+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-3678 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the yeast R2TP complex | |||||||||
 Map data Map data | Structure of the yeast R2TP complex | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationTTT Hsp90 cochaperone complex / R2TP complex / Swr1 complex / Ino80 complex / box C/D snoRNP assembly / NuA4 histone acetyltransferase complex / 3'-5' DNA helicase activity / DNA helicase activity / rRNA processing / 5'-3' DNA helicase activity ...TTT Hsp90 cochaperone complex / R2TP complex / Swr1 complex / Ino80 complex / box C/D snoRNP assembly / NuA4 histone acetyltransferase complex / 3'-5' DNA helicase activity / DNA helicase activity / rRNA processing / 5'-3' DNA helicase activity / DNA helicase / protein stabilization / chromatin remodeling / DNA repair / regulation of transcription by RNA polymerase II / regulation of DNA-templated transcription / chromatin / ATP hydrolysis activity / ATP binding / nucleus Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 8.37 Å | |||||||||
 Authors Authors | Rivera-Calzada A / Pal M / Munoz-Hernandez H / Luque-Ortega JR / Gil-Carton D / Degliesposti G / Skehel JM / Prodromou C / Pearl LH / Llorca O | |||||||||
 Citation Citation |  Journal: Structure / Year: 2017 Journal: Structure / Year: 2017Title: The Structure of the R2TP Complex Defines a Platform for Recruiting Diverse Client Proteins to the HSP90 Molecular Chaperone System. Authors: Angel Rivera-Calzada / Mohinder Pal / Hugo Muñoz-Hernández / Juan R Luque-Ortega / David Gil-Carton / Gianluca Degliesposti / J Mark Skehel / Chrisostomos Prodromou / Laurence H Pearl / Oscar Llorca /   Abstract: The R2TP complex, comprising the Rvb1p-Rvb2p AAA-ATPases, Tah1p, and Pih1p in yeast, is a specialized Hsp90 co-chaperone required for the assembly and maturation of multi-subunit complexes. These ...The R2TP complex, comprising the Rvb1p-Rvb2p AAA-ATPases, Tah1p, and Pih1p in yeast, is a specialized Hsp90 co-chaperone required for the assembly and maturation of multi-subunit complexes. These include the small nucleolar ribonucleoproteins, RNA polymerase II, and complexes containing phosphatidylinositol-3-kinase-like kinases. The structure and stoichiometry of yeast R2TP and how it couples to Hsp90 are currently unknown. Here, we determine the 3D organization of yeast R2TP using sedimentation velocity analysis and cryo-electron microscopy. The 359-kDa complex comprises one Rvb1p/Rvb2p hetero-hexamer with domains II (DIIs) forming an open basket that accommodates a single copy of Tah1p-Pih1p. Tah1p-Pih1p binding to multiple DII domains regulates Rvb1p/Rvb2p ATPase activity. Using domain dissection and cross-linking mass spectrometry, we identified a unique region of Pih1p that is essential for interaction with Rvb1p/Rvb2p. These data provide a structural basis for understanding how R2TP couples an Hsp90 dimer to a diverse set of client proteins and complexes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_3678.map.gz emd_3678.map.gz | 3.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-3678-v30.xml emd-3678-v30.xml emd-3678.xml emd-3678.xml | 17.7 KB 17.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_3678.png emd_3678.png | 305.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-3678 http://ftp.pdbj.org/pub/emdb/structures/EMD-3678 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3678 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3678 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_3678.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_3678.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Structure of the yeast R2TP complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.34 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
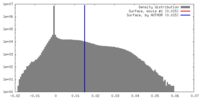
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Yeast R2TP complex
