+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | AgamOR28 structure bound to 2,4,5-trimethylthiazole | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Mosquito olfactory receptor / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationolfactory receptor activity / detection of chemical stimulus involved in sensory perception of smell / odorant binding / signal transduction / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |   Apocrypta bakeri (insect) Apocrypta bakeri (insect) | |||||||||
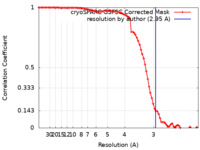
| Method | single particle reconstruction / cryo EM / Resolution: 2.95 Å | |||||||||
 Authors Authors | Zhao J / del Marmol J | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2024 Journal: Science / Year: 2024Title: Structural basis of odor sensing by insect heteromeric odorant receptors. Authors: Jiawei Zhao / Andy Q Chen / Jaewook Ryu / Josefina Del Mármol /  Abstract: Most insects, including human-targeting mosquitoes, detect odors through odorant-activated ion channel complexes consisting of a divergent odorant-binding subunit (OR) and a conserved co-receptor ...Most insects, including human-targeting mosquitoes, detect odors through odorant-activated ion channel complexes consisting of a divergent odorant-binding subunit (OR) and a conserved co-receptor subunit (Orco). As a basis for understanding how odorants activate these heteromeric receptors, we report here cryo-electron microscopy structures of two different heteromeric odorant receptor complexes containing ORs from disease-vector mosquitos or . These structures reveal an unexpected stoichiometry of one OR to three Orco subunits. Comparison of structures in odorant-bound and unbound states indicates that odorant binding to the sole OR subunit is sufficient to open the channel pore, suggesting a mechanism of OR activation and a conceptual framework for understanding evolution of insect odorant receptor sensitivity. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42946.map.gz emd_42946.map.gz | 49.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42946-v30.xml emd-42946-v30.xml emd-42946.xml emd-42946.xml | 16.1 KB 16.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_42946_fsc.xml emd_42946_fsc.xml | 7.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_42946.png emd_42946.png | 138.5 KB | ||
| Filedesc metadata |  emd-42946.cif.gz emd-42946.cif.gz | 6 KB | ||
| Others |  emd_42946_half_map_1.map.gz emd_42946_half_map_1.map.gz emd_42946_half_map_2.map.gz emd_42946_half_map_2.map.gz | 49 MB 49 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42946 http://ftp.pdbj.org/pub/emdb/structures/EMD-42946 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42946 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42946 | HTTPS FTP |
-Related structure data
| Related structure data |  8v3dMC  8v00C  8v02C  8v3cC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_42946.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42946.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.061 Å | ||||||||||||||||||||||||||||||||||||
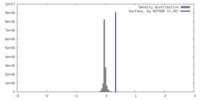
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_42946_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
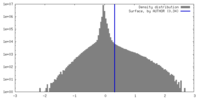
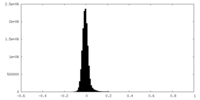
| Density Histograms |
-Half map: #1
| File | emd_42946_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
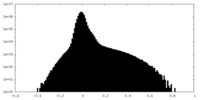
| Density Histograms |
- Sample components
Sample components
-Entire : AgamOR28 heterotetramer bound to 2,4,5-trimethylthiazole
| Entire | Name: AgamOR28 heterotetramer bound to 2,4,5-trimethylthiazole |
|---|---|
| Components |
|
-Supramolecule #1: AgamOR28 heterotetramer bound to 2,4,5-trimethylthiazole
| Supramolecule | Name: AgamOR28 heterotetramer bound to 2,4,5-trimethylthiazole type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|
-Supramolecule #2: Anopheles gambiae OR28
| Supramolecule | Name: Anopheles gambiae OR28 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #2 |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #3: Apocrypta bakeri Orco
| Supramolecule | Name: Apocrypta bakeri Orco / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Apocrypta bakeri (insect) Apocrypta bakeri (insect) |
-Macromolecule #1: Odorant receptor Orco
| Macromolecule | Name: Odorant receptor Orco / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Apocrypta bakeri (insect) Apocrypta bakeri (insect) |
| Molecular weight | Theoretical: 53.204953 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MKFKHQGLVA DLLPNIRVMQ GVGHFMFNYY SEGKKFPHRI YCIVTLLLLL LQYGMMAVNL MMESDDVDDL TANTITMLFF LHPIVKMIY FPVRSKIFYK TLAIWNNPNS HPLFAESNAR FHALAITKMR RLLFCVAGAT IFSVISWTGI TFIEDSVKRI T DPETNETT ...String: MKFKHQGLVA DLLPNIRVMQ GVGHFMFNYY SEGKKFPHRI YCIVTLLLLL LQYGMMAVNL MMESDDVDDL TANTITMLFF LHPIVKMIY FPVRSKIFYK TLAIWNNPNS HPLFAESNAR FHALAITKMR RLLFCVAGAT IFSVISWTGI TFIEDSVKRI T DPETNETT IIPIPRLMIR TFYPFNAMSG AGHVFALIYQ FYYLVISMAV SNSLDVLFCS WLLFACEQLQ HLKAIMKPLM EL SATLDTV VPNSGELFKA GSADHLRESQ GVQPSGNGDN VLDVDLRGIY SNRQDFTATF RPTAGTTFNG GVGPNGLTKK QEM LVRSAI KYWVERHKHV VRLVTAVGDA YGVALLLHML TTTITLTLLA YQATKVNGVN VYAATVIGYL LYTLGQVFLF CIFG NRLIE ESSSVMEAAY SCHWYDGSEE AKTFVQIVCQ QCQKAMSISG AKFFTVSLDL FASVLGAVVT YFMVLVQLK UniProtKB: Odorant receptor |
-Macromolecule #2: OR28
| Macromolecule | Name: OR28 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 45.826898 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MARLVLHEVR YVLMAMLYIS RGMAKQIQNS TIDLYVYWFL TFIPIASLCV PQFTYLVVDT KSLIDFISVL VPITEILLTN GKMIICNVK RGKIINLINQ VQVAWDECAK SEHLEIQTLI TATAKKTKIF VIIYTTSFLL ICVEYSSMPL FKLIYHSAVY G KQSNYTIA ...String: MARLVLHEVR YVLMAMLYIS RGMAKQIQNS TIDLYVYWFL TFIPIASLCV PQFTYLVVDT KSLIDFISVL VPITEILLTN GKMIICNVK RGKIINLINQ VQVAWDECAK SEHLEIQTLI TATAKKTKIF VIIYTTSFLL ICVEYSSMPL FKLIYHSAVY G KQSNYTIA LPYLSRFAYS TESTTSFAWT YFFILLGVYL LALTLSGFDS LFATLVMHVK MMFKVLKFEI EQLGLDLSAG KS HVELQAK LKQIILKHKT NLSLIEQLED GFSFFLMAQF LTSSILVCVV LYELTMVFGW NEDTFKTVTY LPGAILQLFL FCW YAQQIT EEARLVSDHI YNIPWYLADP KLQKDILTFM VKAQKPTGVT ASKFYMVTLQ TFQRISSTSY SYFTLLQTIN QQ UniProtKB: Uncharacterized protein |
-Macromolecule #3: 2,4,5-trimethyl-1,3-thiazole
| Macromolecule | Name: 2,4,5-trimethyl-1,3-thiazole / type: ligand / ID: 3 / Number of copies: 1 / Formula: A1AFC |
|---|---|
| Molecular weight | Theoretical: 127.207 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)