[English] 日本語
 Yorodumi
Yorodumi- EMDB-23911: Cryo-EM reveals partially and fully assembled native glycine rece... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-23911 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
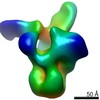
| Title | Cryo-EM reveals partially and fully assembled native glycine receptors,homomeric tetramer | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | glycine receptor / ion channel / homomeric tetramer / MEMBRANE PROTEIN / SIGNALING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationtaurine binding / positive regulation of acrosome reaction / acrosome reaction / negative regulation of transmission of nerve impulse / synaptic transmission, glycinergic / neuromuscular process controlling posture / righting reflex / regulation of respiratory gaseous exchange by nervous system process / extracellularly glycine-gated chloride channel activity / inhibitory synapse ...taurine binding / positive regulation of acrosome reaction / acrosome reaction / negative regulation of transmission of nerve impulse / synaptic transmission, glycinergic / neuromuscular process controlling posture / righting reflex / regulation of respiratory gaseous exchange by nervous system process / extracellularly glycine-gated chloride channel activity / inhibitory synapse / glycinergic synapse / excitatory extracellular ligand-gated monoatomic ion channel activity / adult walking behavior / glycine binding / startle response / inhibitory postsynaptic potential / cellular response to zinc ion / cellular response to ethanol / chloride channel complex / neuronal action potential / visual perception / muscle contraction / ligand-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential / chloride transmembrane transport / cellular response to amino acid stimulus / neuropeptide signaling pathway / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential / transmembrane signaling receptor activity / perikaryon / postsynaptic membrane / external side of plasma membrane / dendrite / zinc ion binding Similarity search - Function | |||||||||
| Biological species |   | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.1 Å | |||||||||
 Authors Authors | Zhu H / Gouaux E | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2021 Journal: Nature / Year: 2021Title: Architecture and assembly mechanism of native glycine receptors. Authors: Hongtao Zhu / Eric Gouaux /  Abstract: Glycine receptors (GlyRs) are pentameric, 'Cys-loop' receptors that form chloride-permeable channels and mediate fast inhibitory signalling throughout the central nervous system. In the spinal cord ...Glycine receptors (GlyRs) are pentameric, 'Cys-loop' receptors that form chloride-permeable channels and mediate fast inhibitory signalling throughout the central nervous system. In the spinal cord and brainstem, GlyRs regulate locomotion and cause movement disorders when mutated. However, the stoichiometry of native GlyRs and the mechanism by which they are assembled remain unclear, despite extensive investigation. Here we report cryo-electron microscopy structures of native GlyRs from pig spinal cord and brainstem, revealing structural insights into heteromeric receptors and their predominant subunit stoichiometry of 4α:1β. Within the heteromeric pentamer, the β(+)-α(-) interface adopts a structure that is distinct from the α(+)-α(-) and α(+)-β(-) interfaces. Furthermore, the β-subunit contains a unique phenylalanine residue that resides within the pore and disrupts the canonical picrotoxin site. These results explain why inclusion of the β-subunit breaks receptor symmetry and alters ion channel pharmacology. We also find incomplete receptor complexes and, by elucidating their structures, reveal the architectures of partially assembled α-trimers and α-tetramers. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_23911.map.gz emd_23911.map.gz | 121.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-23911-v30.xml emd-23911-v30.xml emd-23911.xml emd-23911.xml | 13.9 KB 13.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_23911.png emd_23911.png | 169.4 KB | ||
