[English] 日本語
 Yorodumi
Yorodumi- EMDB-2792: Near-atomic resolution reconstruction of T. acidophilum archaeal ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2792 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Near-atomic resolution reconstruction of T. acidophilum archaeal 20S proteasome using a mid-range electron microscope operated at 200 kV | |||||||||
 Map data Map data | Reconstruction of T. acidophilum archaeal 20S proteasome | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Single-particle electron microscopy / Direct detectors / Near-atomic resolution | |||||||||
| Biological species |   Thermoplasma acidophilum (acidophilic) Thermoplasma acidophilum (acidophilic) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.2 Å | |||||||||
 Authors Authors | Campbell MG / Kearney BM / Cheng A / Potter CS / Johnson JE / Carragher B / Veesler D | |||||||||
 Citation Citation |  Journal: J Struct Biol / Year: 2014 Journal: J Struct Biol / Year: 2014Title: Near-atomic resolution reconstructions using a mid-range electron microscope operated at 200 kV. Abstract: A new era has begun for single particle cryo-electron microscopy (cryoEM) which can now compete with X-ray crystallography for determination of protein structures. The development of direct detectors ...A new era has begun for single particle cryo-electron microscopy (cryoEM) which can now compete with X-ray crystallography for determination of protein structures. The development of direct detectors constitutes a revolution that has led to a wave of near-atomic resolution cryoEM reconstructions. However, regardless of the sample studied, virtually all high-resolution reconstructions reported to date have been achieved using high-end microscopes. We demonstrate that the new generation of direct detectors coupled to a widely used mid-range electron microscope also enables obtaining cryoEM maps of sufficient quality for de novo modeling of protein structures of different sizes and symmetries. We provide an outline of the strategy used to achieve a 3.7 Å resolution reconstruction of Nudaurelia capensis ω virus and a 4.2 Å resolution reconstruction of the Thermoplasma acidophilum T20S proteasome. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2792.map.gz emd_2792.map.gz | 10.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2792-v30.xml emd-2792-v30.xml emd-2792.xml emd-2792.xml | 9.6 KB 9.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_2792.png emd_2792.png | 73.2 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2792 http://ftp.pdbj.org/pub/emdb/structures/EMD-2792 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2792 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2792 | HTTPS FTP |
-Validation report
| Summary document |  emd_2792_validation.pdf.gz emd_2792_validation.pdf.gz | 241.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_2792_full_validation.pdf.gz emd_2792_full_validation.pdf.gz | 240.7 KB | Display | |
| Data in XML |  emd_2792_validation.xml.gz emd_2792_validation.xml.gz | 5.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2792 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2792 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2792 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2792 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_2792.map.gz / Format: CCP4 / Size: 62.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2792.map.gz / Format: CCP4 / Size: 62.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Reconstruction of T. acidophilum archaeal 20S proteasome | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.25 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
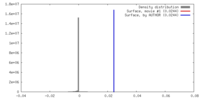
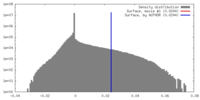
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Near-atomic resolution reconstruction of T. acidophilum archaeal ...
| Entire | Name: Near-atomic resolution reconstruction of T. acidophilum archaeal 20S proteasome using a mid-range electron microscope operated at 200 kV |
|---|---|
| Components |
|
-Supramolecule #1000: Near-atomic resolution reconstruction of T. acidophilum archaeal ...
| Supramolecule | Name: Near-atomic resolution reconstruction of T. acidophilum archaeal 20S proteasome using a mid-range electron microscope operated at 200 kV type: sample / ID: 1000 / Oligomeric state: D7 / Number unique components: 1 |
|---|---|
| Molecular weight | Theoretical: 700 KDa |
-Macromolecule #1: 20S proteasome
| Macromolecule | Name: 20S proteasome / type: protein_or_peptide / ID: 1 / Number of copies: 28 / Oligomeric state: 28mer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:   Thermoplasma acidophilum (acidophilic) Thermoplasma acidophilum (acidophilic) |
| Molecular weight | Theoretical: 690 KDa |
| Recombinant expression | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.21 mg/mL |
|---|---|
| Buffer | pH: 7.8 / Details: 20mM Tris, 150mM NaCl |
| Grid | Details: 1.2/1.3 C-Flat grid, plasma cleaned |
| Vitrification | Cryogen name: ETHANE / Chamber temperature: 95 K / Instrument: GATAN CRYOPLUNGE 3 / Details: Vitrification carried out at room temperature / Method: Blot for 2.5 seconds before plunging |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Alignment procedure | Legacy - Astigmatism: Objective lens astigmatism was corrected at 29,000 times magnification |
| Details | Collected in counting mode |
| Date | Oct 1, 2013 |
| Image recording | Category: CCD / Film or detector model: GATAN K2 (4k x 4k) / Number real images: 166 / Average electron dose: 38 e/Å2 Details: Each movie was acquired over 5 s and was comprised of 25 frames |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 40000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2 mm / Nominal defocus max: 3.3 µm / Nominal defocus min: 0.75 µm / Nominal magnification: 29000 |
| Sample stage | Specimen holder: Liquid nitrogen cooled / Specimen holder model: GATAN LIQUID NITROGEN |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | Initial data processing was performed using the Appion pipeline (including UCSF doseefgpu_driftcorr). Subsequent statistical refinement procedures and motion correction were performed using Relion. |
|---|---|
| CTF correction | Details: Each particle |
| Final reconstruction | Applied symmetry - Point group: D7 (2x7 fold dihedral) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 4.2 Å / Resolution method: OTHER / Software - Name: Relion / Number images used: 21818 |
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)