[English] 日本語
 Yorodumi
Yorodumi- EMDB-35213: Cryo-EM structure of Pseudomonas aeruginosa FtsE(E163Q)X/EnvC/Ami... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of Pseudomonas aeruginosa FtsE(E163Q)X/EnvC/AmiB complex with ATP in peptidisc | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | type VII ABC transporter / divisome / PG hydrolysis / MEMBRANE PROTEIN | |||||||||
| Biological species |  | |||||||||
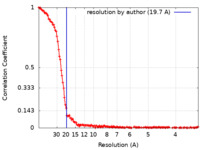
| Method | single particle reconstruction / cryo EM / Resolution: 19.7 Å | |||||||||
 Authors Authors | Xu X / Li J / Luo M | |||||||||
| Funding support |  Singapore, 1 items Singapore, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2023 Journal: Proc Natl Acad Sci U S A / Year: 2023Title: Mechanistic insights into the regulation of cell wall hydrolysis by FtsEX and EnvC at the bacterial division site. Authors: Xin Xu / Jianwei Li / Wan-Zhen Chua / Martin A Pages / Jian Shi / Juan A Hermoso / Thomas Bernhardt / Lok-To Sham / Min Luo /    Abstract: The peptidoglycan (PG) cell wall produced by the bacterial division machinery is initially shared between the daughters and must be split to promote cell separation and complete division. In gram- ...The peptidoglycan (PG) cell wall produced by the bacterial division machinery is initially shared between the daughters and must be split to promote cell separation and complete division. In gram-negative bacteria, enzymes that cleave PG called amidases play major roles in the separation process. To prevent spurious cell wall cleavage that can lead to cell lysis, amidases like AmiB are autoinhibited by a regulatory helix. Autoinhibition is relieved at the division site by the activator EnvC, which is in turn regulated by the ATP-binding cassette (ABC) transporter-like complex called FtsEX. EnvC is also known to be autoinhibited by a regulatory helix (RH), but how its activity is modulated by FtsEX and the mechanism by which it activates the amidases have remained unclear. Here, we investigated this regulation by determining the structure of FtsEX alone with or without bound ATP, in complex with EnvC, and in a FtsEX-EnvC-AmiB supercomplex. In combination with biochemical studies, the structures reveal that ATP binding is likely to activate FtsEX-EnvC and promote its association with AmiB. Furthermore, the AmiB activation mechanism is shown to involve a RH rearrangement. In the activated state of the complex, the inhibitory helix of EnvC is released, freeing it to associate with the RH of AmiB, which liberates its active site for PG cleavage. These regulatory helices are found in many EnvC proteins and amidases throughout gram-negative bacteria, suggesting that the activation mechanism is broadly conserved and a potential target for lysis-inducing antibiotics that misregulate the complex. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_35213.map.gz emd_35213.map.gz | 203.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-35213-v30.xml emd-35213-v30.xml emd-35213.xml emd-35213.xml | 16.9 KB 16.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_35213_fsc.xml emd_35213_fsc.xml | 17.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_35213.png emd_35213.png | 20.1 KB | ||
| Others |  emd_35213_half_map_1.map.gz emd_35213_half_map_1.map.gz emd_35213_half_map_2.map.gz emd_35213_half_map_2.map.gz | 200.2 MB 200.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35213 http://ftp.pdbj.org/pub/emdb/structures/EMD-35213 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35213 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35213 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_35213.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_35213.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.716 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_35213_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_35213_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : FtsEE163QX/EnvC/AmiB-ATP
