+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | MERS-CoV Nsp1 protein bound to the Human 40S Ribosomal subunit | |||||||||
 Map data Map data | Composite Map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | MERS-CoV Nsp1 / translation inhibition / 40S ribosome / betacoronaviruses / RIBOSOME / Ribosome-Viral Protein complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of endoplasmic reticulum unfolded protein response / oxidized pyrimidine DNA binding / response to TNF agonist / positive regulation of base-excision repair / positive regulation of ubiquitin-protein transferase activity / positive regulation of respiratory burst involved in inflammatory response / positive regulation of gastrulation / protein tyrosine kinase inhibitor activity / positive regulation of DNA-templated transcription initiation / positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage ...negative regulation of endoplasmic reticulum unfolded protein response / oxidized pyrimidine DNA binding / response to TNF agonist / positive regulation of base-excision repair / positive regulation of ubiquitin-protein transferase activity / positive regulation of respiratory burst involved in inflammatory response / positive regulation of gastrulation / protein tyrosine kinase inhibitor activity / positive regulation of DNA-templated transcription initiation / positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage / IRE1-RACK1-PP2A complex / positive regulation of Golgi to plasma membrane protein transport / nucleolus organization / TNFR1-mediated ceramide production / negative regulation of RNA splicing / neural crest cell differentiation / supercoiled DNA binding / cytoplasmic translational initiation / NF-kappaB complex / negative regulation of DNA repair / oxidized purine DNA binding / cysteine-type endopeptidase activator activity involved in apoptotic process / rRNA modification in the nucleus and cytosol / negative regulation of intrinsic apoptotic signaling pathway in response to hydrogen peroxide / negative regulation of bicellular tight junction assembly / ubiquitin-like protein conjugating enzyme binding / regulation of establishment of cell polarity / negative regulation of phagocytosis / erythrocyte homeostasis / cytoplasmic side of rough endoplasmic reticulum membrane / Formation of the ternary complex, and subsequently, the 43S complex / ion channel inhibitor activity / protein kinase A binding / laminin receptor activity / Ribosomal scanning and start codon recognition / pigmentation / positive regulation of mitochondrial depolarization / Translation initiation complex formation / negative regulation of Wnt signaling pathway / fibroblast growth factor binding / Protein hydroxylation / monocyte chemotaxis / BH3 domain binding / negative regulation of translational frameshifting / regulation of adenylate cyclase-activating G protein-coupled receptor signaling pathway / SARS-CoV-1 modulates host translation machinery / positive regulation of GTPase activity / mTORC1-mediated signalling / TOR signaling / iron-sulfur cluster binding / Peptide chain elongation / regulation of cell division / host cell membrane / cellular response to ethanol / Selenocysteine synthesis / Formation of a pool of free 40S subunits / positive regulation of intrinsic apoptotic signaling pathway by p53 class mediator / negative regulation of protein binding / Eukaryotic Translation Termination / protein serine/threonine kinase inhibitor activity / SRP-dependent cotranslational protein targeting to membrane / Response of EIF2AK4 (GCN2) to amino acid deficiency / ubiquitin ligase inhibitor activity / negative regulation of respiratory burst involved in inflammatory response / Viral mRNA Translation / endonucleolytic cleavage to generate mature 3'-end of SSU-rRNA from (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / positive regulation of signal transduction by p53 class mediator / negative regulation of ubiquitin-dependent protein catabolic process / GTP hydrolysis and joining of the 60S ribosomal subunit / L13a-mediated translational silencing of Ceruloplasmin expression / Major pathway of rRNA processing in the nucleolus and cytosol / regulation of translational fidelity / positive regulation of microtubule polymerization / phagocytic cup / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / positive regulation of intrinsic apoptotic signaling pathway / spindle assembly / Protein methylation / Nuclear events stimulated by ALK signaling in cancer / endonucleolytic cleavage in ITS1 to separate SSU-rRNA from 5.8S rRNA and LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / translation regulator activity / rough endoplasmic reticulum / positive regulation of cell cycle / ribosomal small subunit export from nucleus / laminin binding / translation initiation factor binding / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / DNA-(apurinic or apyrimidinic site) endonuclease activity / gastrulation / Maturation of protein E / Maturation of protein E / ER Quality Control Compartment (ERQC) / Myoclonic epilepsy of Lafora / FLT3 signaling by CBL mutants / signaling adaptor activity / MDM2/MDM4 family protein binding / negative regulation of protein ubiquitination / IRAK2 mediated activation of TAK1 complex / Prevention of phagosomal-lysosomal fusion Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.6 Å | |||||||||