| Entire | Name: Yeast R2TP complex |
|---|---|
| Components |
|
-Supramolecule #1: Yeast R2TP complex
| Supramolecule | Name: Yeast R2TP complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Molecular weight | Experimental: 359 KDa |
-Macromolecule #1: Rvb1p
| Macromolecule | Name: Rvb1p / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO / EC number: DNA helicase |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MGSSHHHHHH SSGLVPRGSH MMVAISEVKE NPGVNSSNSG AVTRTAAHTH IKGLGLDESG VAKRVEGGFV GQIEAREACG V IVDLIKAK KMSGRAILLA GGPSTGKTAL ALAISQELGP KVPFCPLVGS ELYSVEVKKT ET LMENFRR AIGLRIKETK EVYEGEVTEL ...String: MGSSHHHHHH SSGLVPRGSH MMVAISEVKE NPGVNSSNSG AVTRTAAHTH IKGLGLDESG VAKRVEGGFV GQIEAREACG V IVDLIKAK KMSGRAILLA GGPSTGKTAL ALAISQELGP KVPFCPLVGS ELYSVEVKKT ET LMENFRR AIGLRIKETK EVYEGEVTEL TPEDAENPLG GYGKTISHVI VGLKSAKGTK TLR LDPTIY ESIQREKVSI GDVIYIEANT GAVKRVGRSD AYATEFDLET EEYVPLPKGE VHKK KEIVQ DVTLHDLDVA NARPQGGQDV ISMMGQLLKP KKTEITEKLR QEVNKVVAKY IDQGV AELI PGVLFIDEVN MLDIEIFTYL NKALESNIAP VVVLASNRGM TTVRGTEDVI SPHGVP PDL IDRLLIVRTL PYDKDEIRTI IERRATVERL QVESSALDLL ATMGTETSLR YALQLLA PC GILAQTSNRK EIVVNDVNEA KLLFLDAKRS TKILETSANY L |
-Macromolecule #2: Rvb2p
| Macromolecule | Name: Rvb2p / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO / EC number: DNA helicase |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MEQKLISEED LLRSEEQKLI SEEDLLRSEE QKLISEEDLL RSE SRGSHH HHHHLEVLFQ GPAS MSIQT SDPNETSDLK SLSLIAAHSH ITGLGLDENL QPRPTSEGMV GQLQARRAAG VILKM VQNG TIAGRAVLVA GPPSTGKTAL AMGVSQSLGK DVPFTAIAGS ...String: MEQKLISEED LLRSEEQKLI SEEDLLRSEE QKLISEEDLL RSE SRGSHH HHHHLEVLFQ GPAS MSIQT SDPNETSDLK SLSLIAAHSH ITGLGLDENL QPRPTSEGMV GQLQARRAAG VILKM VQNG TIAGRAVLVA GPPSTGKTAL AMGVSQSLGK DVPFTAIAGS EIFSLELSKT EALTQA FRK SIGIKIKEET ELIEGEVVEI QIDRSITGGH KQGKLTIKTT DMETIYELGN KMIDGLT KE KVLAGDVISI DKASGKITKL GRSFARSRDY DAMGADTRFV QCPEGELQKR KTVVHTVS L HEIDVINSRT QGFLALFTGD TGEIRSEVRD QINTKVAEWK EEGKAEIVPG VLFIDEVHM LDIECFSFIN RALEDEFAPI VMMATNRGVS KTRGTNYKSP HGLPLDLLDR SIIITTKSYN EQEIKTILS IRAQEEEVEL SSDALDLLTK TGVETSLRYS SNLISVAQQI AMKRKNNTVE V EDVKRAYL LFLDSARSVK YVQENESQYI DDQGNVQISI AKSADPDAMD TTE |
-Macromolecule #3: Pih1p
| Macromolecule | Name: Pih1p / type: protein_or_peptide / ID: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MSHHHHHHMA DFLLRPIKQR HRNEDKYVSV DAADGSVSKI EPIADFVIKT KLLSANGPEK LQDGRKVFIN VCHSPLVPKP EVDFNARIVF PLIIQNEWEI PIITSCYRMD HDKKGQECYV WDCCINSDCS RWICDDIQLR EILVEWCLES CEIRDSVVLC RDRIAFPKMK ...String: MSHHHHHHMA DFLLRPIKQR HRNEDKYVSV DAADGSVSKI EPIADFVIKT KLLSANGPEK LQDGRKVFIN VCHSPLVPKP EVDFNARIVF PLIIQNEWEI PIITSCYRMD HDKKGQECYV WDCCINSDCS RWICDDIQLR EILVEWCLES CEIRDSVVLC RDRIAFPKMK KKGAELPALE VLNDELHQDY KAKMHKIIEE EAGDPMSILR GRNDDGDDNN DPDDGTLPPL FPIENKISGA KIEEIDKNEI AHRNLKQAPA PAPAPHEQQE DVPEYEVKMK RFKGAAYKLR ILIENKAPNS KPDRFSPSYN FAENILYING KLSIPLPRDI VVNAADIKIF HIRKERTLYI YI |
-Macromolecule #4: Tah1p
| Macromolecule | Name: Tah1p / type: protein_or_peptide / ID: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MGSSHHHHHH WSHPQFEKGG GSGGGSGGSS AWSHPQFEKL EVLFQGPHMM SQFEKQKEQG NSLFKQGLY REAVHCYDQL ITAQPQNPVG YSNKAMALIK LGEYTQAIQM CQQGLRYTST AEHVAIRSKL QYRLELAQGA VGSVQIPVVE VDELPEGYDR S |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.45 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.8 Component:
| ||||||||||||||||||
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY ARRAY / Pretreatment - Type: GLOW DISCHARGE | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Frames/image: 2-19 / Average exposure time: 12.0 sec. / Average electron dose: 48.0 e/Å2 Details: Data was collected at two different tilts: 0 and 35 degrees. |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 39000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: FLEXIBLE FIT |
|---|
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)