[English] 日本語
 Yorodumi
Yorodumi- EMDB-9636: The structure of Enterovirus D68 A-particle in complex with Fab 11G1 -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9636 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | The structure of Enterovirus D68 A-particle in complex with Fab 11G1 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |   Enterovirus D68 Enterovirus D68 | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 7.2 Å | |||||||||
 Authors Authors | Zheng QB / Zhu R / Xu LF / He MZ / Yan XD / Cheng T / Li SW | |||||||||
 Citation Citation |  Journal: Nat Microbiol / Year: 2019 Journal: Nat Microbiol / Year: 2019Title: Atomic structures of enterovirus D68 in complex with two monoclonal antibodies define distinct mechanisms of viral neutralization. Authors: Qingbing Zheng / Rui Zhu / Longfa Xu / Maozhou He / Xiaodong Yan / Dongxiao Liu / Zhichao Yin / Yangtao Wu / Yongchao Li / Lisheng Yang / Wangheng Hou / Shuxuan Li / Zizhen Li / Zhenqin Chen ...Authors: Qingbing Zheng / Rui Zhu / Longfa Xu / Maozhou He / Xiaodong Yan / Dongxiao Liu / Zhichao Yin / Yangtao Wu / Yongchao Li / Lisheng Yang / Wangheng Hou / Shuxuan Li / Zizhen Li / Zhenqin Chen / Zhihai Li / Hai Yu / Ying Gu / Jun Zhang / Timothy S Baker / Z Hong Zhou / Barney S Graham / Tong Cheng / Shaowei Li / Ningshao Xia /   Abstract: Enterovirus D68 (EV-D68) undergoes structural transformation between mature, cell-entry intermediate (A-particle) and empty forms throughout its life cycle. Structural information for the various ...Enterovirus D68 (EV-D68) undergoes structural transformation between mature, cell-entry intermediate (A-particle) and empty forms throughout its life cycle. Structural information for the various forms and antibody-bound capsids will facilitate the development of effective vaccines and therapeutics against EV-D68 infection, which causes childhood respiratory and paralytic diseases worldwide. Here, we report the structures of three EV-D68 capsid states representing the virus at major phases. We further describe two original monoclonal antibodies (15C5 and 11G1) with distinct structurally defined mechanisms for virus neutralization. 15C5 and 11G1 engage the capsid loci at icosahedral three-fold and five-fold axes, respectively. To block viral attachment, 15C5 binds three forms of capsids, and triggers mature virions to transform into A-particles, mimicking engagement by the functional receptor ICAM-5, whereas 11G1 exclusively recognizes the A-particle. Our data provide a structural and molecular explanation for the transition of picornavirus capsid conformations and demonstrate distinct mechanisms for antibody-mediated neutralization. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9636.map.gz emd_9636.map.gz | 302.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9636-v30.xml emd-9636-v30.xml emd-9636.xml emd-9636.xml | 12 KB 12 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9636.png emd_9636.png | 181.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9636 http://ftp.pdbj.org/pub/emdb/structures/EMD-9636 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9636 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9636 | HTTPS FTP |
-Related structure data
| Related structure data |  9629C  9631C  9632C  9633C  9634C  9635C  6aj0C  6aj2C  6aj3C  6aj7C  6aj9C C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_9636.map.gz / Format: CCP4 / Size: 828.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9636.map.gz / Format: CCP4 / Size: 828.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.128 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
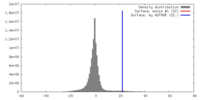
