+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-5983 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Structure of 2G12 (Fab)2 in Complex with Soluble and Fully Glycosylated HIV-1 Env by Negative-Stain Single Particle Electron Microscopy | |||||||||
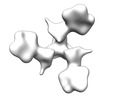
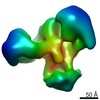
 マップデータ マップデータ | Reconstruction of HIV-1 Env SOSIP BG505.664 bound to soluble, two-domain CD4 (domains 1 and 2, sCD4) and 2G12 Fabs | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | HIV-1 / 2G12 / monoclonal antibodies / Envelope / CD4-bound Env | |||||||||
| 生物種 |   Human immunodeficiency virus 1 (ヒト免疫不全ウイルス) / Human immunodeficiency virus 1 (ヒト免疫不全ウイルス) /  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | 単粒子再構成法 / ネガティブ染色法 / 解像度: 26.0 Å | |||||||||
 データ登録者 データ登録者 | Murin CD / Julien JP / Sok D / Stanfield R / Khayat R / Cupo A / Moore JP / Burton DR / Wilson IA / Ward AB | |||||||||
 引用 引用 |  ジャーナル: J Virol / 年: 2014 ジャーナル: J Virol / 年: 2014タイトル: Structure of 2G12 Fab2 in complex with soluble and fully glycosylated HIV-1 Env by negative-stain single-particle electron microscopy. 著者: Charles D Murin / Jean-Philippe Julien / Devin Sok / Robyn L Stanfield / Reza Khayat / Albert Cupo / John P Moore / Dennis R Burton / Ian A Wilson / Andrew B Ward /  要旨: The neutralizing anti-HIV-1 antibody 2G12 is of particular interest due to the sterilizing protection it provides from viral challenge in animal models. 2G12 is a unique, domain-exchanged antibody ...The neutralizing anti-HIV-1 antibody 2G12 is of particular interest due to the sterilizing protection it provides from viral challenge in animal models. 2G12 is a unique, domain-exchanged antibody that binds exclusively to conserved N-linked glycans that form the high-mannose patch on the gp120 outer domain centered on a glycan at position N332. Several glycans in and around the 2G12 epitope have been shown to interact with other potent, broadly neutralizing antibodies; therefore, this region constitutes a supersite of vulnerability on gp120. While crystal structures of 2G12 and 2G12 bound to high-mannose glycans have been solved, no structural information that describes the interaction of 2G12 with gp120 or the Env trimer is available. Here, we present a negative-stain single-particle electron microscopy reconstruction of 2G12 Fab2 in complex with a soluble, trimeric Env at ∼17-Å resolution that reveals the antibody's interaction with its native and fully glycosylated epitope. We also mapped relevant glycans in this epitope by fitting high-resolution crystal structures and by performing neutralization assays of glycan knockouts. In addition, a reconstruction at ∼26 Å of the ternary complex formed by 2G12 Fab2, soluble CD4, and Env indicates that 2G12 may block membrane fusion by induced steric hindrance upon primary receptor binding, thereby abrogating Env's interaction with coreceptor(s). These structures provide a basis for understanding 2G12 binding and neutralization, and our low-resolution model and glycan assignments provide a basis for higher-resolution studies to determine the molecular nature of the 2G12 epitope. IMPORTANCE: HIV-1 is a human virus that results in the deaths of millions of people around the world each year. While there are several effective therapeutics available to prolong life, a vaccine is ...IMPORTANCE: HIV-1 is a human virus that results in the deaths of millions of people around the world each year. While there are several effective therapeutics available to prolong life, a vaccine is the best long-term solution for curbing this global epidemic. Here, we present structural data that reveal the viral binding site of one of the first HIV-1-neutralizing antibodies isolated, 2G12, and provide a rationale for its effectiveness. These structures provide a basis for higher-resolution studies to determine the molecular nature of the 2G12 epitope, which will aid in vaccine design and antibody-based therapies. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_5983.map.gz emd_5983.map.gz | 2.9 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-5983-v30.xml emd-5983-v30.xml emd-5983.xml emd-5983.xml | 14 KB 14 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_5983.tif emd_5983.tif | 2.2 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-5983 http://ftp.pdbj.org/pub/emdb/structures/EMD-5983 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5983 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5983 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_5983_validation.pdf.gz emd_5983_validation.pdf.gz | 78.4 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_5983_full_validation.pdf.gz emd_5983_full_validation.pdf.gz | 77.5 KB | 表示 | |
| XML形式データ |  emd_5983_validation.xml.gz emd_5983_validation.xml.gz | 494 B | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-5983 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-5983 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-5983 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-5983 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_5983.map.gz / 形式: CCP4 / 大きさ: 3.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_5983.map.gz / 形式: CCP4 / 大きさ: 3.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Reconstruction of HIV-1 Env SOSIP BG505.664 bound to soluble, two-domain CD4 (domains 1 and 2, sCD4) and 2G12 Fabs | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 4.35 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
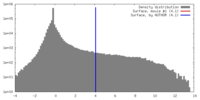
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : Fab fragment of 2G12 monoclonal antibody and soluble, two-domain ...
