 日本語
 Yorodumi
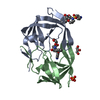
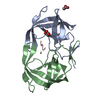
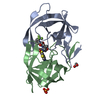
Yorodumi- PDB-1eay: CHEY-BINDING (P2) DOMAIN OF CHEA IN COMPLEX WITH CHEY FROM ESCHER... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1eay | ||||||
|---|---|---|---|---|---|---|---|
| Title | CHEY-BINDING (P2) DOMAIN OF CHEA IN COMPLEX WITH CHEY FROM ESCHERICHIA COLI | ||||||
 Components Components |
| ||||||
 Keywords Keywords | SIGNAL TRANSDUCTION COMPLEX / KINASE / RESPONSE REGULATOR / CHEMOTAXIS | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of protein modification process / methyl accepting chemotaxis protein complex / bacterial-type flagellum basal body, C ring / bacterial-type flagellum rotor complex / bacterial-type flagellum-dependent swimming motility / aerotaxis / regulation of bacterial-type flagellum-dependent cell motility / protein histidine kinase activity / regulation of chemotaxis / internal peptidyl-lysine acetylation ...negative regulation of protein modification process / methyl accepting chemotaxis protein complex / bacterial-type flagellum basal body, C ring / bacterial-type flagellum rotor complex / bacterial-type flagellum-dependent swimming motility / aerotaxis / regulation of bacterial-type flagellum-dependent cell motility / protein histidine kinase activity / regulation of chemotaxis / internal peptidyl-lysine acetylation / thermotaxis / bacterial-type flagellum / phosphorelay response regulator activity / protein acetylation / acetyltransferase activity / phosphorelay sensor kinase activity / histidine kinase / phosphorelay signal transduction system / establishment of localization in cell / chemotaxis / intracellular signal transduction / magnesium ion binding / signal transduction / ATP binding / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MIR, MIR,  MOLECULAR REPLACEMENT / Resolution: 2 Å MOLECULAR REPLACEMENT / Resolution: 2 Å | ||||||
 Authors Authors | Mcevoy, M.M. / Hausrath, A.C. / Randolph, G.B. / Remington, S.J. / Dahlquist, F.W. | ||||||
 Citation Citation |  Journal: Proc.Natl.Acad.Sci.USA / Year: 1998 Journal: Proc.Natl.Acad.Sci.USA / Year: 1998Title: Two binding modes reveal flexibility in kinase/response regulator interactions in the bacterial chemotaxis pathway. Authors: McEvoy, M.M. / Hausrath, A.C. / Randolph, G.B. / Remington, S.J. / Dahlquist, F.W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1eay.cif.gz 1eay.cif.gz | 86.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1eay.ent.gz pdb1eay.ent.gz | 66.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1eay.json.gz 1eay.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ea/1eay https://data.pdbj.org/pub/pdb/validation_reports/ea/1eay ftp://data.pdbj.org/pub/pdb/validation_reports/ea/1eay ftp://data.pdbj.org/pub/pdb/validation_reports/ea/1eay | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 13981.136 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #2: Protein | Mass: 8018.084 Da / Num. of mol.: 2 / Fragment: CHEY-BINDING (P2) DOMAIN / Source method: isolated from a natural source / Source: (natural)  References: UniProt: P07363, Transferases; Transferring phosphorus-containing groups; Phosphotransferases with a nitrogenous group as acceptor #3: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 2 X-RAY DIFFRACTION / Number of used crystals: 2 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.1 Å3/Da / Density % sol: 60 % | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 7 Details: PROTEIN WAS CRYSTALLIZED FROM .63 M NAH2PO4/1.17 M K2HPO4, 10 MM NH4CL, PH 7.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 287 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL9-1 / Wavelength: 0.98 / Beamline: BL9-1 / Wavelength: 0.98 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Apr 1, 1997 |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.98 Å / Relative weight: 1 |
| Reflection | Highest resolution: 2 Å / Num. obs: 30701 / % possible obs: 78 % / Redundancy: 2.5 % / Rmerge(I) obs: 0.055 |
| Reflection | *PLUS Num. measured all: 146840 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MIR, MIR,  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRIES 3CHY AND 1FWP Resolution: 2→20 Å / Isotropic thermal model: TNT BCORREL V1.0 / σ(F): 0 / Stereochemistry target values: TNT PROTGEO V1.0
| ||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: BABINET SCALING / Bsol: 150 Å2 / ksol: 0.7 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→20 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: TNT / Version: 5-F / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor all: 0.217 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller











 PDBj
PDBj



