[English] 日本語
 Yorodumi
Yorodumi- EMDB-5834: A Unique Human Mycoplasma Protein that Generically Blocks Antigen... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-5834 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | A Unique Human Mycoplasma Protein that Generically Blocks Antigen-Antibody Union | |||||||||
 Map data Map data | Protein M and Fab b12 | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  Mycoplasma genitalium (bacteria) / Mycoplasma genitalium (bacteria) /  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 21.3 Å | |||||||||
 Authors Authors | Grover RK / Zhu X / Nieusma T / Jones T / Boreo I / MacLeod AS / Mark A / Niessen S / Kim HJ / Kong L ...Grover RK / Zhu X / Nieusma T / Jones T / Boreo I / MacLeod AS / Mark A / Niessen S / Kim HJ / Kong L / Assad-Garcia N / Kwon K / Chesi M / Salomon DR / Jelinek DF / Kyle RA / Pyles RB / Glass JI / Ward AB / Wilson IA / Lerner RA | |||||||||
 Citation Citation |  Journal: Science / Year: 2014 Journal: Science / Year: 2014Title: A structurally distinct human mycoplasma protein that generically blocks antigen-antibody union. Authors: Rajesh K Grover / Xueyong Zhu / Travis Nieusma / Teresa Jones / Isabel Boreo / Amanda S MacLeod / Adam Mark / Sherry Niessen / Helen J Kim / Leopold Kong / Nacyra Assad-Garcia / Keehwan Kwon ...Authors: Rajesh K Grover / Xueyong Zhu / Travis Nieusma / Teresa Jones / Isabel Boreo / Amanda S MacLeod / Adam Mark / Sherry Niessen / Helen J Kim / Leopold Kong / Nacyra Assad-Garcia / Keehwan Kwon / Marta Chesi / Vaughn V Smider / Daniel R Salomon / Diane F Jelinek / Robert A Kyle / Richard B Pyles / John I Glass / Andrew B Ward / Ian A Wilson / Richard A Lerner /  Abstract: We report the discovery of a broadly reactive antibody-binding protein (Protein M) from human mycoplasma. The crystal structure of the ectodomain of transmembrane Protein M differs from other known ...We report the discovery of a broadly reactive antibody-binding protein (Protein M) from human mycoplasma. The crystal structure of the ectodomain of transmembrane Protein M differs from other known protein structures, as does its mechanism of antibody binding. Protein M binds with high affinity to all types of human and nonhuman immunoglobulin G, predominantly through attachment to the conserved portions of the variable region of the κ and λ light chains. Protein M blocks antibody-antigen union, likely because of its large C-terminal domain extending over the antibody-combining site, blocking entry to large antigens. Similar to the other immunoglobulin-binding proteins such as Protein A, Protein M as well as its orthologs in other Mycoplasma species could become invaluable reagents in the antibody field. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_5834.map.gz emd_5834.map.gz | 626 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-5834-v30.xml emd-5834-v30.xml emd-5834.xml emd-5834.xml | 12.6 KB 12.6 KB | Display Display |  EMDB header EMDB header |
| Images |  400_5834.gif 400_5834.gif 80_5834.gif 80_5834.gif | 21.9 KB 2.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-5834 http://ftp.pdbj.org/pub/emdb/structures/EMD-5834 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5834 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5834 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_5834.map.gz / Format: CCP4 / Size: 670.9 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_5834.map.gz / Format: CCP4 / Size: 670.9 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Protein M and Fab b12 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.05 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
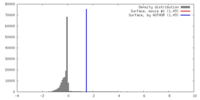
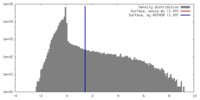
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Fab of human mAb b12 in complex with Full length MG281
| Entire | Name: Fab of human mAb b12 in complex with Full length MG281 |
|---|---|
| Components |
|
-Supramolecule #1000: Fab of human mAb b12 in complex with Full length MG281
| Supramolecule | Name: Fab of human mAb b12 in complex with Full length MG281 type: sample / ID: 1000 Oligomeric state: one monomer of MG281 binds to one Fab monomer Number unique components: 2 |
|---|---|
| Molecular weight | Theoretical: 100 KDa / Method: Western blot |
-Macromolecule #1: MG281
| Macromolecule | Name: MG281 / type: protein_or_peptide / ID: 1 / Name.synonym: protein M / Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Mycoplasma genitalium (bacteria) / synonym: human mycoplasma Mycoplasma genitalium (bacteria) / synonym: human mycoplasma |
| Molecular weight | Experimental: 50 KDa / Theoretical: 50 KDa |
| Recombinant expression | Organism:  |
| Sequence | UniProtKB: Uncharacterized protein MG281 |
-Macromolecule #2: mAb b12 Fab
| Macromolecule | Name: mAb b12 Fab / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Recombinant expression: No / Database: NCBI |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: human Homo sapiens (human) / synonym: human |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Staining | Type: NEGATIVE Details: 3 uL 2% uranyl formate was added to grids adsorbed with 3uL protein sample, then blotted and air-dried. |
|---|---|
| Grid | Details: 400-Cu copper mesh with thin nitrocellulose support and thin carbon, glow discharged |
| Vitrification | Cryogen name: NONE / Instrument: OTHER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI SPIRIT |
|---|---|
| Temperature | Average: 298 K |
| Alignment procedure | Legacy - Astigmatism: Objective astigmatism was corrected at 100,000 time magnification using a live feed of the power spectrum. |
| Date | Jan 15, 2013 |
| Image recording | Category: CCD / Film or detector model: TVIPS TEMCAM-F416 (4k x 4k) / Number real images: 491 Details: 36 images were collected at each angle in 5 degree intervals from 0 degrees to -55 degrees. |
| Tilt angle max | 0 |
| Electron beam | Acceleration voltage: 120 kV / Electron source: LAB6 |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: -1.0 µm / Nominal defocus min: -1.0 µm / Nominal magnification: 52000 |
| Sample stage | Specimen holder model: SIDE ENTRY, EUCENTRIC / Tilt angle min: -55 |
| Experimental equipment |  Model: Tecnai Spirit / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | Particles were selected using automatic (difference-of-Gaussians) picking followed by reference-free classification to eliminate noisy picks or non-target aggregation states. |
|---|---|
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 21.3 Å / Resolution method: OTHER / Software - Name: Appion, spider, EMAN1, Xmipp / Number images used: 20917 |
| Final two d classification | Number classes: 13 |
-Atomic model buiding 1
| Initial model | PDB ID: Chain - #0 - Chain ID: H / Chain - #1 - Chain ID: L / Chain - #2 - Chain ID: M |
|---|---|
| Software | Name:  Chimera Chimera |
| Details | The crystal structure was docked to the EM volume using the fitting function in UCSF Chimera. |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)