+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human propionyl-coenzyme A carboxylase | |||||||||
 Map data Map data | Human propionyl-coenzyme A carboxylase | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Cryo-EM / PCC / Mitochondria / CYTOSOLIC PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationshort-chain fatty acid catabolic process / branched-chain amino acid metabolic process / Propionyl-CoA catabolism / propionyl-CoA carboxylase / Defective HLCS causes multiple carboxylase deficiency / propionyl-CoA carboxylase activity / Biotin transport and metabolism / biotin binding / catalytic complex / Mitochondrial protein degradation ...short-chain fatty acid catabolic process / branched-chain amino acid metabolic process / Propionyl-CoA catabolism / propionyl-CoA carboxylase / Defective HLCS causes multiple carboxylase deficiency / propionyl-CoA carboxylase activity / Biotin transport and metabolism / biotin binding / catalytic complex / Mitochondrial protein degradation / fatty acid metabolic process / mitochondrial matrix / enzyme binding / mitochondrion / ATP binding / metal ion binding / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.2 Å | |||||||||
 Authors Authors | Su JY / Liu DS | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2025 Journal: Nat Struct Mol Biol / Year: 2025Title: Structural insight into synergistic activation of human 3-methylcrotonyl-CoA carboxylase. Authors: Jiayue Su / Xuyang Tian / Hang Cheng / Desheng Liu / Ziyi Wang / Shan Sun / Hong-Wei Wang / Sen-Fang Sui /   Abstract: The enzymes 3-methylcrotonyl-coenzyme A (CoA) carboxylase (MCC), pyruvate carboxylase and propionyl-CoA carboxylase belong to the biotin-dependent carboxylase family located in mitochondria. They ...The enzymes 3-methylcrotonyl-coenzyme A (CoA) carboxylase (MCC), pyruvate carboxylase and propionyl-CoA carboxylase belong to the biotin-dependent carboxylase family located in mitochondria. They participate in various metabolic pathways in human such as amino acid metabolism and tricarboxylic acid cycle. Many human diseases are caused by mutations in those enzymes but their structures have not been fully resolved so far. Here we report an optimized purification strategy to obtain high-resolution structures of intact human endogenous MCC, propionyl-CoA carboxylase and pyruvate carboxylase in different conformational states. We also determine the structures of MCC bound to different substrates. Analysis of MCC structures in different states reveals the mechanism of the substrate-induced, multi-element synergistic activation of MCC. These results provide important insights into the catalytic mechanism of the biotin-dependent carboxylase family and are of great value for the development of new drugs for the treatment of related diseases. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33729.map.gz emd_33729.map.gz | 90.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33729-v30.xml emd-33729-v30.xml emd-33729.xml emd-33729.xml | 15.4 KB 15.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_33729.png emd_33729.png | 142.2 KB | ||
| Filedesc metadata |  emd-33729.cif.gz emd-33729.cif.gz | 5.8 KB | ||
| Others |  emd_33729_half_map_1.map.gz emd_33729_half_map_1.map.gz emd_33729_half_map_2.map.gz emd_33729_half_map_2.map.gz | 165 MB 165 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33729 http://ftp.pdbj.org/pub/emdb/structures/EMD-33729 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33729 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33729 | HTTPS FTP |
-Related structure data
| Related structure data |  7ybuMC  8hwlC  8j4zC  8j73C  8j78C  8j7dC  8j7oC  8jakC  8jawC  8jxlC  8jxmC  8jxnC  8k2vC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_33729.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33729.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Human propionyl-coenzyme A carboxylase | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0825 Å | ||||||||||||||||||||||||||||||||||||
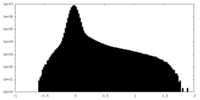
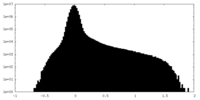
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Human propionyl-coenzyme A carboxylase half map A
