[English] 日本語
 Yorodumi
Yorodumi- EMDB-35524: Cryo-EM structure of the eicosapentaenoic acid bound GPR120-Gi1 c... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the eicosapentaenoic acid bound GPR120-Gi1 complex(mask on receptor) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.91 Å | |||||||||
 Authors Authors | Mao C / Xiao P / Tao X / Qin J / He Q / Zhang C / Yu X / Zhang Y / Sun J | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2023 Journal: Science / Year: 2023Title: Unsaturated bond recognition leads to biased signal in a fatty acid receptor. Authors: Chunyou Mao / Peng Xiao / Xiao-Na Tao / Jiao Qin / Qing-Tao He / Chao Zhang / Sheng-Chao Guo / Ya-Qin Du / Li-Nan Chen / Dan-Dan Shen / Zhi-Shuai Yang / Han-Qiong Zhang / Shen-Ming Huang / ...Authors: Chunyou Mao / Peng Xiao / Xiao-Na Tao / Jiao Qin / Qing-Tao He / Chao Zhang / Sheng-Chao Guo / Ya-Qin Du / Li-Nan Chen / Dan-Dan Shen / Zhi-Shuai Yang / Han-Qiong Zhang / Shen-Ming Huang / Yong-Hao He / Jie Cheng / Ya-Ni Zhong / Pan Shang / Jun Chen / Dao-Lai Zhang / Qian-Lang Wang / Mei-Xia Liu / Guo-Yu Li / Yongyuan Guo / H Eric Xu / Chuanxin Wang / Cheng Zhang / Shiqing Feng / Xiao Yu / Yan Zhang / Jin-Peng Sun /  Abstract: Individual free fatty acids (FAs) play important roles in metabolic homeostasis, many through engagement with more than 40G protein-coupled receptors. Searching for receptors to sense beneficial ...Individual free fatty acids (FAs) play important roles in metabolic homeostasis, many through engagement with more than 40G protein-coupled receptors. Searching for receptors to sense beneficial omega-3 FAs of fish oil enabled the identification of GPR120, which is involved in a spectrum of metabolic diseases. Here, we report six cryo-electron microscopy structures of GPR120 in complex with FA hormones or TUG891 and G or G trimers. Aromatic residues inside the GPR120 ligand pocket were responsible for recognizing different double-bond positions of these FAs and connect ligand recognition to distinct effector coupling. We also investigated synthetic ligand selectivity and the structural basis of missense single-nucleotide polymorphisms. We reveal how GPR120 differentiates rigid double bonds and flexible single bonds. The knowledge gleaned here may facilitate rational drug design targeting to GPR120. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_35524.map.gz emd_35524.map.gz | 13.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-35524-v30.xml emd-35524-v30.xml emd-35524.xml emd-35524.xml | 13.5 KB 13.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_35524.png emd_35524.png | 54.6 KB | ||
| Others |  emd_35524_half_map_1.map.gz emd_35524_half_map_1.map.gz emd_35524_half_map_2.map.gz emd_35524_half_map_2.map.gz | 25 MB 25 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35524 http://ftp.pdbj.org/pub/emdb/structures/EMD-35524 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35524 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35524 | HTTPS FTP |
-Validation report
| Summary document |  emd_35524_validation.pdf.gz emd_35524_validation.pdf.gz | 666.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_35524_full_validation.pdf.gz emd_35524_full_validation.pdf.gz | 666.1 KB | Display | |
| Data in XML |  emd_35524_validation.xml.gz emd_35524_validation.xml.gz | 10.4 KB | Display | |
| Data in CIF |  emd_35524_validation.cif.gz emd_35524_validation.cif.gz | 12.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35524 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35524 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35524 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35524 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_35524.map.gz / Format: CCP4 / Size: 27 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_35524.map.gz / Format: CCP4 / Size: 27 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.014 Å | ||||||||||||||||||||||||||||||||||||
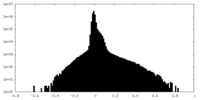
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_35524_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
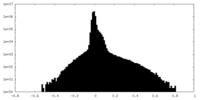
| Density Histograms |
-Half map: #2
| File | emd_35524_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
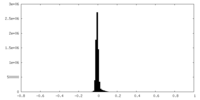
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of the eicosapentaenoic acid bound GPR120-Gi1 c...
| Entire | Name: Cryo-EM structure of the eicosapentaenoic acid bound GPR120-Gi1 complex(mask on receptor) |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of the eicosapentaenoic acid bound GPR120-Gi1 c...
| Supramolecule | Name: Cryo-EM structure of the eicosapentaenoic acid bound GPR120-Gi1 complex(mask on receptor) type: complex / ID: 1 / Chimera: Yes / Parent: 0 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 52.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.91 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 880881 |
|---|---|
| Initial angle assignment | Type: RANDOM ASSIGNMENT |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)