[English] 日本語
 Yorodumi
Yorodumi- EMDB-26128: Structure of the L. blandensis dGTPase H125A mutant bound to dGTP -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the L. blandensis dGTPase H125A mutant bound to dGTP | ||||||||||||
 Map data Map data | DeepEMhancer post-processed map | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | dGTPase / nucleotide binding / HYDROLASE | ||||||||||||
| Function / homology |  Function and homology information Function and homology information | ||||||||||||
| Biological species |  Leeuwenhoekiella blandensis MED217 (bacteria) Leeuwenhoekiella blandensis MED217 (bacteria) | ||||||||||||
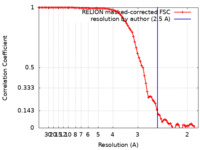
| Method | single particle reconstruction / cryo EM / Resolution: 2.5 Å | ||||||||||||
 Authors Authors | Klemm BP / Sikkema AP | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: J Biol Chem / Year: 2022 Journal: J Biol Chem / Year: 2022Title: High-resolution structures of the SAMHD1 dGTPase homolog from Leeuwenhoekiella blandensis reveal a novel mechanism of allosteric activation by dATP. Authors: Bradley P Klemm / Andrew P Sikkema / Allen L Hsu / James C Horng / Traci M Tanaka Hall / Mario J Borgnia / Roel M Schaaper /  Abstract: Deoxynucleoside triphosphate (dNTP) triphosphohydrolases (dNTPases) are important enzymes that may perform multiple functions in the cell, including regulating the dNTP pools and contributing to ...Deoxynucleoside triphosphate (dNTP) triphosphohydrolases (dNTPases) are important enzymes that may perform multiple functions in the cell, including regulating the dNTP pools and contributing to innate immunity against viruses. Among the homologs that are best studied are human sterile alpha motif and HD domain-containing protein 1 (SAMHD1), a tetrameric dNTPase, and the hexameric Escherichia coli dGTPase; however, it is unclear whether these are representative of all dNTPases given their wide distribution throughout life. Here, we investigated a hexameric homolog from the marine bacterium Leeuwenhoekiella blandensis, revealing that it is a dGTPase that is subject to allosteric activation by dATP, specifically. Allosteric regulation mediated solely by dATP represents a novel regulatory feature among dNTPases that may facilitate maintenance of cellular dNTP pools in L. blandensis. We present high-resolution X-ray crystallographic structures (1.80-2.26 Å) in catalytically important conformations as well as cryo-EM structures (2.1-2.7 Å) of the enzyme bound to dGTP and dATP ligands. The structures, the highest resolution cryo-EM structures of any SAMHD1-like dNTPase to date, reveal an intact metal-binding site with the dGTP substrate coordinated to three metal ions. These structural and biochemical data yield insights into the catalytic mechanism and support a conserved catalytic mechanism for the tetrameric and hexameric dNTPase homologs. We conclude that the allosteric activation by dATP appears to rely on structural connectivity between the allosteric and active sites, as opposed to the changes in oligomeric state upon ligand binding used by SAMHD1. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26128.map.gz emd_26128.map.gz | 40.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26128-v30.xml emd-26128-v30.xml emd-26128.xml emd-26128.xml | 26.5 KB 26.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_26128_fsc.xml emd_26128_fsc.xml | 8.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_26128.png emd_26128.png | 112.8 KB | ||
| Masks |  emd_26128_msk_1.map emd_26128_msk_1.map | 47.6 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-26128.cif.gz emd-26128.cif.gz | 6.7 KB | ||
| Others |  emd_26128_additional_1.map.gz emd_26128_additional_1.map.gz emd_26128_additional_2.map.gz emd_26128_additional_2.map.gz emd_26128_additional_3.map.gz emd_26128_additional_3.map.gz emd_26128_half_map_1.map.gz emd_26128_half_map_1.map.gz emd_26128_half_map_2.map.gz emd_26128_half_map_2.map.gz | 44 MB 36.5 MB 6.2 MB 36.7 MB 36.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26128 http://ftp.pdbj.org/pub/emdb/structures/EMD-26128 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26128 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26128 | HTTPS FTP |
-Related structure data
