+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-22121 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of V-ATPase from bovine brain, state 1 | |||||||||
 Map data Map data | Structure of V-ATPase from bovine brain, state 1 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | PROTON TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology informationROS and RNS production in phagocytes / Insulin receptor recycling / Transferrin endocytosis and recycling / Amino acids regulate mTORC1 / Ion channel transport / Metabolism of Angiotensinogen to Angiotensins / pH reduction / RHOA GTPase cycle / plasma membrane proton-transporting V-type ATPase complex / synaptic vesicle lumen acidification ...ROS and RNS production in phagocytes / Insulin receptor recycling / Transferrin endocytosis and recycling / Amino acids regulate mTORC1 / Ion channel transport / Metabolism of Angiotensinogen to Angiotensins / pH reduction / RHOA GTPase cycle / plasma membrane proton-transporting V-type ATPase complex / synaptic vesicle lumen acidification / cellular response to increased oxygen levels / vacuolar transport / vacuolar proton-transporting V-type ATPase, V1 domain / vacuolar proton-transporting V-type ATPase, V0 domain / clathrin-coated vesicle membrane / lysosomal lumen acidification / endosomal lumen acidification / proton-transporting V-type ATPase complex / protein localization to cilium / vacuolar proton-transporting V-type ATPase complex / vacuolar acidification / : / cell projection organization / dendritic spine membrane / Neutrophil degranulation / ATPase activator activity / autophagosome membrane / proton-transporting ATPase activity, rotational mechanism / cilium assembly / regulation of macroautophagy / positive regulation of Wnt signaling pathway / H+-transporting two-sector ATPase / transport vesicle / ATP metabolic process / receptor-mediated endocytosis of virus by host cell / RNA endonuclease activity / endoplasmic reticulum-Golgi intermediate compartment membrane / endomembrane system / receptor-mediated endocytosis / proton transmembrane transport / transmembrane transport / endocytosis / melanosome / positive regulation of canonical Wnt signaling pathway / synaptic vesicle membrane / ATPase binding / signaling receptor activity / Hydrolases; Acting on ester bonds / intracellular iron ion homeostasis / early endosome / lysosome / endosome / endosome membrane / apical plasma membrane / nuclear speck / cilium / external side of plasma membrane / axon / lysosomal membrane / hydrolase activity / centrosome / endoplasmic reticulum membrane / perinuclear region of cytoplasm / Golgi apparatus / ATP hydrolysis activity / ATP binding / membrane / plasma membrane / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.37 Å | |||||||||
 Authors Authors | Wang R / Li X | |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2020 Journal: Nat Commun / Year: 2020Title: Cryo-EM structures of intact V-ATPase from bovine brain. Authors: Rong Wang / Tao Long / Abdirahman Hassan / Jin Wang / Yingyuan Sun / Xiao-Song Xie / Xiaochun Li /  Abstract: The vacuolar-type H-ATPases (V-ATPase) hydrolyze ATP to pump protons across the plasma or intracellular membrane, secreting acids to the lumen or acidifying intracellular compartments. It has been ...The vacuolar-type H-ATPases (V-ATPase) hydrolyze ATP to pump protons across the plasma or intracellular membrane, secreting acids to the lumen or acidifying intracellular compartments. It has been implicated in tumor metastasis, renal tubular acidosis, and osteoporosis. Here, we report two cryo-EM structures of the intact V-ATPase from bovine brain with all the subunits including the subunit H, which is essential for ATPase activity. Two type-I transmembrane proteins, Ac45 and (pro)renin receptor, along with subunit c", constitute the core of the c-ring. Three different conformations of A/B heterodimers suggest a mechanism for ATP hydrolysis that triggers a rotation of subunits DF, inducing spinning of subunit d with respect to the entire c-ring. Moreover, many lipid molecules have been observed in the Vo domain to mediate the interactions between subunit c, c", (pro)renin receptor, and Ac45. These two structures reveal unique features of mammalian V-ATPase and suggest a mechanism of V1-Vo torque transmission. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_22121.map.gz emd_22121.map.gz | 469.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-22121-v30.xml emd-22121-v30.xml emd-22121.xml emd-22121.xml | 27.9 KB 27.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_22121.png emd_22121.png | 39.3 KB | ||
| Filedesc metadata |  emd-22121.cif.gz emd-22121.cif.gz | 9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-22121 http://ftp.pdbj.org/pub/emdb/structures/EMD-22121 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22121 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22121 | HTTPS FTP |
-Related structure data
| Related structure data |  6xbwMC  6xbyC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_22121.map.gz / Format: CCP4 / Size: 506 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_22121.map.gz / Format: CCP4 / Size: 506 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Structure of V-ATPase from bovine brain, state 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.833 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
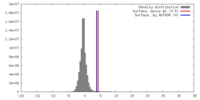
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : Cryo-EM structure of V-ATPase complex from bovine brain, state 1
+Supramolecule #1: Cryo-EM structure of V-ATPase complex from bovine brain, state 1
+Macromolecule #1: V-type proton ATPase catalytic subunit A
+Macromolecule #2: V-type proton ATPase subunit B, brain isoform
+Macromolecule #3: V-type proton ATPase subunit C 1
+Macromolecule #4: V-type proton ATPase subunit D
+Macromolecule #5: V-type proton ATPase subunit E 1
+Macromolecule #6: V-type proton ATPase subunit F
+Macromolecule #7: V-type proton ATPase subunit G
+Macromolecule #8: V-type proton ATPase subunit H
+Macromolecule #9: V-type proton ATPase subunit a
+Macromolecule #10: V-type proton ATPase 21 kDa proteolipid subunit
+Macromolecule #11: V-type proton ATPase subunit d 1
+Macromolecule #12: V-type proton ATPase subunit e 2
+Macromolecule #13: V-type proton ATPase subunit S1
+Macromolecule #14: Renin receptor
+Macromolecule #15: V-type proton ATPase 16 kDa proteolipid subunit
+Macromolecule #16: Ribonuclease kappa
+Macromolecule #17: MAGNESIUM ION
+Macromolecule #18: ADENOSINE-5'-DIPHOSPHATE
+Macromolecule #19: (2S)-3-(hexadecanoyloxy)-2-[(9Z)-octadec-9-enoyloxy]propyl 2-(tri...
+Macromolecule #20: 2-acetamido-2-deoxy-beta-D-glucopyranose
+Macromolecule #21: OLEIC ACID
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: EMDB MAP |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.37 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 84345 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION |
| Final angle assignment | Type: ANGULAR RECONSTITUTION |
 Movie
Movie Controller
Controller















 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)