+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-8841 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | cryo-EM of a lysosomal protein | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | human TRPML1 / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of lysosome organization / calcium ion export / intracellularly phosphatidylinositol-3,5-bisphosphate-gated monatomic cation channel activity / phagosome maturation / NAADP-sensitive calcium-release channel activity / iron ion transmembrane transporter activity / iron ion transmembrane transport / Transferrin endocytosis and recycling / cellular response to pH / monoatomic anion channel activity ...positive regulation of lysosome organization / calcium ion export / intracellularly phosphatidylinositol-3,5-bisphosphate-gated monatomic cation channel activity / phagosome maturation / NAADP-sensitive calcium-release channel activity / iron ion transmembrane transporter activity / iron ion transmembrane transport / Transferrin endocytosis and recycling / cellular response to pH / monoatomic anion channel activity / intracellular zinc ion homeostasis / ligand-gated calcium channel activity / TRP channels / sodium channel activity / intracellular vesicle / autophagosome maturation / potassium channel activity / monoatomic cation transport / monoatomic cation channel activity / phagocytic cup / release of sequestered calcium ion into cytosol / cellular response to calcium ion / transferrin transport / phagocytic vesicle membrane / calcium ion transmembrane transport / calcium channel activity / late endosome / late endosome membrane / protein homotetramerization / adaptive immune response / lysosome / signaling receptor complex / endosome membrane / lysosomal membrane / lipid binding / Golgi apparatus / nucleoplasm / membrane / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.49 Å | |||||||||
 Authors Authors | Schmiege P / Li X | |||||||||
 Citation Citation |  Journal: Nature / Year: 2017 Journal: Nature / Year: 2017Title: Human TRPML1 channel structures in open and closed conformations. Authors: Philip Schmiege / Michael Fine / Günter Blobel / Xiaochun Li /  Abstract: Transient receptor potential mucolipin 1 (TRPML1) is a Ca-releasing cation channel that mediates the calcium signalling and homeostasis of lysosomes. Mutations in TRPML1 lead to mucolipidosis type ...Transient receptor potential mucolipin 1 (TRPML1) is a Ca-releasing cation channel that mediates the calcium signalling and homeostasis of lysosomes. Mutations in TRPML1 lead to mucolipidosis type IV, a severe lysosomal storage disorder. Here we report two electron cryo-microscopy structures of full-length human TRPML1: a 3.72-Å apo structure at pH 7.0 in the closed state, and a 3.49-Å agonist-bound structure at pH 6.0 in an open state. Several aromatic and hydrophobic residues in pore helix 1, helices S5 and S6, and helix S6 of a neighbouring subunit, form a hydrophobic cavity to house the agonist, suggesting a distinct agonist-binding site from that found in TRPV1, a TRP channel from a different subfamily. The opening of TRPML1 is associated with distinct dilations of its lower gate together with a slight structural movement of pore helix 1. Our work reveals the regulatory mechanism of TRPML channels, facilitates better understanding of TRP channel activation, and provides insights into the molecular basis of mucolipidosis type IV pathogenesis. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_8841.map.gz emd_8841.map.gz | 95.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-8841-v30.xml emd-8841-v30.xml emd-8841.xml emd-8841.xml | 9.6 KB 9.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_8841.png emd_8841.png | 59.5 KB | ||
| Filedesc metadata |  emd-8841.cif.gz emd-8841.cif.gz | 5.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-8841 http://ftp.pdbj.org/pub/emdb/structures/EMD-8841 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8841 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8841 | HTTPS FTP |
-Related structure data
| Related structure data |  5wj9MC  8840C  5wj5C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_8841.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_8841.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : TRPML1
| Entire | Name: TRPML1 |
|---|---|
| Components |
|
-Supramolecule #1: TRPML1
| Supramolecule | Name: TRPML1 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Mucolipin-1
| Macromolecule | Name: Mucolipin-1 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 65.084996 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MTAPAGPRGS ETERLLTPNP GYGTQAGPSP APPTPPEEED LRRRLKYFFM SPCDKFRAKG RKPCKLMLQV VKILVVTVQL ILFGLSNQL AVTFREENTI AFRHLFLLGY SDGADDTFAA YTREQLYQAI FHAVDQYLAL PDVSLGRYAY VRGGGDPWTN G SGLALCQR ...String: MTAPAGPRGS ETERLLTPNP GYGTQAGPSP APPTPPEEED LRRRLKYFFM SPCDKFRAKG RKPCKLMLQV VKILVVTVQL ILFGLSNQL AVTFREENTI AFRHLFLLGY SDGADDTFAA YTREQLYQAI FHAVDQYLAL PDVSLGRYAY VRGGGDPWTN G SGLALCQR YYHRGHVDPA NDTFDIDPMV VTDCIQVDPP ERPPPPPSDD LTLLESSSSY KNLTLKFHKL VNVTIHFRLK TI NLQSLIN NEIPDCYTFS VLITFDNKAH SGRIPISLET QAHIQECKHP SVFQHGDNSF RLLFDVVVIL TCSLSFLLCA RSL LRGFLL QNEFVGFMWR QRGRVISLWE RLEFVNGWYI LLVTSDVLTI SGTIMKIGIE AKNLASYDVC SILLGTSTLL VWVG VIRYL TFFHNYNILI ATLRVALPSV MRFCCCVAVI YLGYCFCGWI VLGPYHVKFR SLSMVSECLF SLINGDDMFV TFAAM QAQQ GRSSLVWLFS QLYLYSFISL FIYMVLSLFI ALITGAYDTI KHPGGAGAEE SELQAYIAQC QDSPTSGKFR RGSGSA CSL LCCCGRDPSE EHSLLVN UniProtKB: Mucolipin-1 |
-Macromolecule #2: 2-{2-oxo-2-[(4S)-2,2,4-trimethyl-3,4-dihydroquinolin-1(2H)-yl]eth...
| Macromolecule | Name: 2-{2-oxo-2-[(4S)-2,2,4-trimethyl-3,4-dihydroquinolin-1(2H)-yl]ethyl}-1H-isoindole-1,3(2H)-dione type: ligand / ID: 2 / Number of copies: 4 / Formula: AQV |
|---|---|
| Molecular weight | Theoretical: 362.422 Da |
| Chemical component information | 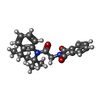 ChemComp-AQV: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 7 mg/mL |
|---|---|
| Buffer | pH: 7 |
| Vitrification | Cryogen name: NITROGEN |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 1.6 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: EMDB MAP EMDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.49 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: FREALIGN / Number images used: 56105 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION |
| Final angle assignment | Type: ANGULAR RECONSTITUTION |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)