[English] 日本語
 Yorodumi
Yorodumi- EMDB-43088: Cryogenic electron microscopy structure of human serum albumin in... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryogenic electron microscopy structure of human serum albumin in complex with teniposide | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Human Serum Albumin / teniposide / TRANSPORT PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationbilirubin transport / Ciprofloxacin ADME / exogenous protein binding / cellular response to calcium ion starvation / enterobactin binding / Heme biosynthesis / HDL remodeling / molecular carrier activity / negative regulation of mitochondrial depolarization / Prednisone ADME ...bilirubin transport / Ciprofloxacin ADME / exogenous protein binding / cellular response to calcium ion starvation / enterobactin binding / Heme biosynthesis / HDL remodeling / molecular carrier activity / negative regulation of mitochondrial depolarization / Prednisone ADME / Heme degradation / Aspirin ADME / antioxidant activity / Scavenging of heme from plasma / toxic substance binding / Recycling of bile acids and salts / platelet alpha granule lumen / fatty acid binding / cellular response to starvation / Post-translational protein phosphorylation / Cytoprotection by HMOX1 / response to nutrient levels / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / Platelet degranulation / pyridoxal phosphate binding / protein-folding chaperone binding / blood microparticle / endoplasmic reticulum lumen / copper ion binding / endoplasmic reticulum / Golgi apparatus / protein-containing complex / : / DNA binding / extracellular exosome / extracellular region / identical protein binding / nucleus Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Catalano C / Lucier KW / To D / Senko S / Tran NL / Farwell AC / Silva SM / Dip PV / Poweleit N / Scapin G | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: J Struct Biol / Year: 2024 Journal: J Struct Biol / Year: 2024Title: The CryoEM structure of human serum albumin in complex with ligands. Authors: Claudio Catalano / Kyle W Lucier / Dennis To / Skerdi Senko / Nhi L Tran / Ashlyn C Farwell / Sabrina M Silva / Phat V Dip / Nicole Poweleit / Giovanna Scapin /  Abstract: Human serum albumin (HSA) is the most prevalent plasma protein in the human body, accounting for 60 % of the total plasma protein. HSA plays a major pharmacokinetic function, serving as a ...Human serum albumin (HSA) is the most prevalent plasma protein in the human body, accounting for 60 % of the total plasma protein. HSA plays a major pharmacokinetic function, serving as a facilitator in the distribution of endobiotics and xenobiotics within the organism. In this paper we report the cryoEM structures of HSA in the apo form and in complex with two ligands (salicylic acid and teniposide) at a resolution of 3.5, 3.7 and 3.4 Å, respectively. We expand upon previously published work and further demonstrate that sub-4 Å maps of ∼60 kDa proteins can be routinely obtained using a 200 kV microscope, employing standard workflows. Most importantly, these maps allowed for the identification of small molecule ligands, emphasizing the practical applicability of this methodology and providing a starting point for subsequent computational modeling and in silico optimization. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_43088.map.gz emd_43088.map.gz | 306.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-43088-v30.xml emd-43088-v30.xml emd-43088.xml emd-43088.xml | 17 KB 17 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_43088_fsc.xml emd_43088_fsc.xml | 14.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_43088.png emd_43088.png | 49.6 KB | ||
| Masks |  emd_43088_msk_1.map emd_43088_msk_1.map | 325 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-43088.cif.gz emd-43088.cif.gz | 6.2 KB | ||
| Others |  emd_43088_half_map_1.map.gz emd_43088_half_map_1.map.gz emd_43088_half_map_2.map.gz emd_43088_half_map_2.map.gz | 300.4 MB 300.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-43088 http://ftp.pdbj.org/pub/emdb/structures/EMD-43088 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43088 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43088 | HTTPS FTP |
-Related structure data
| Related structure data |  8vacMC  8vaeC  8vafC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_43088.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_43088.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.566 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_43088_msk_1.map emd_43088_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
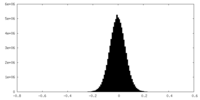
| Density Histograms |
-Half map: #2
| File | emd_43088_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
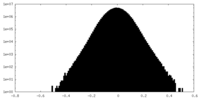
| Density Histograms |
-Half map: #1
| File | emd_43088_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : human serum albumin with teniposide
| Entire | Name: human serum albumin with teniposide |
|---|---|
| Components |
|
-Supramolecule #1: human serum albumin with teniposide
| Supramolecule | Name: human serum albumin with teniposide / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Serum albumin
| Macromolecule | Name: Serum albumin / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 66.571219 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: DAHKSEVAHR FKDLGEENFK ALVLIAFAQY LQQCPFEDHV KLVNEVTEFA KTCVADESAE NCDKSLHTLF GDKLCTVATL RETYGEMAD CCAKQEPERN ECFLQHKDDN PNLPRLVRPE VDVMCTAFHD NEETFLKKYL YEIARRHPYF YAPELLFFAK R YKAAFTEC ...String: DAHKSEVAHR FKDLGEENFK ALVLIAFAQY LQQCPFEDHV KLVNEVTEFA KTCVADESAE NCDKSLHTLF GDKLCTVATL RETYGEMAD CCAKQEPERN ECFLQHKDDN PNLPRLVRPE VDVMCTAFHD NEETFLKKYL YEIARRHPYF YAPELLFFAK R YKAAFTEC CQAADKAACL LPKLDELRDE GKASSAKQRL KCASLQKFGE RAFKAWAVAR LSQRFPKAEF AEVSKLVTDL TK VHTECCH GDLLECADDR ADLAKYICEN QDSISSKLKE CCEKPLLEKS HCIAEVENDE MPADLPSLAA DFVESKDVCK NYA EAKDVF LGMFLYEYAR RHPDYSVVLL LRLAKTYETT LEKCCAAADP HECYAKVFDE FKPLVEEPQN LIKQNCELFE QLGE YKFQN ALLVRYTKKV PQVSTPTLVE VSRNLGKVGS KCCKHPEAKR MPCAEDYLSV VLNQLCVLHE KTPVSDRVTK CCTES LVNR RPCFSALEVD ETYVPKEFNA ETFTFHADIC TLSEKERQIK KQTALVELVK HKPKATKEQL KAVMDDFAAF VEKCCK ADD KETCFAEEGK KLVAASQAAL GL UniProtKB: Albumin |
-Macromolecule #2: (5S,5aR,8aR,9R)-9-(4-hydroxy-3,5-dimethoxyphenyl)-8-oxo-5,5a,6,8,...
| Macromolecule | Name: (5S,5aR,8aR,9R)-9-(4-hydroxy-3,5-dimethoxyphenyl)-8-oxo-5,5a,6,8,8a,9-hexahydrofuro[3',4':6,7]naphtho[2,3-d][1,3]dioxol -5-yl 4,6-O-(thiophen-2-ylmethylidene)-beta-D-glucopyranoside type: ligand / ID: 2 / Number of copies: 1 / Formula: 9TP |
|---|---|
| Molecular weight | Theoretical: 656.654 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 / Component - Name: PBS |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS GLACIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Number grids imaged: 1 / Number real images: 12075 / Average exposure time: 3.7 sec. / Average electron dose: 36.43 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 240000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)