+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-23657 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Canine parvovirus and Fab14 at partial occupancy | |||||||||
 Map data Map data | Canine parvovirus and Fab14 at partial occupancy | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | canine parvovirus / CPV / VIRUS | |||||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont entry into host cell via permeabilization of host membrane / T=1 icosahedral viral capsid / clathrin-dependent endocytosis of virus by host cell / virion attachment to host cell / structural molecule activity Similarity search - Function | |||||||||
| Biological species |  Canine parvovirus type 2 / Canine parvovirus type 2 /  Canine parvovirus Canine parvovirus | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.26 Å | |||||||||
 Authors Authors | Goteschius DJ / Hartmann SR | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2021 Journal: Proc Natl Acad Sci U S A / Year: 2021Title: High-resolution asymmetric structure of a Fab-virus complex reveals overlap with the receptor binding site. Authors: Daniel J Goetschius / Samantha R Hartmann / Lindsey J Organtini / Heather Callaway / Kai Huang / Carol M Bator / Robert E Ashley / Alexander M Makhov / James F Conway / Colin R Parrish / Susan L Hafenstein /  Abstract: Canine parvovirus is an important pathogen causing severe diseases in dogs, including acute hemorrhagic enteritis, myocarditis, and cerebellar disease. Overlap on the surface of parvovirus capsids ...Canine parvovirus is an important pathogen causing severe diseases in dogs, including acute hemorrhagic enteritis, myocarditis, and cerebellar disease. Overlap on the surface of parvovirus capsids between the antigenic epitope and the receptor binding site has contributed to cross-species transmission, giving rise to closely related variants. It has been shown that Mab 14 strongly binds and neutralizes canine but not feline parvovirus, suggesting this antigenic site also controls species-specific receptor binding. To visualize the conformational epitope at high resolution, we solved the cryogenic electron microscopy (cryo-EM) structure of the Fab-virus complex. We also created custom software, Icosahedral Subparticle Extraction and Correlated Classification, to solve a Fab-virus complex with only a few Fab bound per capsid and visualize local structures of the Fab-bound and -unbound antigenic sites extracted from the same complex map. Our results identified the antigenic epitope that had significant overlap with the receptor binding site, and the structures revealed that binding of Fab induced conformational changes to the virus. We were also able to assign the order and position of attached Fabs to allow assessment of complementarity between the Fabs bound to different positions. This approach therefore provides a method for using cryo-EM to investigate complementarity of antibody binding. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_23657.map.gz emd_23657.map.gz | 323.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-23657-v30.xml emd-23657-v30.xml emd-23657.xml emd-23657.xml | 13.7 KB 13.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_23657.png emd_23657.png | 248.6 KB | ||
| Filedesc metadata |  emd-23657.cif.gz emd-23657.cif.gz | 5.2 KB | ||
| Others |  emd_23657_half_map_1.map.gz emd_23657_half_map_1.map.gz emd_23657_half_map_2.map.gz emd_23657_half_map_2.map.gz | 297 MB 295.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-23657 http://ftp.pdbj.org/pub/emdb/structures/EMD-23657 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23657 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23657 | HTTPS FTP |
-Related structure data
| Related structure data |  7m3mMC  7m3lC  7m3nC  7m3oC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_23657.map.gz / Format: CCP4 / Size: 371.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_23657.map.gz / Format: CCP4 / Size: 371.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Canine parvovirus and Fab14 at partial occupancy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.1 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
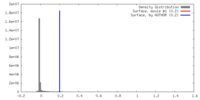
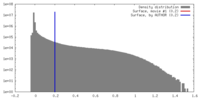
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: Half-map 1
| File | emd_23657_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half-map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
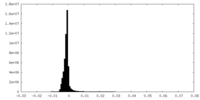
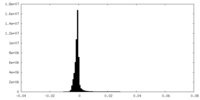
| Density Histograms |
-Half map: Half-map 2
| File | emd_23657_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half-map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
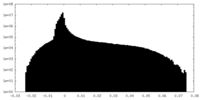
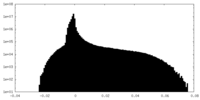
| Density Histograms |
- Sample components
Sample components
-Entire : Canine parvovirus
| Entire | Name:  Canine parvovirus Canine parvovirus |
|---|---|
| Components |
|
-Supramolecule #1: Canine parvovirus
| Supramolecule | Name: Canine parvovirus / type: virus / ID: 1 / Parent: 0 / Macromolecule list: all / NCBI-ID: 10788 / Sci species name: Canine parvovirus / Virus type: VIRION / Virus isolate: SPECIES / Virus enveloped: No / Virus empty: No |
|---|
-Macromolecule #1: Capsid protein 2
| Macromolecule | Name: Capsid protein 2 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Canine parvovirus type 2 Canine parvovirus type 2 |
| Molecular weight | Theoretical: 64.705559 KDa |
| Sequence | String: MSDGAVQPDG GQPAVRNERA TGSGNGSGGG GGGGSGGVGI STGTFNNQTE FKFLENGWVE ITANSSRLVH LNMPESENYR RVVVNNMDK TAVNGNMALD DIHAQIVTPW SLVDANAWGV WFNPGDWQLI VNTMSELHLV SFEQEIFNVV LKTVSESATQ P PTKVYNND ...String: MSDGAVQPDG GQPAVRNERA TGSGNGSGGG GGGGSGGVGI STGTFNNQTE FKFLENGWVE ITANSSRLVH LNMPESENYR RVVVNNMDK TAVNGNMALD DIHAQIVTPW SLVDANAWGV WFNPGDWQLI VNTMSELHLV SFEQEIFNVV LKTVSESATQ P PTKVYNND LTASLMVALD SNNTMPFTPA AMRSETLGFY PWKPTIPTPW RYYFQWDRTL IPSHTGTSGT PTNIYHGTDP DD VQFYTIE NSVPVHLLRT GDEFATGTFF FDCKPCRLTH TWQTNRALGL PPFLNSLPQS EGATNFGDIG VQQDKRRGVT QMG NTNYIT EATIMRPAEV GYSAPYYSFE ASTQGPFKTP IAAGRGGAQT DENQAADGNP RYAFGRQHGQ KTTTTGETPE RFTY IAHQD TGRYPEGDWI QNINFNLPVT NDNVLLPTDP IGGKTGINYT NIFNTYGPLT ALNNVPPVYP NGQIWDKEFD TDLKP RLHV NAPFVCQNNC PGQLFVKVAP NLTNEYDPDA SANMSRIVTY SDFWWKGKLV FKAKLRASHT WNPIQQMSIN VDNQFN YVP SNIGGMKIVY EKSQLAPRKL Y UniProtKB: Capsid protein 2 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.26 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 162627 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: OTHER |
 Movie
Movie Controller
Controller



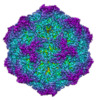















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)