+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7533 | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Yeast RNA polymerase III elongation complex | |||||||||||||||||||||
 Map data Map data | Yeast RNA polymerase III elongation complex | |||||||||||||||||||||
 Sample Sample |
| |||||||||||||||||||||
 Keywords Keywords | RNA polymerase III / TFIIIB / tRNA / TRANSCRIPTION / transcription-dna complex | |||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationRNA polymerase III core binding / TFIIA-class transcription factor complex binding / DNA-templated transcription open complex formation / RNA polymerase III transcription regulatory region sequence-specific DNA binding / RNA polymerase III preinitiation complex assembly / transcription factor TFIIIB complex / TFIIIC-class transcription factor complex binding / RNA polymerase III type 3 promoter sequence-specific DNA binding / regulation of transcription by RNA polymerase III / RNA polymerase III general transcription initiation factor activity ...RNA polymerase III core binding / TFIIA-class transcription factor complex binding / DNA-templated transcription open complex formation / RNA polymerase III transcription regulatory region sequence-specific DNA binding / RNA polymerase III preinitiation complex assembly / transcription factor TFIIIB complex / TFIIIC-class transcription factor complex binding / RNA polymerase III type 3 promoter sequence-specific DNA binding / regulation of transcription by RNA polymerase III / RNA polymerase III general transcription initiation factor activity / RNA polymerase I general transcription initiation factor binding / nuclear DNA-directed RNA polymerase complex / transcription factor TFIIA complex / RNA polymerase I preinitiation complex assembly / RNA Polymerase I Transcription Initiation / DNA binding, bending / transcription preinitiation complex / Processing of Capped Intron-Containing Pre-mRNA / RNA Polymerase III Transcription Initiation From Type 2 Promoter / RNA Pol II CTD phosphorylation and interaction with CE / Formation of the Early Elongation Complex / mRNA Capping / RNA polymerase II transcribes snRNA genes / TP53 Regulates Transcription of DNA Repair Genes / RNA Polymerase II Promoter Escape / RNA Polymerase II Transcription Pre-Initiation And Promoter Opening / RNA Polymerase II Transcription Initiation / RNA Polymerase II Transcription Initiation And Promoter Clearance / RNA Polymerase II Pre-transcription Events / termination of RNA polymerase III transcription / RNA-templated transcription / transcription factor TFIID complex / RNA polymerase II general transcription initiation factor activity / Formation of TC-NER Pre-Incision Complex / RNA Polymerase I Promoter Escape / transcription initiation at RNA polymerase III promoter / termination of RNA polymerase I transcription / Gap-filling DNA repair synthesis and ligation in TC-NER / transcription initiation at RNA polymerase I promoter / nucleolar large rRNA transcription by RNA polymerase I / Estrogen-dependent gene expression / transcription by RNA polymerase III / Dual incision in TC-NER / RNA polymerase II core promoter sequence-specific DNA binding / RNA polymerase I complex / RNA polymerase III complex / RNA polymerase II preinitiation complex assembly / transcription elongation by RNA polymerase I / RNA polymerase II, core complex / tRNA transcription by RNA polymerase III / transcription by RNA polymerase I / nucleotidyltransferase activity / TBP-class protein binding / transcription initiation at RNA polymerase II promoter / DNA-templated transcription initiation / transcription elongation by RNA polymerase II / ribonucleoside binding / DNA-directed RNA polymerase / disordered domain specific binding / DNA-directed RNA polymerase activity / peroxisome / single-stranded DNA binding / ribosome biogenesis / transcription regulator complex / transcription by RNA polymerase II / DNA-binding transcription factor binding / nucleic acid binding / RNA polymerase II-specific DNA-binding transcription factor binding / protein dimerization activity / nucleotide binding / negative regulation of DNA-templated transcription / chromatin binding / regulation of DNA-templated transcription / nucleolus / positive regulation of transcription by RNA polymerase II / protein-containing complex / mitochondrion / DNA binding / zinc ion binding / nucleoplasm / metal ion binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||||||||||||||
| Biological species |   | |||||||||||||||||||||
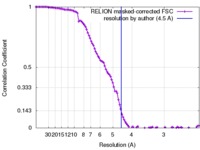
| Method | single particle reconstruction / cryo EM / Resolution: 4.5 Å | |||||||||||||||||||||
