+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-6792 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of p300 | |||||||||
 Map data Map data | Cryo-EM structure of p300. Transcriptional coactivator p300 is a large, multidomain protein that possess (HAT) histone acetyltransferase activity. | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationbehavioral defense response / negative regulation of protein oligomerization / peptidyl-lysine propionylation / histone lactyltransferase (CoA-dependent) activity / peptidyl-lysine crotonylation / peptidyl-lysine butyrylation / histone butyryltransferase activity / histone isonicotinyltransferase activity / histone H1K75 acetyltransferase activity / swimming ...behavioral defense response / negative regulation of protein oligomerization / peptidyl-lysine propionylation / histone lactyltransferase (CoA-dependent) activity / peptidyl-lysine crotonylation / peptidyl-lysine butyrylation / histone butyryltransferase activity / histone isonicotinyltransferase activity / histone H1K75 acetyltransferase activity / swimming / histone H3K122 acetyltransferase activity / internal protein amino acid acetylation / peptide butyryltransferase activity / regulation of tubulin deacetylation / histone H2B acetyltransferase activity / peptide 2-hydroxyisobutyryltransferase activity / histone crotonyltransferase activity / NOTCH2 intracellular domain regulates transcription / protein propionyltransferase activity / thigmotaxis / L-lysine N6-acetyltransferase activity, acting on acetyl phosphate as donor / positive regulation of TORC2 signaling / internal peptidyl-lysine acetylation / histone H4 acetyltransferase activity / histone H3 acetyltransferase activity / negative regulation of chromosome condensation / NFE2L2 regulating ER-stress associated genes / cellular response to L-leucine / acetylation-dependent protein binding / NFE2L2 regulating inflammation associated genes / Activation of the TFAP2 (AP-2) family of transcription factors / NGF-stimulated transcription / histone H3K18 acetyltransferase activity / N-terminal peptidyl-lysine acetylation / histone H3K27 acetyltransferase activity / LRR FLII-interacting protein 1 (LRRFIP1) activates type I IFN production / NFE2L2 regulates pentose phosphate pathway genes / NFE2L2 regulating MDR associated enzymes / STAT3 nuclear events downstream of ALK signaling / Polo-like kinase mediated events / negative regulation of brown fat cell differentiation / regulation of mitochondrion organization / regulation of androgen receptor signaling pathway / host-mediated activation of viral transcription / TGFBR3 expression / Regulation of gene expression in late stage (branching morphogenesis) pancreatic bud precursor cells / Regulation of FOXO transcriptional activity by acetylation / RUNX3 regulates NOTCH signaling / NOTCH4 Intracellular Domain Regulates Transcription / Regulation of NFE2L2 gene expression / Regulation of gene expression by Hypoxia-inducible Factor / Nuclear events mediated by NFE2L2 / face morphogenesis / platelet formation / NOTCH3 Intracellular Domain Regulates Transcription / TRAF6 mediated IRF7 activation / regulation of glycolytic process / NFE2L2 regulating tumorigenic genes / NFE2L2 regulating anti-oxidant/detoxification enzymes / protein acetylation / megakaryocyte development / nuclear androgen receptor binding / acyltransferase activity / STAT family protein binding / Formation of paraxial mesoderm / acetyltransferase activity / histone acetyltransferase activity / FOXO-mediated transcription of cell death genes / stimulatory C-type lectin receptor signaling pathway / positive regulation of transforming growth factor beta receptor signaling pathway / Zygotic genome activation (ZGA) / PI5P Regulates TP53 Acetylation / DNA repair-dependent chromatin remodeling / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / histone acetyltransferase complex / : / RUNX3 regulates p14-ARF / intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator / negative regulation of gluconeogenesis / pre-mRNA intronic binding / canonical Wnt signaling pathway / Attenuation phase / protein-lysine-acetyltransferase activity / positive regulation of T-helper 17 cell lineage commitment / somitogenesis / NF-kappaB binding / fat cell differentiation / histone acetyltransferase / negative regulation of protein-containing complex assembly / skeletal muscle tissue development / NR1H3 & NR1H2 regulate gene expression linked to cholesterol transport and efflux / regulation of cellular response to heat / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / Regulation of TP53 Activity through Acetylation / canonical NF-kappaB signal transduction / Transcriptional and post-translational regulation of MITF-M expression and activity / positive regulation of TORC1 signaling / Transferases; Acyltransferases; Transferring groups other than aminoacyl groups / SUMOylation of transcription cofactors / RORA,B,C and NR1D1 (REV-ERBA) regulate gene expression Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
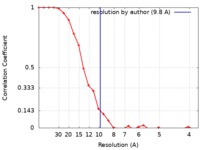
| Method | single particle reconstruction / cryo EM / Resolution: 9.8 Å | |||||||||
 Authors Authors | Ghosh R / Roy S / Sengupta J | |||||||||
