[English] 日本語
 Yorodumi
Yorodumi- EMDB-6807: Class3 cryoEM structure of human M-type phospholipase A2 receptor -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-6807 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Class3 cryoEM structure of human M-type phospholipase A2 receptor | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 10.0 Å | |||||||||
 Authors Authors | Dong Y / Cao L / Shi X / Tang H / He Y | |||||||||
 Citation Citation |  Journal: J Mol Biol / Year: 2017 Journal: J Mol Biol / Year: 2017Title: Structure of Human M-type Phospholipase A2 Receptor Revealed by Cryo-Electron Microscopy. Authors: Yue Dong / Longxing Cao / Hua Tang / Xiangyi Shi / Yongning He /  Abstract: M-type phospholipase A2 receptor (M-PLA2R) is a member of the mannose receptor family and known as the receptor of secretory phospholipase A2s. It has also been identified as the major autoantigen of ...M-type phospholipase A2 receptor (M-PLA2R) is a member of the mannose receptor family and known as the receptor of secretory phospholipase A2s. It has also been identified as the major autoantigen of idiopathic membranous nephropathy, one of the most common causes for nephrotic syndrome in adults. Here we determine the structure of human M-PLA2R ectodomain by cryo-electron microscopy. The results show that the ectodomain has high internal flexibility and forms a compact dual-ring-shaped conformation at acidic pH and adopts extended conformations at basic pH. The inter-domain interactions of human M-PLA2R are explored by the binding studies with individual domains, showing the mechanism of the conformational change. In addition, the biochemical data suggest that mouse M-PLA2R recognizes mouse secretory phospholipase A2-G1B only at physiological or basic pH, rather than at acidic pH. These results suggest that the pH-dependent conformational change might play important roles in the functional activities of M-PLA2R such as ligand binding and release, and may also be relevant to the immunogenicity in membranous nephropathy. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_6807.map.gz emd_6807.map.gz | 377.4 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-6807-v30.xml emd-6807-v30.xml emd-6807.xml emd-6807.xml | 13.7 KB 13.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_6807.png emd_6807.png | 33.2 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6807 http://ftp.pdbj.org/pub/emdb/structures/EMD-6807 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6807 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6807 | HTTPS FTP |
-Validation report
| Summary document |  emd_6807_validation.pdf.gz emd_6807_validation.pdf.gz | 78.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_6807_full_validation.pdf.gz emd_6807_full_validation.pdf.gz | 77.4 KB | Display | |
| Data in XML |  emd_6807_validation.xml.gz emd_6807_validation.xml.gz | 494 B | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6807 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6807 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6807 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6807 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_6807.map.gz / Format: CCP4 / Size: 432.6 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_6807.map.gz / Format: CCP4 / Size: 432.6 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.3 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
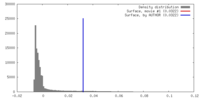
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : The ectodomain of human M-type phospholipase A2 receptor
| Entire | Name: The ectodomain of human M-type phospholipase A2 receptor |
|---|---|
| Components |
|
-Supramolecule #1: The ectodomain of human M-type phospholipase A2 receptor
| Supramolecule | Name: The ectodomain of human M-type phospholipase A2 receptor type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Experimental: 180 KDa |
| Recombinant expression | Organism:  |
-Macromolecule #1: PLA2R1
| Macromolecule | Name: PLA2R1 / type: other / ID: 1 / Classification: other |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MLLSPSLLLL LLLGAPRGCA EGVAAALTPE RLLEWQDKGI FVIQSESLKK CIQAGKSVLT LENCKQANKH MLWKWVSNH GLFNIGGSGC LGLNFSAPEQ PLSLYECDST LVSLRWRCNR KMITGPLQYS VQVAHDNTVV A SRKYIHKW ISYGSGGGDI CEYLHKDLHT ...String: MLLSPSLLLL LLLGAPRGCA EGVAAALTPE RLLEWQDKGI FVIQSESLKK CIQAGKSVLT LENCKQANKH MLWKWVSNH GLFNIGGSGC LGLNFSAPEQ PLSLYECDST LVSLRWRCNR KMITGPLQYS VQVAHDNTVV A SRKYIHKW ISYGSGGGDI CEYLHKDLHT IKGNTHGMPC MFPFQYNHQW HHECTREGRE DDLLWCATTS RY ERDEKWG FCPDPTSAEV GCDTIWEKDL NSHICYQFNL LSSLSWSEAH SSCQMQGGTL LSITDETEEN FIR EHMSSK TVEVWMGLNQ LDEHAGWQWS DGTPLNYLNW SPEVNFEPFV EDHCGTFSSF MPSAWRSRDC ESTL PYICK KYLNHIDHEI VEKDAWKYYA THCEPGWNPY NRNCYKLQKE EKTWHEALRS CQADNSALID ITSLA EVEF LVTLLGDENA SETWIGLSSN KIPVSFEWSN DSSVIFTNWH TLEPHIFPNR SQLCVSAEQS EGHWKV KNC EERLFYICKK AGHVLSDAES GCQEGWERHG GFCYKIDTVL RSFDQASSGY YCPPALVTIT NRFEQAF IT SLISSVVKMK DSYFWIALQD QNDTGEYTWK PVGQKPEPVQ YTHWNTHQPR YSGGCVAMRG RHPLGRWE V KHCRHFKAMS LCKQPVENQE KAEYEERWPF HPCYLDWESE PGLASCFKVF HSEKVLMKRT WREAEAFCE EFGAHLASFA HIEEENFVNE LLHSKFNWTE ERQFWIGFNK RNPLNAGSWE WSDRTPVVSS FLDNTYFGED ARNCAVYKA NKTLLPLHCG SKREWICKIP RDVKPKIPFW YQYDVPWLFY QDAEYLFHTF ASEWLNFEFV C SWLHSDLL TIHSAHEQEF IHSKIKALSK YGASWWIGLQ EERANDEFRW RDGTPVIYQN WDTGRERTVN NQ SQRCGFI SSITGLWGSE ECSVSMPSIC KRKKVWLIEK KKDTPKQHGT CPKGWLYFNY KCLLLNIPKD PSS WKNWTH AQHFCAEEGG TLVAIESEVE QAFITMNLFG QTTSVWIGLQ NDDYETWLNG KPVVYSNWSP FDII NIPSH NTTEVQKHIP LCALLSSNPN FHFTGKWYFE DCGKEGYGFV CEKMQDTSGH GVNTSDMYPM PNTLE YGNR TYKIINANMT WYAAIKTCLM HKAQLVSITD QYHQSFLTVV LNRLGYAHWI GLFTTDNGLN FDWSDG TKS SFTFWKDEES SLLGDCVFAD SNGRWHSTAC ESFLQGAICH VPPETRQSEH PELCSETSIP WIKFKSN CY SFSTVLDSMS FEAAHEFCKK EGSNLLTIKD EAENAFLLEE LFAFGSSVQM VWLNAQFDGN NETIKWFD G TPTDQSNWGI RKPDTDYFKP HHCVALRIPE GLWQLSPCQE KKGFICKMEA DIHTAEALPE KG |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.3 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 6 Component:
| |||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: OTHER | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 283.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 14 pixel / Digitization - Dimensions - Height: 14 pixel / Digitization - Frames/image: 4-37 / Average exposure time: 0.2 sec. / Average electron dose: 1.25 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: OTHER |
|---|
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)