| Filedesc metadata |  emd-23911.cif.gz emd-23911.cif.gz | 5.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-23911 http://ftp.pdbj.org/pub/emdb/structures/EMD-23911 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23911 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23911 | HTTPS FTP |
-Related structure data
| Related structure data |  7mlvMC  7mluC  7mlyC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_23911.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_23911.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.826 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Native homomeric glycine receptor tetramer bound with 3D1 fab
| Entire | Name: Native homomeric glycine receptor tetramer bound with 3D1 fab |
|---|---|
| Components |
|
-Supramolecule #1: Native homomeric glycine receptor tetramer bound with 3D1 fab
| Supramolecule | Name: Native homomeric glycine receptor tetramer bound with 3D1 fab type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Molecular weight | Theoretical: 400 KDa |
-Supramolecule #2: Native homomeric glycine receptor tetramer
| Supramolecule | Name: Native homomeric glycine receptor tetramer / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #3 |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #3: 3D1 fab
| Supramolecule | Name: 3D1 fab / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: 3D1 Fab Light Chain
| Macromolecule | Name: 3D1 Fab Light Chain / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 11.743124 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: DIVMTQSHKF MSTSVGDRVS ITCKASQDVS TAVAWYQQKP GQSPKLLIYW ASTRHTGVPG RFTGSGSGTD YTLTISSVQA EDLSLYYCQ QHYSTPRTFG GGTKLEIK |
-Macromolecule #2: 3D1 Fab Heavy Chain
| Macromolecule | Name: 3D1 Fab Heavy Chain / type: protein_or_peptide / ID: 2 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 12.983512 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: QVQLQQSGAE LMKPGAAVKI SCKATGHTIS RYWIDWLKQR PGHGLEWIGE ILPGSGSTNY NEKFKGKATF TAEKSSNTAY MQLSSLTSE DSAVYYCAMG VRGNYFDYWG QGTTLTVSS |
-Macromolecule #3: Glycine receptor alpha 1
| Macromolecule | Name: Glycine receptor alpha 1 / type: protein_or_peptide / ID: 3 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 52.64475 KDa |
| Sequence | String: MYRFNTLRLY LWETIVFFSL AASKEAEAAR SASKPMSPSD FLDKLMGRTS GYDARIRPNF KGPPVNVSCN IFINSFGSIA ETTMDYRVN IFLRQQWNDP RLAYNEYPDD SLDLDPSMLD SIWKPDLFFA NEKGAHFHEI TTDNKLLRIS RNGNVLYSIR I TLTLACPM ...String: MYRFNTLRLY LWETIVFFSL AASKEAEAAR SASKPMSPSD FLDKLMGRTS GYDARIRPNF KGPPVNVSCN IFINSFGSIA ETTMDYRVN IFLRQQWNDP RLAYNEYPDD SLDLDPSMLD SIWKPDLFFA NEKGAHFHEI TTDNKLLRIS RNGNVLYSIR I TLTLACPM DLKNFPMDVQ TCIMQLESFG YTMNDLIFEW QEQGAVQVAD GLTLPQFILK EEKDLRYCTK HYNTGKFTCI EA RFHLERQ MGYYLIQMYI PSLLIVILSW ISFWINMDAA PARVGLGITT VLTMTTQSSG SRASLPKVSY VKAIDIWMAV CLL FVFSAL LEYAAVNFVS RQHKELLRFR RKRRHHKSPM LNLFQEDEAG EGRFNFSAYG MGPACLQAKD GISVKGANNT TTNP PPAPS KSPEEMRKLF IQRAKKIDKI SRIGFPMAFL IFNMFYWIIY KIVRREDVHN Q UniProtKB: Glycine receptor alpha 1 |
-Macromolecule #7: alpha-D-mannopyranose
| Macromolecule | Name: alpha-D-mannopyranose / type: ligand / ID: 7 / Number of copies: 4 / Formula: MAN |
|---|---|
| Molecular weight | Theoretical: 180.156 Da |
| Chemical component information |  ChemComp-MAN: |
-Macromolecule #8: beta-D-mannopyranose
| Macromolecule | Name: beta-D-mannopyranose / type: ligand / ID: 8 / Number of copies: 1 / Formula: BMA |
|---|---|
| Molecular weight | Theoretical: 180.156 Da |
| Chemical component information |  ChemComp-BMA: |
-Macromolecule #9: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 9 / Number of copies: 2 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.05 mg/mL |
|---|---|
| Buffer | pH: 8 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 28.2 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 4.1 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC / Number images used: 129772 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION / Software - Name: cryoSPARC |
| Final angle assignment | Type: ANGULAR RECONSTITUTION / Software - Name: cryoSPARC |
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