 Citation Citation |  Journal: Biochemistry / Year: 2019 Journal: Biochemistry / Year: 2019Title: Tumor Suppressor p53-Mediated Structural Reorganization of the Transcriptional Coactivator p300. Authors: Raka Ghosh / Stephanie Kaypee / Manidip Shasmal / Tapas K Kundu / Siddhartha Roy / Jayati Sengupta /  Abstract: Transcriptional coactivator p300, a critical player in eukaryotic gene regulation, primarily functions as a histone acetyltransferase (HAT). It is also an important player in acetylation of a number ...Transcriptional coactivator p300, a critical player in eukaryotic gene regulation, primarily functions as a histone acetyltransferase (HAT). It is also an important player in acetylation of a number of nonhistone proteins, p53 being the most prominent one. Recruitment of p300 to p53 is pivotal in the regulation of p53-dependent genes. Emerging evidence suggests that p300 adopts an active conformation upon binding to the tetrameric p53, resulting in its enhanced acetylation activity. As a modular protein, p300 consists of multiple well-defined domains, where the structured domains are interlinked with unstructured linker regions. A crystal structure of the central domain of p300 encompassing Bromo, RING, PHD, and HAT domains demonstrates a compact module, where the HAT active site stays occluded by the RING domain. However, although p300 has a significant role in mediating the transcriptional activity of p53, only a few structural details on the complex of these two full-length proteins are available. Here, we present a cryo-electron microscopy (cryo-EM) study on the p300-p53 complex. The three-dimensional cryo-EM density map of the p300-p53 complex, when compared to the cryo-EM map of free p300, revealed that substantial change in the relative arrangement of Bromo and HAT domains occurs upon complex formation, which is likely required for exposing HAT active site and subsequent acetyltransferase activity. Our observation correlates well with previous studies showing that the presence of Bromodomain is obligatory for effective acetyltransferase activity of HAT. Thus, our result sheds new light on the mechanism whereby p300, following binding with p53, gets activated. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_6792.map.gz emd_6792.map.gz | 951.5 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-6792-v30.xml emd-6792-v30.xml emd-6792.xml emd-6792.xml | 13.1 KB 13.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_6792_fsc.xml emd_6792_fsc.xml | 2.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_6792.png emd_6792.png | 122.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6792 http://ftp.pdbj.org/pub/emdb/structures/EMD-6792 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6792 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6792 | HTTPS FTP |
-Related structure data
| Related structure data |  6k4nMC  6791C  5xzcC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_6792.map.gz / Format: CCP4 / Size: 1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_6792.map.gz / Format: CCP4 / Size: 1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of p300. Transcriptional coactivator p300 is a large, multidomain protein that possess (HAT) histone acetyltransferase activity. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.89 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : p300 protein
| Entire | Name: p300 protein |
|---|---|
| Components |
|
-Supramolecule #1: p300 protein
| Supramolecule | Name: p300 protein / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  |
-Macromolecule #1: Histone acetyltransferase p300
| Macromolecule | Name: Histone acetyltransferase p300 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: histone acetyltransferase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 72.100062 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: KKIFKPEELR QALMPTLEAL YRQDPESLPF RQPVDPQLLG IPDYFDIVKS PMDLSTIKRK LDTGQYQEPW QYVDDIWLMF NNAWLYNRK TSRVYKYCSK LSEVFEQEID PVMQSLGYCC GRKLEFSPQT LCCYGKQLCT IPRDATYYSY QNRYHFCEKC F NEIQGESV ...String: KKIFKPEELR QALMPTLEAL YRQDPESLPF RQPVDPQLLG IPDYFDIVKS PMDLSTIKRK LDTGQYQEPW QYVDDIWLMF NNAWLYNRK TSRVYKYCSK LSEVFEQEID PVMQSLGYCC GRKLEFSPQT LCCYGKQLCT IPRDATYYSY QNRYHFCEKC F NEIQGESV SLGDDPSQPQ TTINKEQFSK RKNDTLDPEL FVECTECGRK MHQICVLHHE IIWPAGFVCD GCLKKSARTR KE NKFSAKR LPSTRLGTFL ENRVNDFLRR QNHPESGEVT VRVVHASDKT VEVKPGMKAR FVDSGEMAES FPYRTKALFA FEE IDGVDL CFFGMHVQEY GSDCPPPNQR RVYISYLDSV HFFRPKCLRT AVYHEILIGY LEYVKKLGYT TGHIWACPPS EGDD YIFHC HPPDQKIPKP KRLQEWYKKM LDKAVSERIV HDYKDIFKQA TEDRLTSAKE LPYFEGDFWP NVLEESIKEL EQEEE ERKR EENTSNESTD VTKGDSKNAK KKNNKKTSKN KSSLSRGNKK KPGMPNVSND LSQKLYATME KHKEVFFVIR LIAGPA ANS LPPIVDPDPL IPCDLMDGRD AFLTLARDKH LEFSSLRRAQ WSTMCMLVEL HTQSQD |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 Component:
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 279 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Image recording | Film or detector model: FEI EAGLE (4k x 4k) / Average exposure time: 1.0 sec. / Average electron dose: 15.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 150.0 µm / Calibrated defocus max: 4.5 µm / Calibrated defocus min: 1.5 µm / Calibrated magnification: 78894 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 4.5 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 59000 |
| Sample stage | Specimen holder model: GATAN 910 MULTI-SPECIMEN SINGLE TILT CRYO TRANSFER HOLDER Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)