| Related structure data |  7tu7MC  7tu0C  7tu1C  7tu2C  7tu3C  7tu4C  7tu5C  7tu6C  7tu8C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26128.map.gz / Format: CCP4 / Size: 47.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26128.map.gz / Format: CCP4 / Size: 47.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | DeepEMhancer post-processed map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.932 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_26128_msk_1.map emd_26128_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: PHENIX auto-sharpened map
| File | emd_26128_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | PHENIX auto-sharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Full map from RELION refinement
| File | emd_26128_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Full map from RELION refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: RELION post-processed map
| File | emd_26128_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | RELION post-processed map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half-map 1
| File | emd_26128_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half-map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half-map 2
| File | emd_26128_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half-map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : dGTP triphosphohydrolase
| Entire | Name: dGTP triphosphohydrolase |
|---|---|
| Components |
|
-Supramolecule #1: dGTP triphosphohydrolase
| Supramolecule | Name: dGTP triphosphohydrolase / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Leeuwenhoekiella blandensis MED217 (bacteria) Leeuwenhoekiella blandensis MED217 (bacteria) |
-Macromolecule #1: dGTP triphosphohydrolase
| Macromolecule | Name: dGTP triphosphohydrolase / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO / EC number: dGTPase |
|---|---|
| Source (natural) | Organism:  Leeuwenhoekiella blandensis MED217 (bacteria) / Strain: CECT 7118 / CCUG 51940 / MED217 Leeuwenhoekiella blandensis MED217 (bacteria) / Strain: CECT 7118 / CCUG 51940 / MED217 |
| Molecular weight | Theoretical: 52.656176 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MHHHHHHSSG VDLGTENLYF QSNANWEHLL SLKRQGDTAK RLRIEQDDTR LGFEVDYDRI IFSAPFRSLQ DKTQVIPLSK TDFVHTRLT HSLEVSVVGR SLGRMVGKKL LEKYPHLEQV YGYKFNDFGA IVAAAALAHD IGNPPFGASG EKAIGEFFKN G YGKRYKDS ...String: MHHHHHHSSG VDLGTENLYF QSNANWEHLL SLKRQGDTAK RLRIEQDDTR LGFEVDYDRI IFSAPFRSLQ DKTQVIPLSK TDFVHTRLT HSLEVSVVGR SLGRMVGKKL LEKYPHLEQV YGYKFNDFGA IVAAAALAHD IGNPPFGASG EKAIGEFFKN G YGKRYKDS LTAKEYQDLI KFEGNANGFK VLSQSKPGAQ GGLRLSYATL GAFMKYPKES LPHKPSDHIA DKKYGFFQSE RA LFEDVAQ ELGLLKRSTT DDVSWSRHPL AYLVEAADDI CYTIIDFEDG INLGLIPEEY ALEYMVKLVG QTIDRNKYNA LQE TSDRVS YLRALAIGTL INESVDTFMK YEEEILAGTF DQSLIDKSNY QAQITDIINL SIERIYNSRE VIEKEIAGYE ILST LLEAR CRALDNNDTH YNQLIQQLLA PNDHSEKSLY ENLIQICAEV STMTDGKALR NYKKIKGLD UniProtKB: dGTP triphosphohydrolase |
-Macromolecule #2: 2'-DEOXYGUANOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: 2'-DEOXYGUANOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 2 / Number of copies: 6 / Formula: DGT |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information | 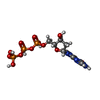 ChemComp-DGT: |
-Macromolecule #3: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 3 / Number of copies: 18 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| ||||||||||||
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 290 K / Instrument: LEICA EM GP / Details: 2.5 second blot time (front). | ||||||||||||
| Details | Hexamer concentration is listed. 10 mM dGTP was added to the peak fraction after gel filtration. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 3838 pixel / Digitization - Dimensions - Height: 3710 pixel / Number grids imaged: 1 / Number real images: 391 / Average exposure time: 8.4 sec. / Average electron dose: 54.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 45000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Details | Apo Cryo-EM model was fit into the EM map for building. dGTP ligands were initially built into the density using Coot. |
|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
| Output model |  PDB-7tu7: |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)