+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of OSCA1.2-DOPC-1:50-betaCD state | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | OSCA/TMEM63 channel / mechanosensitive channel / PLANT PROTEIN | ||||||||||||
| Biological species |  | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 6.28 Å | ||||||||||||
 Authors Authors | Zhang Y / Han Y | ||||||||||||
| Funding support |  China, China,  Australia, 3 items Australia, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Mechanical activation opens a lipid-lined pore in OSCA ion channels. Authors: Yaoyao Han / Zijing Zhou / Ruitao Jin / Fei Dai / Yifan Ge / Xisan Ju / Xiaonuo Ma / Sitong He / Ling Yuan / Yingying Wang / Wei Yang / Xiaomin Yue / Zhongwen Chen / Yadong Sun / Ben Corry / ...Authors: Yaoyao Han / Zijing Zhou / Ruitao Jin / Fei Dai / Yifan Ge / Xisan Ju / Xiaonuo Ma / Sitong He / Ling Yuan / Yingying Wang / Wei Yang / Xiaomin Yue / Zhongwen Chen / Yadong Sun / Ben Corry / Charles D Cox / Yixiao Zhang /   Abstract: OSCA/TMEM63 channels are the largest known family of mechanosensitive channels, playing critical roles in plant and mammalian mechanotransduction. Here we determined 44 cryogenic electron microscopy ...OSCA/TMEM63 channels are the largest known family of mechanosensitive channels, playing critical roles in plant and mammalian mechanotransduction. Here we determined 44 cryogenic electron microscopy structures of OSCA/TMEM63 channels in different environments to investigate the molecular basis of OSCA/TMEM63 channel mechanosensitivity. In nanodiscs, we mimicked increased membrane tension and observed a dilated pore with membrane access in one of the OSCA1.2 subunits. In liposomes, we captured the fully open structure of OSCA1.2 in the inside-in orientation, in which the pore shows a large lateral opening to the membrane. Unusually for ion channels, structural, functional and computational evidence supports the existence of a 'proteo-lipidic pore' in which lipids act as a wall of the ion permeation pathway. In the less tension-sensitive homologue OSCA3.1, we identified an 'interlocking' lipid tightly bound in the central cleft, keeping the channel closed. Mutation of the lipid-coordinating residues induced OSCA3.1 activation, revealing a conserved open conformation of OSCA channels. Our structures provide a global picture of the OSCA channel gating cycle, uncover the importance of bound lipids and show that each subunit can open independently. This expands both our understanding of channel-mediated mechanotransduction and channel pore formation, with important mechanistic implications for the TMEM16 and TMC protein families. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_38722.map.gz emd_38722.map.gz | 31.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-38722-v30.xml emd-38722-v30.xml emd-38722.xml emd-38722.xml | 13 KB 13 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_38722.png emd_38722.png | 53.4 KB | ||
| Filedesc metadata |  emd-38722.cif.gz emd-38722.cif.gz | 3.9 KB | ||
| Others |  emd_38722_half_map_1.map.gz emd_38722_half_map_1.map.gz emd_38722_half_map_2.map.gz emd_38722_half_map_2.map.gz | 30.7 MB 30.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-38722 http://ftp.pdbj.org/pub/emdb/structures/EMD-38722 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38722 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38722 | HTTPS FTP |
-Related structure data
| Related structure data |  8xajC  8xngC  8xryC  8xs0C  8xs4C  8xs5C  8xvxC  8xvyC  8xvzC  8xw0C  8xw1C  8xw2C  8xw3C  8xw4C C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_38722.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_38722.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.055 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_38722_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
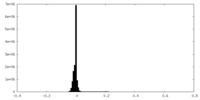
| Density Histograms |
-Half map: #2
| File | emd_38722_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
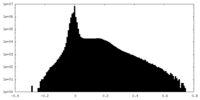
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of OSCA1.2-DOPC-1:50-betaCD state
| Entire | Name: Cryo-EM structure of OSCA1.2-DOPC-1:50-betaCD state |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of OSCA1.2-DOPC-1:50-betaCD state
| Supramolecule | Name: Cryo-EM structure of OSCA1.2-DOPC-1:50-betaCD state / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.9 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 49.41 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.4 µm / Nominal defocus min: 1.4000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 6.28 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 72614 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)