| File | emd_33729_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Human propionyl-coenzyme A carboxylase half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
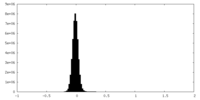
| Density Histograms |
-Half map: Human propionyl-coenzyme A carboxylase half map B
| File | emd_33729_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Human propionyl-coenzyme A carboxylase half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
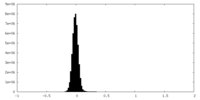
| Density Histograms |
- Sample components
Sample components
-Entire : Human propionyl-coenzyme A carboxylase heterododecomer
| Entire | Name: Human propionyl-coenzyme A carboxylase heterododecomer |
|---|---|
| Components |
|
-Supramolecule #1: Human propionyl-coenzyme A carboxylase heterododecomer
| Supramolecule | Name: Human propionyl-coenzyme A carboxylase heterododecomer type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Propionyl-CoA carboxylase alpha chain, mitochondrial
| Macromolecule | Name: Propionyl-CoA carboxylase alpha chain, mitochondrial / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO / EC number: propionyl-CoA carboxylase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 80.161922 KDa |
| Sequence | String: MAGFWVGTAP LVAAGRRGRW PPQQLMLSAA LRTLKHVLYY SRQCLMVSRN LGSVGYDPNE KTFDKILVAN RGEIACRVIR TCKKMGIKT VAIHSDVDAS SVHVKMADEA VCVGPAPTSK SYLNMDAIME AIKKTRAQAV HPGYGFLSEN KEFARCLAAE D VVFIGPDT ...String: MAGFWVGTAP LVAAGRRGRW PPQQLMLSAA LRTLKHVLYY SRQCLMVSRN LGSVGYDPNE KTFDKILVAN RGEIACRVIR TCKKMGIKT VAIHSDVDAS SVHVKMADEA VCVGPAPTSK SYLNMDAIME AIKKTRAQAV HPGYGFLSEN KEFARCLAAE D VVFIGPDT HAIQAMGDKI ESKLLAKKAE VNTIPGFDGV VKDAEEAVRI AREIGYPVMI KASAGGGGKG MRIAWDDEET RD GFRLSSQ EAASSFGDDR LLIEKFIDNP RHIEIQVLGD KHGNALWLNE RECSIQRRNQ KVVEEAPSIF LDAETRRAMG EQA VALARA VKYSSAGTVE FLVDSKKNFY FLEMNTRLQV EHPVTECITG LDLVQEMIRV AKGYPLRHKQ ADIRINGWAV ECRV YAEDP YKSFGLPSIG RLSQYQEPLH LPGVRVDSGI QPGSDISIYY DPMISKLITY GSDRTEALKR MADALDNYVI RGVTH NIAL LREVIINSRF VKGDISTKFL SDVYPDGFKG HMLTKSEKNQ LLAIASSLFV AFQLRAQHFQ ENSRMPVIKP DIANWE LSV KLHDKVHTVV ASNNGSVFSV EVDGSKLNVT STWNLASPLL SVSVDGTQRT VQCLSREAGG NMSIQFLGTV YKVNILT RL AAELNKFMLE KVTEDTSSVL RSPMPGVVVA VSVKPGDAVA EGQEICVIEA MKMQNSMTAG KTGTVKSVHC QAGDTVGE G DLLVELE UniProtKB: Propionyl-CoA carboxylase alpha chain, mitochondrial |
-Macromolecule #2: Propionyl-CoA carboxylase beta chain, mitochondrial
| Macromolecule | Name: Propionyl-CoA carboxylase beta chain, mitochondrial / type: protein_or_peptide / ID: 2 / Number of copies: 6 / Enantiomer: LEVO / EC number: propionyl-CoA carboxylase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 58.284488 KDa |
| Sequence | String: MAAALRVAAV GARLSVLASG LRAAVRSLCS QATSVNERIE NKRRTALLGG GQRRIDAQHK RGKLTARERI SLLLDPGSFV ESDMFVEHR CADFGMAADK NKFPGDSVVT GRGRINGRLV YVFSQDFTVF GGSLSGAHAQ KICKIMDQAI TVGAPVIGLN D SGGARIQE ...String: MAAALRVAAV GARLSVLASG LRAAVRSLCS QATSVNERIE NKRRTALLGG GQRRIDAQHK RGKLTARERI SLLLDPGSFV ESDMFVEHR CADFGMAADK NKFPGDSVVT GRGRINGRLV YVFSQDFTVF GGSLSGAHAQ KICKIMDQAI TVGAPVIGLN D SGGARIQE GVESLAGYAD IFLRNVTASG VIPQISLIMG PCAGGAVYSP ALTDFTFMVK DTSYLFITGP DVVKSVTNED VT QEELGGA KTHTTMSGVA HRAFENDVDA LCNLRDFFNY LPLSSQDPAP VRECHDPSDR LVPELDTIVP LESTKAYNMV DII HSVVDE REFFEIMPNY AKNIIVGFAR MNGRTVGIVG NQPKVASGCL DINSSVKGAR FVRFCDAFNI PLITFVDVPG FLPG TAQEY GGIIRHGAKL LYAFAEATVP KVTVITRKAY GGAYDVMSSK HLCGDTNYAW PTAEIAVMGA KGAVEIIFKG HENVE AAQA EYIEKFANPF PAAVRGFVDD IIQPSSTRAR ICCDLDVLAS KKVQRPWRKH ANIPL UniProtKB: Propionyl-CoA carboxylase beta chain, mitochondrial |
-Macromolecule #3: 5-(HEXAHYDRO-2-OXO-1H-THIENO[3,4-D]IMIDAZOL-6-YL)PENTANAL
| Macromolecule | Name: 5-(HEXAHYDRO-2-OXO-1H-THIENO[3,4-D]IMIDAZOL-6-YL)PENTANAL type: ligand / ID: 3 / Number of copies: 6 / Formula: BTI |
|---|---|
| Molecular weight | Theoretical: 228.311 Da |
| Chemical component information |  ChemComp-BTI: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.8 µm / Nominal defocus min: 1.3 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.2 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 99377 |
| Initial angle assignment | Type: RANDOM ASSIGNMENT |
| Final angle assignment | Type: COMMON LINE |
 Movie
Movie Controller
Controller






















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)