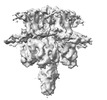
登録情報 データベース : EMDB / ID : EMD-40510タイトル KCNQ1 with voltage sensor in the down conformation 複合体 : Complex of KCNQ1 (Kv7.1) channel bound to calmodulin-Ca2+タンパク質・ペプチド : Potassium voltage-gated channel subfamily KQT member 1タンパク質・ペプチド : Calmodulin-1 / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / / 解像度 : 6.8 Å Mandala VS / MacKinnon R 資金援助 Organization Grant number 国 Howard Hughes Medical Institute (HHMI)
ジャーナル : Proc Natl Acad Sci U S A / 年 : 2023タイトル : The membrane electric field regulates the PIP-binding site to gate the KCNQ1 channel.著者 : Venkata Shiva Mandala / Roderick MacKinnon / 要旨 : Voltage-dependent ion channels underlie the propagation of action potentials and other forms of electrical activity in cells. In these proteins, voltage sensor domains (VSDs) regulate opening and ... Voltage-dependent ion channels underlie the propagation of action potentials and other forms of electrical activity in cells. In these proteins, voltage sensor domains (VSDs) regulate opening and closing of the pore through the displacement of their positive-charged S4 helix in response to the membrane voltage. The movement of S4 at hyperpolarizing membrane voltages in some channels is thought to directly clamp the pore shut through the S4-S5 linker helix. The KCNQ1 channel (also known as K7.1), which is important for heart rhythm, is regulated not only by membrane voltage but also by the signaling lipid phosphatidylinositol 4,5-bisphosphate (PIP). KCNQ1 requires PIP to open and to couple the movement of S4 in the VSD to the pore. To understand the mechanism of this voltage regulation, we use cryogenic electron microscopy to visualize the movement of S4 in the human KCNQ1 channel in lipid membrane vesicles with a voltage difference across the membrane, i.e., an applied electric field in the membrane. Hyperpolarizing voltages displace S4 in such a manner as to sterically occlude the PIP-binding site. Thus, in KCNQ1, the voltage sensor acts primarily as a regulator of PIP binding. The voltage sensors' influence on the channel's gate is indirect through the reaction sequence: voltage sensor movement → alter PIP ligand affinity → alter pore opening. 履歴 登録 2023年4月16日 - ヘッダ(付随情報) 公開 2023年5月31日 - マップ公開 2023年5月31日 - 更新 2025年6月4日 - 現状 2025年6月4日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報
 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト)
Homo sapiens (ヒト) データ登録者
データ登録者 米国, 1件
米国, 1件  引用
引用 ジャーナル: Proc Natl Acad Sci U S A / 年: 2023
ジャーナル: Proc Natl Acad Sci U S A / 年: 2023
 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_40510.map.gz
emd_40510.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-40510-v30.xml
emd-40510-v30.xml emd-40510.xml
emd-40510.xml EMDBヘッダ
EMDBヘッダ emd_40510.png
emd_40510.png emd-40510.cif.gz
emd-40510.cif.gz emd_40510_additional_1.map.gz
emd_40510_additional_1.map.gz emd_40510_half_map_1.map.gz
emd_40510_half_map_1.map.gz emd_40510_half_map_2.map.gz
emd_40510_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-40510
http://ftp.pdbj.org/pub/emdb/structures/EMD-40510 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40510
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40510 emd_40510_validation.pdf.gz
emd_40510_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_40510_full_validation.pdf.gz
emd_40510_full_validation.pdf.gz emd_40510_validation.xml.gz
emd_40510_validation.xml.gz emd_40510_validation.cif.gz
emd_40510_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-40510
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-40510 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-40510
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-40510 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_40510.map.gz / 形式: CCP4 / 大きさ: 20.8 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_40510.map.gz / 形式: CCP4 / 大きさ: 20.8 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素 Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 ムービー
ムービー コントローラー
コントローラー





























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)