[English] 日本語
 Yorodumi
Yorodumi- EMDB-26080: CaKip3[2-482] - AMP-PNP in complex with a dolastatin-10-stabilize... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | CaKip3[2-482] - AMP-PNP in complex with a dolastatin-10-stabilized tubulin ring | ||||||||||||
 Map data Map data | Primary map, locally refined on the central asymmetric unit | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | Kip3 / kinesin / tubulin / dolastatin-10 / MOTOR PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationplus-end specific microtubule depolymerization / tubulin-dependent ATPase activity / microtubule plus-end directed mitotic chromosome migration / regulation of mitotic spindle elongation / meiotic sister chromatid segregation / mitotic spindle astral microtubule / mitotic chromosome movement towards spindle pole / mitotic spindle midzone / nuclear microtubule / nuclear migration along microtubule ...plus-end specific microtubule depolymerization / tubulin-dependent ATPase activity / microtubule plus-end directed mitotic chromosome migration / regulation of mitotic spindle elongation / meiotic sister chromatid segregation / mitotic spindle astral microtubule / mitotic chromosome movement towards spindle pole / mitotic spindle midzone / nuclear microtubule / nuclear migration along microtubule / microtubule plus-end / microtubule nucleation / plus-end-directed microtubule motor activity / Microtubule-dependent trafficking of connexons from Golgi to the plasma membrane / Resolution of Sister Chromatid Cohesion / Hedgehog 'off' state / Cilium Assembly / Intraflagellar transport / COPI-dependent Golgi-to-ER retrograde traffic / Mitotic Prometaphase / Carboxyterminal post-translational modifications of tubulin / RHOH GTPase cycle / EML4 and NUDC in mitotic spindle formation / Sealing of the nuclear envelope (NE) by ESCRT-III / Kinesins / PKR-mediated signaling / Separation of Sister Chromatids / The role of GTSE1 in G2/M progression after G2 checkpoint / Aggrephagy / mitotic spindle disassembly / microtubule depolymerization / RHO GTPases activate IQGAPs / RHO GTPases Activate Formins / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / MHC class II antigen presentation / Recruitment of NuMA to mitotic centrosomes / kinesin complex / COPI-mediated anterograde transport / microtubule-based movement / negative regulation of microtubule polymerization / establishment of mitotic spindle orientation / mitotic sister chromatid segregation / mitotic spindle assembly / microtubule-based process / mitotic spindle organization / structural constituent of cytoskeleton / kinetochore / microtubule cytoskeleton organization / mitotic cell cycle / microtubule cytoskeleton / microtubule binding / microtubule / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / GTPase activity / GTP binding / ATP hydrolysis activity / ATP binding / metal ion binding / nucleus / cytoplasm Similarity search - Function | ||||||||||||
| Biological species |  Candida albicans (yeast) / Candida albicans (yeast) /  | ||||||||||||
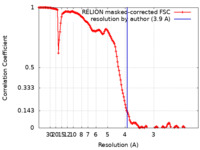
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | ||||||||||||
 Authors Authors | Benoit MPMH / Asenjo AB / Hunter B / Allingham JS / Sosa H | ||||||||||||
| Funding support |  United States, United States,  Canada, 3 items Canada, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Kinesin-8-specific loop-2 controls the dual activities of the motor domain according to tubulin protofilament shape. Authors: Byron Hunter / Matthieu P M H Benoit / Ana B Asenjo / Caitlin Doubleday / Daria Trofimova / Corey Frazer / Irsa Shoukat / Hernando Sosa / John S Allingham /   Abstract: Kinesin-8s are dual-activity motor proteins that can move processively on microtubules and depolymerize microtubule plus-ends, but their mechanism of combining these distinct activities remains ...Kinesin-8s are dual-activity motor proteins that can move processively on microtubules and depolymerize microtubule plus-ends, but their mechanism of combining these distinct activities remains unclear. We addressed this by obtaining cryo-EM structures (2.6-3.9 Å) of Candida albicans Kip3 in different catalytic states on the microtubule lattice and on a curved microtubule end mimic. We also determined a crystal structure of microtubule-unbound CaKip3-ADP (2.0 Å) and analyzed the biochemical activity of CaKip3 and kinesin-1 mutants. These data reveal that the microtubule depolymerization activity of kinesin-8 originates from