| 全体 | 名称: Fab fragment of 2G12 monoclonal antibody and soluble, two-domain CD4 (domains 1 and 2, sCD4) bound to HIV-1 Env BG505.664 |
|---|---|
| 要素 |
|
-超分子 #1000: Fab fragment of 2G12 monoclonal antibody and soluble, two-domain ...
| 超分子 | 名称: Fab fragment of 2G12 monoclonal antibody and soluble, two-domain CD4 (domains 1 and 2, sCD4) bound to HIV-1 Env BG505.664 タイプ: sample / ID: 1000 集合状態: Env trimer bound to three 2G12 domain-swapped Fabs and three sCD4s Number unique components: 3 |
|---|---|
| 分子量 | 実験値: 730 KDa / 理論値: 729 KDa / 手法: SEC-MALS |
-分子 #1: HIV-1 Env
| 分子 | 名称: HIV-1 Env / タイプ: protein_or_peptide / ID: 1 / Name.synonym: SOSIP BG505.664 / コピー数: 1 / 集合状態: Trimer / 組換発現: Yes |
|---|---|
| 由来(天然) | 生物種:   Human immunodeficiency virus 1 (ヒト免疫不全ウイルス) Human immunodeficiency virus 1 (ヒト免疫不全ウイルス)別称: HIV-1 |
| 分子量 | 実験値: 360 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) / 組換細胞: HEK 293F Homo sapiens (ヒト) / 組換細胞: HEK 293F |
-分子 #2: soluble CD4
| 分子 | 名称: soluble CD4 / タイプ: protein_or_peptide / ID: 2 / Name.synonym: sCD4 / コピー数: 1 / 集合状態: monomer / 組換発現: Yes |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) / 別称: Human Homo sapiens (ヒト) / 別称: Human |
| 分子量 | 理論値: 23 KDa |
| 組換発現 | 生物種:  |
-分子 #3: Human Monoclonal Antibody 2G12 IgG1 Fab Fragment
| 分子 | 名称: Human Monoclonal Antibody 2G12 IgG1 Fab Fragment / タイプ: protein_or_peptide / ID: 3 / Name.synonym: 2G12 Fab / 集合状態: Dimer of Fabs / 組換発現: Yes |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) / 別称: Human Homo sapiens (ヒト) / 別称: Human |
| 分子量 | 実験値: 100 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) / 組換細胞: HEK 293F / 組換プラスミド: phCMV3 Homo sapiens (ヒト) / 組換細胞: HEK 293F / 組換プラスミド: phCMV3 |
-実験情報
-構造解析
| 手法 | ネガティブ染色法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 0.03 mg/mL |
|---|---|
| 緩衝液 | pH: 7.4 / 詳細: 150 mM NaCl, 10 mM Tris |
| 染色 | タイプ: NEGATIVE 詳細: Grids with adsorbed protein were floated on 2% w/v uranyl formate for 30 seconds. |
| グリッド | 詳細: 400 mesh copper grid with thin carbon support, Gatan plasma cleaned |
| 凍結 | 凍結剤: NONE / 装置: OTHER |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TECNAI SPIRIT |
|---|---|
| アライメント法 | Legacy - 非点収差: Objective lens astigmatism was corrected at 100,000 times magnification. |
| 特殊光学系 | エネルギーフィルター - 名称: FEI |
| 日付 | 2012年7月12日 |
| 撮影 | カテゴリ: CCD / フィルム・検出器のモデル: OTHER / 実像数: 149 / 平均電子線量: 30 e/Å2 |
| Tilt angle min | 0 |
| 電子線 | 加速電圧: 120 kV / 電子線源: TUNGSTEN HAIRPIN |
| 電子光学系 | 倍率(補正後): 52000 / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 0.8 µm / 最小 デフォーカス(公称値): 0.5 µm / 倍率(公称値): 52000 |
| 試料ステージ | 試料ホルダーモデル: HOME BUILD |
| 実験機器 |  モデル: Tecnai Spirit / 画像提供: FEI Company |
- 画像解析
画像解析
| 詳細 | The particles were selected using automatic selection program Leginon and processed using the Appion system. |
|---|---|
| 最終 再構成 | アルゴリズム: OTHER / 解像度のタイプ: BY AUTHOR / 解像度: 26.0 Å / 解像度の算出法: OTHER / ソフトウェア - 名称: EMAN2 / 詳細: Final map was low pass filtered to 20 Angstrom. / 使用した粒子像数: 5372 |
| 最終 2次元分類 | クラス数: 100 |
-原子モデル構築 1
| 初期モデル | PDB ID: |
|---|---|
| ソフトウェア | 名称:  Chimera Chimera |
| 詳細 | Three structures of HIV-1 gp120 bound to sCD4 were fit into the HIV-1 Env trimer portion of the map. |
| 精密化 | 空間: REAL / プロトコル: RIGID BODY FIT |
-原子モデル構築 2
| 初期モデル | PDB ID:  1op5 |
|---|---|
| ソフトウェア | 名称:  Chimera Chimera |
| 詳細 | 2G12 Fab structures were fit by rigid body fitting using the UCSF Chimera volume fit option, simulating a map at an estimated resolution of 20 Angstrom. |
| 精密化 | 空間: REAL / プロトコル: RIGID BODY FIT |
 ムービー
ムービー コントローラー
コントローラー







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)