+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-6076 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Reconstruction of the N-terminal domain of human TFG | |||||||||
 Map data Map data | Reconstruction of the N-terminal domain of human TFG | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | TFG / COPII / secretory pathway | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / negative staining / Resolution: 30.0 Å | |||||||||
 Authors Authors | Johnson A / Bhattacharya N / Hanna M / Schuh AL / Pennington JG / Otegui MS / Stagg SM / Audhya A | |||||||||
 Citation Citation |  Journal: EMBO J / Year: 2015 Journal: EMBO J / Year: 2015Title: TFG clusters COPII-coated transport carriers and promotes early secretory pathway organization. Authors: Adam Johnson / Nilakshee Bhattacharya / Michael Hanna / Janice G Pennington / Amber L Schuh / Lei Wang / Marisa S Otegui / Scott M Stagg / Anjon Audhya /  Abstract: In mammalian cells, cargo-laden secretory vesicles leave the endoplasmic reticulum (ER) en route to ER-Golgi intermediate compartments (ERGIC) in a manner dependent on the COPII coat complex. We ...In mammalian cells, cargo-laden secretory vesicles leave the endoplasmic reticulum (ER) en route to ER-Golgi intermediate compartments (ERGIC) in a manner dependent on the COPII coat complex. We report here that COPII-coated transport carriers traverse a submicron, TFG (Trk-fused gene)-enriched zone at the ER/ERGIC interface. The architecture of TFG complexes as determined by three-dimensional electron microscopy reveals the formation of flexible, octameric cup-like structures, which are able to self-associate to generate larger polymers in vitro. In cells, loss of TFG function dramatically slows protein export from the ER and results in the accumulation of COPII-coated carriers throughout the cytoplasm. Additionally, the tight association between ER and ERGIC membranes is lost in the absence of TFG. We propose that TFG functions at the ER/ERGIC interface to locally concentrate COPII-coated transport carriers and link exit sites on the ER to ERGIC membranes. Our findings provide a new mechanism by which COPII-coated carriers are retained near their site of formation to facilitate rapid fusion with neighboring ERGIC membranes upon uncoating, thereby promoting interorganellar cargo transport. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_6076.map.gz emd_6076.map.gz | 5.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-6076-v30.xml emd-6076-v30.xml emd-6076.xml emd-6076.xml | 8.1 KB 8.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_6076.jpg emd_6076.jpg | 26.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6076 http://ftp.pdbj.org/pub/emdb/structures/EMD-6076 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6076 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6076 | HTTPS FTP |
-Validation report
| Summary document |  emd_6076_validation.pdf.gz emd_6076_validation.pdf.gz | 78.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_6076_full_validation.pdf.gz emd_6076_full_validation.pdf.gz | 77.3 KB | Display | |
| Data in XML |  emd_6076_validation.xml.gz emd_6076_validation.xml.gz | 495 B | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6076 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6076 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6076 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6076 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_6076.map.gz / Format: CCP4 / Size: 11.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_6076.map.gz / Format: CCP4 / Size: 11.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Reconstruction of the N-terminal domain of human TFG | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.5 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
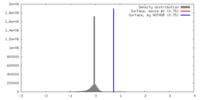
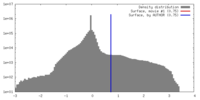
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : N-terminal domain of human TFG
| Entire | Name: N-terminal domain of human TFG |
|---|---|
| Components |
|
-Supramolecule #1000: N-terminal domain of human TFG
| Supramolecule | Name: N-terminal domain of human TFG / type: sample / ID: 1000 / Oligomeric state: octamer / Number unique components: 1 |
|---|
-Supramolecule #1: TFG
| Supramolecule | Name: TFG / type: organelle_or_cellular_component / ID: 1 / Number of copies: 8 / Oligomeric state: octamer / Recombinant expression: Yes / Database: NCBI |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: human / Location in cell: cytoplasm Homo sapiens (human) / synonym: human / Location in cell: cytoplasm |
-Experimental details
-Structure determination
| Method | negative staining, cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.6 / Details: 20 mM HEPES, 100 mM NaCl |
|---|---|
| Staining | Type: NEGATIVE / Details: Stained with 2% uranyl formate |
| Grid | Details: carbon grids |
| Vitrification | Cryogen name: NITROGEN / Instrument: OTHER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Date | Jun 6, 2012 |
| Image recording | Category: FILM / Film or detector model: GATAN ULTRASCAN 4000 (4k x 4k) / Digitization - Scanner: TEMSCAN / Number real images: 629 |
| Electron beam | Acceleration voltage: 120 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Nominal magnification: 37000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Tilt angle min: -55 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| CTF correction | Details: whole image |
|---|---|
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 30.0 Å / Resolution method: OTHER / Software - Name: MRC, EMAN / Number images used: 18000 |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)