 Authors Authors | Han Y / He Y | |||||||||||||||||||||
| Funding support |  United States, 6 items United States, 6 items
| |||||||||||||||||||||
 Citation Citation |  Journal: Cell Discov / Year: 2018 Journal: Cell Discov / Year: 2018Title: Structural visualization of RNA polymerase III transcription machineries. Authors: Yan Han / Chunli Yan / Susan Fishbain / Ivaylo Ivanov / Yuan He /  Abstract: RNA polymerase III (Pol III) transcription initiation requires the action of the transcription factor IIIB (TFIIIB) and is highly regulated. Here, we determine the structures of Pol III pre- ...RNA polymerase III (Pol III) transcription initiation requires the action of the transcription factor IIIB (TFIIIB) and is highly regulated. Here, we determine the structures of Pol III pre-initiation complexes (PICs) using single particle cryo-electron microscopy (cryo-EM). We observe stable Pol III-TFIIIB complexes using nucleic acid scaffolds mimicking various functional states, in which TFIIIB tightly encircles the upstream promoter DNA. There is an intricate interaction between TFIIIB and Pol III, which stabilizes the winged-helix domains of the C34 subunit of Pol III over the active site cleft. The architecture of Pol III PIC more resembles that of the Pol II PIC than the Pol I PIC. In addition, we also obtain a 3D reconstruction of Pol III in complex with TFIIIB using the elongation complex (EC) scaffold, shedding light on the mechanism of facilitated recycling of Pol III prior to transcription re-initiation. | |||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7533.map.gz emd_7533.map.gz | 54.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7533-v30.xml emd-7533-v30.xml emd-7533.xml emd-7533.xml | 39.6 KB 39.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_7533_fsc.xml emd_7533_fsc.xml | 10.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_7533.png emd_7533.png | 196.9 KB | ||
| Filedesc metadata |  emd-7533.cif.gz emd-7533.cif.gz | 11.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7533 http://ftp.pdbj.org/pub/emdb/structures/EMD-7533 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7533 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7533 | HTTPS FTP |
-Related structure data
| Related structure data |  6cnfMC  7530C  7531C  7532C  7534C  6cnbC  6cncC  6cndC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_7533.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7533.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Yeast RNA polymerase III elongation complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.18 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
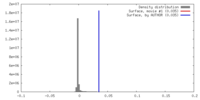
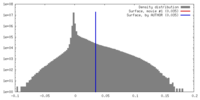
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Yeast RNA polymerase III elongation complex
+Supramolecule #1: Yeast RNA polymerase III elongation complex
+Macromolecule #1: DNA-directed RNA polymerase III subunit RPC1
+Macromolecule #2: DNA-directed RNA polymerase III subunit RPC2
+Macromolecule #3: DNA-directed RNA polymerases I and III subunit RPAC1
+Macromolecule #4: DNA-directed RNA polymerase III subunit RPC9
+Macromolecule #5: DNA-directed RNA polymerases I, II, and III subunit RPABC1
+Macromolecule #6: DNA-directed RNA polymerases I, II, and III subunit RPABC2
+Macromolecule #7: DNA-directed RNA polymerase III subunit RPC8
+Macromolecule #8: DNA-directed RNA polymerases I, II, and III subunit RPABC3
+Macromolecule #9: DNA-directed RNA polymerase III subunit RPC10
+Macromolecule #10: DNA-directed RNA polymerases I, II, and III subunit RPABC5
+Macromolecule #11: DNA-directed RNA polymerases I and III subunit RPAC2
+Macromolecule #12: DNA-directed RNA polymerases I, II, and III subunit RPABC4
+Macromolecule #13: DNA-directed RNA polymerase III subunit RPC5
+Macromolecule #14: DNA-directed RNA polymerase III subunit RPC4
+Macromolecule #15: DNA-directed RNA polymerase III subunit RPC3
+Macromolecule #16: DNA-directed RNA polymerase III subunit RPC6
+Macromolecule #17: DNA-directed RNA polymerase III subunit RPC7,DNA-directed RNA pol...
+Macromolecule #18: Transcription factor IIIB 70 kDa subunit,TATA-box-binding protein...
+Macromolecule #19: Transcription factor TFIIIB component B'',Transcription factor TF...
+Macromolecule #20: DNA (79-MER)
+Macromolecule #21: DNA (79-MER)
+Macromolecule #22: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.9 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | JEOL 3200FS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 68.9 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)