[English] 日本語
 Yorodumi
Yorodumi- EMDB-53345: Cryo-EM structure of aquaporin 3 at pH 8.0 with hydrogen peroxide -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of aquaporin 3 at pH 8.0 with hydrogen peroxide | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | membrane protein / glycerol channel / hydrogen peroxide transport / tetramer | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of immune system process / glycerol channel activity / Passive transport by Aquaporins / regulation of keratinocyte differentiation / glycerol transmembrane transport / urea transmembrane transporter activity / renal water absorption / water channel activity / water transport / Differentiation of Keratinocytes in Interfollicular Epidermis in Mammalian Skin ...positive regulation of immune system process / glycerol channel activity / Passive transport by Aquaporins / regulation of keratinocyte differentiation / glycerol transmembrane transport / urea transmembrane transporter activity / renal water absorption / water channel activity / water transport / Differentiation of Keratinocytes in Interfollicular Epidermis in Mammalian Skin / response to vitamin D / odontogenesis / response to retinoic acid / renal water homeostasis / establishment of localization in cell / response to ischemia / response to calcium ion / cell-cell junction / Vasopressin regulates renal water homeostasis via Aquaporins / cellular response to hypoxia / basolateral plasma membrane / nucleoplasm / membrane / identical protein binding / plasma membrane / cytoplasm Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.0 Å | ||||||||||||
 Authors Authors | Huang P / Venskutonyte R / Lindkvist-Petersson K | ||||||||||||
| Funding support |  Sweden, 3 items Sweden, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2025 Journal: Nat Commun / Year: 2025Title: Structural insights into AQP3 channel closure upon pH and redox changes reveal an autoregulatory molecular mechanism. Authors: Peng Huang / Raminta Venskutonytė / Carter J Wilson / Sara Bsharat / Rashmi B Prasad / Pontus Gourdon / Isabella Artner / Bert L de Groot / Karin Lindkvist-Petersson /    Abstract: Regulation of intracellular levels of reactive oxygen species (ROS) remains poorly understood. Aquaporin 3 (AQP3) facilitates the membrane transport of hydrogen peroxide (HO), a key ROS signaling ...Regulation of intracellular levels of reactive oxygen species (ROS) remains poorly understood. Aquaporin 3 (AQP3) facilitates the membrane transport of hydrogen peroxide (HO), a key ROS signaling molecule. Here we elucidate the molecular mechanism of AQP3 and show that its regulatory properties are both pH dependent and autoregulated by HO. Using single particle cryo-electron microscopy, we present open and closed conformations of human AQP3. At pH 8.0, the channel adopts an open state, while acidic pH or exposure to HO promotes closure via a large conformational rearrangement of extracellular loop E. These findings reveal a mechanism for autoregulation of HO transport and establish AQP3 as a key modulator of redox homeostasis in human pancreatic β-cells. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_53345.map.gz emd_53345.map.gz | 59.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-53345-v30.xml emd-53345-v30.xml emd-53345.xml emd-53345.xml | 17 KB 17 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_53345_fsc.xml emd_53345_fsc.xml | 8.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_53345.png emd_53345.png | 81.3 KB | ||
| Filedesc metadata |  emd-53345.cif.gz emd-53345.cif.gz | 5.8 KB | ||
| Others |  emd_53345_half_map_1.map.gz emd_53345_half_map_1.map.gz emd_53345_half_map_2.map.gz emd_53345_half_map_2.map.gz | 59.1 MB 59.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-53345 http://ftp.pdbj.org/pub/emdb/structures/EMD-53345 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-53345 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-53345 | HTTPS FTP |
-Related structure data
| Related structure data |  9qszMC  9qsxC  9qsyC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_53345.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_53345.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.83 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_53345_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_53345_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Aquaporin 3
| Entire | Name: Aquaporin 3 |
|---|---|
| Components |
|
-Supramolecule #1: Aquaporin 3
| Supramolecule | Name: Aquaporin 3 / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Aquaporin-3
| Macromolecule | Name: Aquaporin-3 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 32.396549 KDa |
| Recombinant expression | Organism:  Komagataella pastoris (fungus) Komagataella pastoris (fungus) |
| Sequence | String: MGRQKELVSR CGEMLHIRYR LLRQALAECL GTLILVMFGC GSVAQVVLSR GTHGGFLTIN LAFGFAVTLG ILIAGQVSGA HLNPAVTFA MCFLAREPWI KLPIYTLAQT LGAFLGAGIV FGLYYDAIWH FADNQLFVSG PNGTAGIFAT YPSGHLDMIN G FFDQFIGT ...String: MGRQKELVSR CGEMLHIRYR LLRQALAECL GTLILVMFGC GSVAQVVLSR GTHGGFLTIN LAFGFAVTLG ILIAGQVSGA HLNPAVTFA MCFLAREPWI KLPIYTLAQT LGAFLGAGIV FGLYYDAIWH FADNQLFVSG PNGTAGIFAT YPSGHLDMIN G FFDQFIGT ASLIVCVLAI VDPYNNPVPR GLEAFTVGLV VLVIGTSMGF NSGYAVNPAR DFGPRLFTAL AGWGSAVFTT GQ HWWWVPI VSPLLGSIAG VFVYQLMIGC HLEQPPPSNE EENVKLAHVK HKEQIHHHHH H UniProtKB: Aquaporin-3 |
-Macromolecule #2: HYDROGEN PEROXIDE
| Macromolecule | Name: HYDROGEN PEROXIDE / type: ligand / ID: 2 / Number of copies: 16 / Formula: PEO |
|---|---|
| Molecular weight | Theoretical: 34.015 Da |
| Chemical component information | 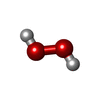 ChemComp-PEO: |
-Macromolecule #3: water
| Macromolecule | Name: water / type: ligand / ID: 3 / Number of copies: 12 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 Details: 20mM Tris-HCL (pH 8), 100mM NaCl with supplemented H2O2 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number real images: 15001 / Average exposure time: 1.8 sec. / Average electron dose: 52.825 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 105000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





































