+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
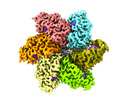
| Title | CryoEM structure of LTag bound to SV40 EP half origin DNA | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Helicase / ATPase / EP half origin / AMP-PNP / DNA replication / TRANSLOCASE | |||||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont-mediated suppression of host JAK-STAT cascade via inhibition of JAK1 activity / bidirectional double-stranded viral DNA replication / viral DNA genome replication / symbiont-mediated perturbation of host cell cycle G1/S transition checkpoint / 3'-5' DNA helicase activity / DNA 3'-5' helicase / DNA replication origin binding / single-stranded DNA binding / double-stranded DNA binding / symbiont-mediated perturbation of host ubiquitin-like protein modification ...symbiont-mediated suppression of host JAK-STAT cascade via inhibition of JAK1 activity / bidirectional double-stranded viral DNA replication / viral DNA genome replication / symbiont-mediated perturbation of host cell cycle G1/S transition checkpoint / 3'-5' DNA helicase activity / DNA 3'-5' helicase / DNA replication origin binding / single-stranded DNA binding / double-stranded DNA binding / symbiont-mediated perturbation of host ubiquitin-like protein modification / DNA replication / symbiont-mediated suppression of host innate immune response / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / host cell nucleus / ATP hydrolysis activity / zinc ion binding / ATP binding / identical protein binding Similarity search - Function | |||||||||
| Biological species |  Betapolyomavirus macacae / synthetic construct (others) Betapolyomavirus macacae / synthetic construct (others) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Danazumi AU / Shahid T / Tehseen M / Alhudhali L / Clark A / Savva CG / Hamdan SM / De Biasio A | |||||||||
| Funding support |  Saudi Arabia, 1 items Saudi Arabia, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2025 Journal: Nature / Year: 2025Title: Structural dynamics of DNA unwinding by a replicative helicase. Authors: Taha Shahid / Ammar U Danazumi / Muhammad Tehseen / Lubna Alhudhali / Alice R Clark / Christos G Savva / Samir M Hamdan / Alfredo De Biasio /   Abstract: Hexameric helicases are nucleotide-driven molecular machines that unwind DNA to initiate replication across all domains of life. Despite decades of intensive study, several critical aspects of their ...Hexameric helicases are nucleotide-driven molecular machines that unwind DNA to initiate replication across all domains of life. Despite decades of intensive study, several critical aspects of their function remain unresolved: the site and mechanism of DNA strand separation, the mechanics of unwinding propagation, and the dynamic relationship between nucleotide hydrolysis and DNA movement. Here, using cryo-electron microscopy (cryo-EM), we show that the simian virus 40 large tumour antigen (LTag) helicase assembles in the form of head-to-head hexamers at replication origins, melting DNA at two symmetrically positioned sites to establish bidirectional replication forks. Through continuous heterogeneity analysis, we characterize the conformational landscape of LTag on forked DNA under catalytic conditions, demonstrating coordinated motions that drive DNA translocation and unwinding. We show that the helicase pulls the tracking strand through DNA-binding loops lining the central channel, while directing the non-tracking strand out of the rear, in a cyclic process. ATP hydrolysis functions as an 'entropy switch', removing blocks to translocation rather than directly powering DNA movement. Our structures show the allosteric couplings between nucleotide turnover and subunit motions that enable DNA unwinding while maintaining dedicated exit paths for the separated strands. These findings provide a comprehensive model for replication fork establishment and progression that extends from viral to eukaryotic systems. More broadly, they introduce fundamental principles of the mechanism by which ATP-dependent enzymes achieve efficient mechanical work through entropy-driven allostery. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_62206.map.gz emd_62206.map.gz | 87.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-62206-v30.xml emd-62206-v30.xml emd-62206.xml emd-62206.xml | 23.7 KB 23.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_62206_fsc.xml emd_62206_fsc.xml | 11.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_62206.png emd_62206.png | 102.6 KB | ||