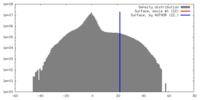
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Enterovirus D68
| Entire | Name:  Enterovirus D68 Enterovirus D68 |
|---|---|
| Components |
|
-Supramolecule #1: Enterovirus D68
| Supramolecule | Name: Enterovirus D68 / type: virus / ID: 1 / Parent: 0 / Macromolecule list: all / NCBI-ID: 42789 / Sci species name: Enterovirus D68 / Virus type: VIRION / Virus isolate: STRAIN / Virus enveloped: No / Virus empty: No |
|---|
-Macromolecule #1: VL of Fab 11G1
| Macromolecule | Name: VL of Fab 11G1 / type: other / ID: 1 / Classification: other |
|---|---|
| Source (natural) | Organism:  |
| Sequence | String: DIVMSQSPSS LAVSVGEKVT MSCKSSQSLL YSNNQKNYLA WYQQKPGQSP QLLIYWASTR ESGVPDRFTG SESGTDFTLT ISSVKAEDLA VFYCQQYYNY PWTFGGGTKL EIK |
-Macromolecule #2: VH of Fab 11G1
| Macromolecule | Name: VH of Fab 11G1 / type: other / ID: 2 / Classification: other |
|---|---|
| Source (natural) | Organism:  |
| Sequence | String: QVQLQQSGAE LVRPGVSVKI SCKGSGYIFT DYVMHWVKQS HSKSLEWIGV ISTYYGDGSY NQKFKGKATM TVDKSSSTAY MELARLTSED SAIYYCVRGE NYYGVSGTMD YWGQGTSVTV SS |
-Macromolecule #3: VP1
| Macromolecule | Name: VP1 / type: other / ID: 3 / Classification: other |
|---|---|
| Source (natural) | Organism:  Enterovirus D68 Enterovirus D68 |
| Sequence | String: IESIIKTATD TVKSEINAEL GVVPSLNAVE TGATSNTEPE EAIQTRTVIN QHGVSETLVE NFLSRAALVS KRSFEYKDHT SSAAQADKNF FKWTINTRSF VQLRRKLELF TYLRFDAEIT ILTTVAVNGS GNNTYVGLPD LTLQAMFVPT GALTPEKQDS FHWQSGSNAS ...String: IESIIKTATD TVKSEINAEL GVVPSLNAVE TGATSNTEPE EAIQTRTVIN QHGVSETLVE NFLSRAALVS KRSFEYKDHT SSAAQADKNF FKWTINTRSF VQLRRKLELF TYLRFDAEIT ILTTVAVNGS GNNTYVGLPD LTLQAMFVPT GALTPEKQDS FHWQSGSNAS VFFKISDPPA RITIPFMCIN SAYSVFYDGF AGFEKNGLYG INPADTIGNL CVRIVNEHQP VGFTVTVRVY MKPKHIKAWA PRPPRTLPYM SIANANYKGK ERAPNALNAI IGNRDSVKTM PHNIV |
-Macromolecule #4: VP2
| Macromolecule | Name: VP2 / type: other / ID: 4 / Classification: other |
|---|---|
| Source (natural) | Organism:  Enterovirus D68 Enterovirus D68 |
| Sequence | String: SPSAEACGYS DRVLQLKLGN SAIVTQEAAN YCCAYGEWPN YLPDHEAVAI DKPTQPETAT DRFYTLKSVK WETGSTGWWW KLPDALNNIG MFGQNVQHHY LYRSGFLIHV QCNATKFHQG ALLVVAIPEH QRGAHNTNTS PGFDDIMKGE EGGTFNHPYV LDDGTSLACA ...String: SPSAEACGYS DRVLQLKLGN SAIVTQEAAN YCCAYGEWPN YLPDHEAVAI DKPTQPETAT DRFYTLKSVK WETGSTGWWW KLPDALNNIG MFGQNVQHHY LYRSGFLIHV QCNATKFHQG ALLVVAIPEH QRGAHNTNTS PGFDDIMKGE EGGTFNHPYV LDDGTSLACA TIFPHQWINL RTNNSATIVL PWMNAAPMDF PLRHNQWTLA IIPVVPLGTR TTSSMVPITV SIAPMCCEFN GLRHAITQ |
-Macromolecule #5: VP3
| Macromolecule | Name: VP3 / type: other / ID: 5 / Classification: other |
|---|---|
| Source (natural) | Organism:  Enterovirus D68 Enterovirus D68 |
| Sequence | String: GVPTYLLPGS GQFLTTDDHS SAPALPCFNP TPEMHIPGQV RNMLEVVQVE SMMEINNTES AVGMERLKVD ISALTDVDQL LFNIPLDIQL DGPLRNTLVG NISRYYTHWS GSLEMTFMFC GSFMATGKLI LCYTPPGGSC PTTRETAMLG THIVWDFGLQ SSVTLIIPWI ...String: GVPTYLLPGS GQFLTTDDHS SAPALPCFNP TPEMHIPGQV RNMLEVVQVE SMMEINNTES AVGMERLKVD ISALTDVDQL LFNIPLDIQL DGPLRNTLVG NISRYYTHWS GSLEMTFMFC GSFMATGKLI LCYTPPGGSC PTTRETAMLG THIVWDFGLQ SSVTLIIPWI SGSHYRMFNN DAKSTNANVG YVTCFMQTNL IVPSESSDTC SLIGFIAAKD DFSLRLMRDS PDIGQLDHLH AAEAAYQ |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F30 |
|---|---|
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Average electron dose: 25.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Tecnai F30 / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Point group: I (icosahedral) / Resolution.type: BY AUTHOR / Resolution: 7.2 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 414 |
|---|---|
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: OTHER |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)