conformational changes of its motor core that are amplified by dynamic contacts between its extended loop-2 and tubulin. On curved microtubule ends, loop-1 inserts into preceding motor domains, forming head-to-tail arrays of kinesin-8s that complement loop-2 contacts with curved tubulin and assist depolymerization. On straight tubulin protofilaments in the microtubule lattice, loop-2-tubulin contacts inhibit conformational changes in the motor core, but in the ADP-Pi state these contacts are relaxed, allowing neck-linker docking for motility. We propose that these tubulin shape-induced alternations between pro-microtubule-depolymerization and pro-motility kinesin states, regulated by loop-2, are the key to the dual activity of kinesin-8 motors. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26080.map.gz emd_26080.map.gz | 6.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26080-v30.xml emd-26080-v30.xml emd-26080.xml emd-26080.xml | 26 KB 26 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_26080_fsc.xml emd_26080_fsc.xml | 14.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_26080.png emd_26080.png | 188.6 KB | ||
| Masks |  emd_26080_msk_1.map emd_26080_msk_1.map emd_26080_msk_2.map emd_26080_msk_2.map emd_26080_msk_3.map emd_26080_msk_3.map emd_26080_msk_4.map emd_26080_msk_4.map | 274.6 MB 274.6 MB 274.6 MB 274.6 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-26080.cif.gz emd-26080.cif.gz | 8 KB | ||
| Others |  emd_26080_additional_1.map.gz emd_26080_additional_1.map.gz emd_26080_half_map_1.map.gz emd_26080_half_map_1.map.gz emd_26080_half_map_2.map.gz emd_26080_half_map_2.map.gz | 6.7 MB 219 MB 218.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26080 http://ftp.pdbj.org/pub/emdb/structures/EMD-26080 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26080 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26080 | HTTPS FTP |
-Related structure data
| Related structure data |  7tr3MC  7lffC  7tqxC  7tqyC  7tqzC  7tr0C  7tr1C  7tr2C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26080.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26080.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Primary map, locally refined on the central asymmetric unit | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.083 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_26080_msk_1.map emd_26080_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Mask #2
| File |  emd_26080_msk_2.map emd_26080_msk_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Mask #3
| File |  emd_26080_msk_3.map emd_26080_msk_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Mask #4
| File |  emd_26080_msk_4.map emd_26080_msk_4.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Map locally refined on the 3 central asymmetric units
| File | emd_26080_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Map locally refined on the 3 central asymmetric units | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map from the gold Standard refinement
| File | emd_26080_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map from the gold Standard refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map from the gold Standard refinement
| File | emd_26080_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map from the gold Standard refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : caKip3[2-482] - AMP-PNP in complex with a dolastatin-10-stabilize...
+Supramolecule #1: caKip3[2-482] - AMP-PNP in complex with a dolastatin-10-stabilize...
+Supramolecule #2: caKip3[2-482] - AMP-PNP
+Supramolecule #3: dolastatin-10 stabilized tubulin ring
+Macromolecule #1: Tubulin alpha-1B chain
+Macromolecule #2: Tubulin beta-2B chain
+Macromolecule #3: Kinesin-like protein
+Macromolecule #4: GUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #5: MAGNESIUM ION
+Macromolecule #6: GUANOSINE-5'-DIPHOSPHATE
+Macromolecule #7: dolastatin-10
+Macromolecule #8: PHOSPHOAMINOPHOSPHONIC ACID-ADENYLATE ESTER
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 6.8 Component:
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: UltrAuFoil R2/2 / Material: GOLD / Support film - Material: GOLD / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Frames/image: 1-50 / Average electron dose: 64.01 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.67 µm / Nominal defocus min: 0.81 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT |
|---|---|
| Output model |  PDB-7tr3: |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)