| Entire | Name: FtsEE163QX/EnvC/AmiB-ATP |
|---|---|
| Components |
|
-Supramolecule #1: FtsEE163QX/EnvC/AmiB-ATP
| Supramolecule | Name: FtsEE163QX/EnvC/AmiB-ATP / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 216 KDa |
-Macromolecule #1: FstEE163Q
| Macromolecule | Name: FstEE163Q / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MIRFEQVGKR YPNGHVGLHE VSFRVHRGEI LFVTGHSGAG KSTLLRLILA MERPTSGKLL LGGQDLGRIT TAQIPFLRRQ IGVVFQNHQL LTDRTVADNI ALPLQILGMP KPEIAKRVAS ALERVNLKEK GEALPSDLST GQQQRVGIAR AIVHQPALLL ADQPTGNLDP ...String: MIRFEQVGKR YPNGHVGLHE VSFRVHRGEI LFVTGHSGAG KSTLLRLILA MERPTSGKLL LGGQDLGRIT TAQIPFLRRQ IGVVFQNHQL LTDRTVADNI ALPLQILGMP KPEIAKRVAS ALERVNLKEK GEALPSDLST GQQQRVGIAR AIVHQPALLL ADQPTGNLDP RLASEIMGVF EDINRLGTTV LIASHDLALI ARMRHRMLTL QRGRIIADRE DEA |
-Macromolecule #2: FtsX
| Macromolecule | Name: FtsX / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MSANDLPRGP EEGAPERKTR EKPSQEQTDW SGSFSAYLES HRASLVDSLR RLFGHPFGSF FTCLVMGITL SLPMGLSLLL NNVERLGGSW QRAAQISLFL DLKTSENQGQ DLREQIERLP DVIEAQLISR EQALSELQEQ SGLGEALKEL PENPLPPVIS VTPKQIDRAG ...String: MSANDLPRGP EEGAPERKTR EKPSQEQTDW SGSFSAYLES HRASLVDSLR RLFGHPFGSF FTCLVMGITL SLPMGLSLLL NNVERLGGSW QRAAQISLFL DLKTSENQGQ DLREQIERLP DVIEAQLISR EQALSELQEQ SGLGEALKEL PENPLPPVIS VTPKQIDRAG LEALRQQLAE LPHVQQAQLD LVWVERLSAI LKLGERFVFG LTILLVLTLL LVVGNTIRLH IENRRNEIEV IKLVGGTDGY VRRPFLYMGA LYGLGAGILS WALLAYSLNW LNGSVVNLSG LYGSDFGLQG VPLDDGLSLT VGAVLLGWVG AWLAVARHLR ELAPR |
-Macromolecule #3: EnvC
| Macromolecule | Name: EnvC / type: protein_or_peptide / ID: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: DERADTQRQL EQTQKDIGEL KKLLDGIQQE KSGVQKQLKS TETEMGDLEK QIKALQDELD KSEAELKRLD GEKKKLQDAR IEQQRLLAIQ ARAAYQSGRE EYLKLLLNQE HPEKFSRTLT YYDYINKARL EQLASFNETL RQLANVEQDI SAQKAEQLSK QGELDSRREA ...String: DERADTQRQL EQTQKDIGEL KKLLDGIQQE KSGVQKQLKS TETEMGDLEK QIKALQDELD KSEAELKRLD GEKKKLQDAR IEQQRLLAIQ ARAAYQSGRE EYLKLLLNQE HPEKFSRTLT YYDYINKARL EQLASFNETL RQLANVEQDI SAQKAEQLSK QGELDSRREA LAATRKERQQ ALAKLNSDYR ERDQKLKSRQ QDQAELAKVL RTIEETLARQ AREAAAAAER ERQRALAAER ERARQQQAAP GRVTSPPREP APGPLVSSTG AVYGGAFGSA RGKLPWPVNG RVVARFGSQR GDDPRAKWDG VLISASAGST VRAVHGGRVV FADWLRGAGL LVILDHGGGY LSLYGHNQSL LKDAGDTVKA GDPIATVGTS GGQSSPAVYF AIRHQGRPAD PTTWCRAQG |
-Macromolecule #4: AmiB
| Macromolecule | Name: AmiB / type: protein_or_peptide / ID: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: QIKSVRIWRA PDNTRLVFDL SGPVQHSLFT LAAPNRIVID VSGAQLATQL NGLKLGNTPI TAVRSAQRTP NDLRMVLDLS AQVTPKSFVL PPNQQYGNRL VVDLYDQGAD LTPDVPATPT PSVPVTPVTP TQPVAKLPLP TKGGTRDIVI AIDAGHGGED PGALGPGGLH ...String: QIKSVRIWRA PDNTRLVFDL SGPVQHSLFT LAAPNRIVID VSGAQLATQL NGLKLGNTPI TAVRSAQRTP NDLRMVLDLS AQVTPKSFVL PPNQQYGNRL VVDLYDQGAD LTPDVPATPT PSVPVTPVTP TQPVAKLPLP TKGGTRDIVI AIDAGHGGED PGALGPGGLH EKNITLSIAR ELQRQINQVR GYRAELTRTG DYFIPLRKRT EIARKKGADL FVSIHADAAP SRSAFGASVF ALSDRGATSE TARWLADSEN RSDLIGGDGS VSLGDKDQML AGVLLDLSMT ATLSSSLDVG HKVLTNVGRI TSLHKRRVEQ AGFMVLKSPD IPSILVETGF ISNVNESRKL ASASHQQALA RSITSGIRQY FQQSPPPGTY IASLRAQGKL SMGPREHVVR PGETLAMIAQ RYEVSMAALR SSNSLSSDNL KVGQALSIPS TALAAQ |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3 mg/mL |
|---|---|
| Buffer | pH: 7.5 / Details: 25 mM Tris pH 7.5, 150 mM NaCl, 2mM ATP/MgCl2 |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 293.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 1.2 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)