 Authors Authors | Devarkar SC / Xiong Y | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2023 Journal: Cell Rep / Year: 2023Title: Structural basis for translation inhibition by MERS-CoV Nsp1 reveals a conserved mechanism for betacoronaviruses. Authors: Swapnil C Devarkar / Michael Vetick / Shravani Balaji / Ivan B Lomakin / Luojia Yang / Danni Jin / Wendy V Gilbert / Sidi Chen / Yong Xiong /  Abstract: All betacoronaviruses (β-CoVs) encode non-structural protein 1 (Nsp1), an essential pathogenicity factor that potently restricts host gene expression. Among the β-CoV family, MERS-CoV is the most ...All betacoronaviruses (β-CoVs) encode non-structural protein 1 (Nsp1), an essential pathogenicity factor that potently restricts host gene expression. Among the β-CoV family, MERS-CoV is the most distantly related member to SARS-CoV-2, and the mechanism for host translation inhibition by MERS-CoV Nsp1 remains controversial. Herein, we show that MERS-CoV Nsp1 directly interacts with the 40S ribosomal subunit. Using cryogenic electron microscopy (cryo-EM), we report a 2.6-Å structure of the MERS-CoV Nsp1 bound to the human 40S ribosomal subunit. The extensive interactions between C-terminal domain of MERS-CoV Nsp1 and the mRNA entry channel of the 40S ribosomal subunit are critical for its translation inhibition function. This mechanism of MERS-CoV Nsp1 is strikingly similar to SARS-CoV and SARS-CoV-2 Nsp1, despite modest sequence conservation. Our results reveal that the mechanism of host translation inhibition is conserved across β-CoVs and highlight a potential therapeutic target for the development of antivirals that broadly restrict β-CoVs. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41039.map.gz emd_41039.map.gz | 100 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41039-v30.xml emd-41039-v30.xml emd-41039.xml emd-41039.xml | 54.2 KB 54.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_41039_fsc.xml emd_41039_fsc.xml emd_41039_fsc_2.xml emd_41039_fsc_2.xml emd_41039_fsc_3.xml emd_41039_fsc_3.xml | 12.4 KB 12.4 KB 12.4 KB | Display Display Display |  FSC data file FSC data file |
| Images |  emd_41039.png emd_41039.png | 67.1 KB | ||
| Filedesc metadata |  emd-41039.cif.gz emd-41039.cif.gz | 11.9 KB | ||
| Others |  emd_41039_additional_1.map.gz emd_41039_additional_1.map.gz emd_41039_additional_2.map.gz emd_41039_additional_2.map.gz emd_41039_additional_3.map.gz emd_41039_additional_3.map.gz | 106.1 MB 105.9 MB 106 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41039 http://ftp.pdbj.org/pub/emdb/structures/EMD-41039 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41039 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41039 | HTTPS FTP |
-Related structure data
| Related structure data |  8t4sMC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_41039.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41039.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composite Map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
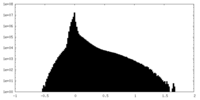
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: 40S Head subvolume
| File | emd_41039_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 40S Head subvolume | ||||||||||||
| Projections & Slices |
| ||||||||||||
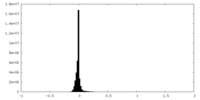
| Density Histograms |
-Additional map: Consensus Refinement Map
| File | emd_41039_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Consensus Refinement Map | ||||||||||||
| Projections & Slices |
| ||||||||||||
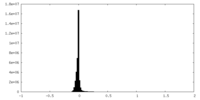
| Density Histograms |
-Additional map: 40S Body subvolume
| File | emd_41039_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 40S Body subvolume | ||||||||||||
| Projections & Slices |
| ||||||||||||
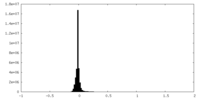
| Density Histograms |
- Sample components
Sample components
+Entire : MERS-CoV Nsp1 bound to human 40S ribosomal subunit
+Supramolecule #1: MERS-CoV Nsp1 bound to human 40S ribosomal subunit
+Macromolecule #1: 18S rRNA
+Macromolecule #2: 40S ribosomal protein SA
+Macromolecule #3: 40S ribosomal protein S3a
+Macromolecule #4: 40S ribosomal protein S2
+Macromolecule #5: 40S ribosomal protein S3
+Macromolecule #6: 40S ribosomal protein S4, X isoform
+Macromolecule #7: 40S ribosomal protein S5
+Macromolecule #8: 40S ribosomal protein S6
+Macromolecule #9: 40S ribosomal protein S7
+Macromolecule #10: 40S ribosomal protein S8
+Macromolecule #11: 40S ribosomal protein S9
+Macromolecule #12: 40S ribosomal protein S10
+Macromolecule #13: 40S ribosomal protein S11
+Macromolecule #14: 40S ribosomal protein S12
+Macromolecule #15: 40S ribosomal protein S13
+Macromolecule #16: 40S ribosomal protein S14
+Macromolecule #17: 40S ribosomal protein S15
+Macromolecule #18: 40S ribosomal protein S16
+Macromolecule #19: 40S ribosomal protein S17
+Macromolecule #20: 40S ribosomal protein S18
+Macromolecule #21: 40S ribosomal protein S19
+Macromolecule #22: 40S ribosomal protein S20
+Macromolecule #23: 40S ribosomal protein S21
+Macromolecule #24: 40S ribosomal protein S15a
+Macromolecule #25: 40S ribosomal protein S23
+Macromolecule #26: 40S ribosomal protein S24
+Macromolecule #27: 40S ribosomal protein S25
+Macromolecule #28: 40S ribosomal protein S26
+Macromolecule #29: 40S ribosomal protein S27
+Macromolecule #30: 40S ribosomal protein S28
+Macromolecule #31: 40S ribosomal protein S29
+Macromolecule #32: FAU ubiquitin-like and ribosomal protein S30
+Macromolecule #33: Ubiquitin-40S ribosomal protein S27a
+Macromolecule #34: Receptor of activated protein C kinase 1
+Macromolecule #35: 60S ribosomal protein L41
+Macromolecule #36: Replicase polyprotein 1ab
+Macromolecule #37: MAGNESIUM ION
+Macromolecule #38: ZINC ION
+Macromolecule #39: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 81000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)