| Masks |  emd_62206_msk_1.map emd_62206_msk_1.map | 125 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-62206.cif.gz emd-62206.cif.gz | 6.6 KB | ||
| Others |  emd_62206_additional_1.map.gz emd_62206_additional_1.map.gz emd_62206_half_map_1.map.gz emd_62206_half_map_1.map.gz emd_62206_half_map_2.map.gz emd_62206_half_map_2.map.gz | 62.4 MB 116.1 MB 116.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-62206 http://ftp.pdbj.org/pub/emdb/structures/EMD-62206 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-62206 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-62206 | HTTPS FTP |
-Related structure data
| Related structure data |  9kaeMC  9evhC  9evpC  9exdC  9f3tC  9f3uC  9f5iC  9f73C  9f74C  9f75C  9f7nC  9f9nC  9f9oC  9f9wC  9f9xC  9fa1C  9fa2C  9fb0C  9fb4C  9fb5C  9fb6C  9kakC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_62206.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_62206.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.93 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_62206_msk_1.map emd_62206_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: #1
| File | emd_62206_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_62206_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_62206_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of SV40 LTag with SV40 EP half origin
| Entire | Name: Complex of SV40 LTag with SV40 EP half origin |
|---|---|
| Components |
|
-Supramolecule #1: Complex of SV40 LTag with SV40 EP half origin
| Supramolecule | Name: Complex of SV40 LTag with SV40 EP half origin / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|
-Supramolecule #2: SV40 LTag
| Supramolecule | Name: SV40 LTag / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Betapolyomavirus macacae Betapolyomavirus macacae |
-Supramolecule #3: DNA
| Supramolecule | Name: DNA / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2-#3 |
|---|---|
| Source (natural) | Organism: synthetic construct (others) / Synthetically produced: Yes |
-Macromolecule #1: Large T antigen
| Macromolecule | Name: Large T antigen / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO / EC number: DNA 3'-5' helicase |
|---|---|
| Source (natural) | Organism:  Betapolyomavirus macacae Betapolyomavirus macacae |
| Molecular weight | Theoretical: 41.812664 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: KQVSWKLVTE YAMETKCDDV LLLLGMYLEF QYSFEMCLKC IKKEQPSHYK YHEKHYANAA IFADSKNQKT ICQQAVDTVL AKKRVDSLQ LTREQMLTNR FNDLLDRMDI MFGSTGSADI EEWMAGVAWL HCLLPKMDSV VYDFLKCMVY NIPKKRYWLF K GPIDSGKT ...String: KQVSWKLVTE YAMETKCDDV LLLLGMYLEF QYSFEMCLKC IKKEQPSHYK YHEKHYANAA IFADSKNQKT ICQQAVDTVL AKKRVDSLQ LTREQMLTNR FNDLLDRMDI MFGSTGSADI EEWMAGVAWL HCLLPKMDSV VYDFLKCMVY NIPKKRYWLF K GPIDSGKT TLAAALLELC GGKALNVNLP LDRLNFELGV AIDQFLVVFE DVKGTGGESR DLPSGQGINN LDNLRDYLDG SV KVNLEKK HLNKRTQIFP PGIVTMNEYS VPKTLQARFV KQIDFRPKDY LKHCLERSEF LLEKRIIQSG IALLLMLIWY RPV AEFAQS IQSRIVEWKE RLDKEFSLSV YQKMKFNVAM GIGVLD UniProtKB: Large T antigen |
-Macromolecule #2: DNA
| Macromolecule | Name: DNA / type: dna / ID: 2 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 3.08711 KDa |
| Sequence | String: (DA)(DA)(DA)(DA)(DA)(DA)(DA)(DA)(DA)(DA) |
-Macromolecule #3: DNA
| Macromolecule | Name: DNA / type: dna / ID: 3 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 4.517935 KDa |
| Sequence | String: (DT)(DT)(DT)(DT)(DT)(DT)(DT)(DT)(DT)(DT) (DT)(DT)(DT)(DT)(DT) |
-Macromolecule #4: PHOSPHOAMINOPHOSPHONIC ACID-ADENYLATE ESTER
| Macromolecule | Name: PHOSPHOAMINOPHOSPHONIC ACID-ADENYLATE ESTER / type: ligand / ID: 4 / Number of copies: 6 / Formula: ANP |
|---|---|
| Molecular weight | Theoretical: 506.196 Da |
| Chemical component information |  ChemComp-ANP: |
-Macromolecule #5: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 5 / Number of copies: 6 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.5 mg/mL |
|---|---|
| Buffer | pH: 8 |
| Grid | Model: Quantifoil R2/2 / Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average exposure time: 5.0 sec. / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.6 µm / Nominal defocus min: 1.1 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: FLEXIBLE FIT |
|---|---|
| Output model |  PDB-9kae: |
 Movie
Movie Controller
Controller










































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)