[English] 日本語
 Yorodumi
Yorodumi- EMDB-22048: C3 symmetric reconstruction of CD4- and 17-bound B41 HIV-1 Env SO... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-22048 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | C3 symmetric reconstruction of CD4- and 17-bound B41 HIV-1 Env SOSIP in complex with small molecule GO52 | ||||||||||||
 Map data Map data | Sharpened map | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | HIV-1 / Env / CD4 / receptor-bound state / small molecule / VIRAL PROTEIN / VIRAL PROTEIN-Immune System complex | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationhelper T cell enhancement of adaptive immune response / interleukin-16 binding / interleukin-16 receptor activity / response to methamphetamine hydrochloride / maintenance of protein location in cell / cellular response to ionomycin / T cell selection / symbiont-mediated perturbation of host defense response / MHC class II protein binding / positive regulation of kinase activity ...helper T cell enhancement of adaptive immune response / interleukin-16 binding / interleukin-16 receptor activity / response to methamphetamine hydrochloride / maintenance of protein location in cell / cellular response to ionomycin / T cell selection / symbiont-mediated perturbation of host defense response / MHC class II protein binding / positive regulation of kinase activity / interleukin-15-mediated signaling pathway / cellular response to granulocyte macrophage colony-stimulating factor stimulus / positive regulation of monocyte differentiation / Nef Mediated CD4 Down-regulation / Alpha-defensins / regulation of T cell activation / response to vitamin D / extracellular matrix structural constituent / Other interleukin signaling / T cell receptor complex / enzyme-linked receptor protein signaling pathway / Translocation of ZAP-70 to Immunological synapse / Phosphorylation of CD3 and TCR zeta chains / positive regulation of protein kinase activity / regulation of calcium ion transport / positive regulation of calcium ion transport into cytosol / macrophage differentiation / Generation of second messenger molecules / immunoglobulin binding / T cell differentiation / Co-inhibition by PD-1 / Binding and entry of HIV virion / coreceptor activity / positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / positive regulation of T cell proliferation / positive regulation of interleukin-2 production / positive regulation of calcium-mediated signaling / cell surface receptor protein tyrosine kinase signaling pathway / protein tyrosine kinase binding / host cell endosome membrane / Vpu mediated degradation of CD4 / clathrin-coated endocytic vesicle membrane / calcium-mediated signaling / positive regulation of protein phosphorylation / MHC class II protein complex binding / transmembrane signaling receptor activity / Downstream TCR signaling / response to estradiol / Cargo recognition for clathrin-mediated endocytosis / signaling receptor activity / Clathrin-mediated endocytosis / virus receptor activity / response to ethanol / defense response to Gram-negative bacterium / clathrin-dependent endocytosis of virus by host cell / adaptive immune response / positive regulation of viral entry into host cell / early endosome / cell surface receptor signaling pathway / positive regulation of ERK1 and ERK2 cascade / positive regulation of canonical NF-kappaB signal transduction / cell adhesion / positive regulation of MAPK cascade / viral protein processing / immune response / membrane raft / endoplasmic reticulum lumen / fusion of virus membrane with host plasma membrane / external side of plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / lipid binding / symbiont entry into host cell / endoplasmic reticulum membrane / protein kinase binding / positive regulation of DNA-templated transcription / virion attachment to host cell / host cell plasma membrane / virion membrane / structural molecule activity / enzyme binding / signal transduction / protein homodimerization activity / zinc ion binding / identical protein binding / membrane / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |   Human immunodeficiency virus 1 / Human immunodeficiency virus 1 /  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
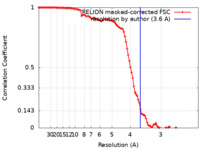
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | ||||||||||||
 Authors Authors | Ozorowski G / Torres JL | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2020 Journal: Cell Rep / Year: 2020Title: A Strain-Specific Inhibitor of Receptor-Bound HIV-1 Targets a Pocket near the Fusion Peptide. Authors: Gabriel Ozorowski / Jonathan L Torres / Diogo Santos-Martins / Stefano Forli / Andrew B Ward /  Abstract: Disruption of viral fusion represents a viable, albeit under-explored, target for HIV therapeutics. Here, while studying the receptor-bound envelope glycoprotein conformation by cryoelectron ...Disruption of viral fusion represents a viable, albeit under-explored, target for HIV therapeutics. Here, while studying the receptor-bound envelope glycoprotein conformation by cryoelectron microscopy (cryo-EM), we identify a pocket near the base of the trimer containing a bound detergent molecule and perform in silico drug screening by using a library of drug-like and commercially available molecules. After down-selection, we solve cryo-EM structures that validate the binding of two small molecule hits in very similar manners to the predicted binding poses, including interactions with aromatic residues within the fusion peptide. One of the molecules demonstrates low micromolar inhibition of the autologous virus by using a very rare phenylalanine in the fusion peptide and stabilizing the surrounding region. This work demonstrates that small molecules can target the fusion process, providing an additional target for anti-HIV therapeutics, and highlights the need to explore how fusion peptide sequence variations affect receptor-mediated conformational states across diverse HIV strains. #1:  Journal: Biorxiv / Year: 2020 Journal: Biorxiv / Year: 2020Title: A strain-specific inhibitor of receptor-bound HIV-1 targets a pocket near the fusion peptide and offers a template for drug design Authors: Ozorowski G / Torres JL / Santos-Martins D / Forli S / Ward AB | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_22048.map.gz emd_22048.map.gz | 85.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-22048-v30.xml emd-22048-v30.xml emd-22048.xml emd-22048.xml | 24.6 KB 24.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_22048_fsc.xml emd_22048_fsc.xml | 10.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_22048.png emd_22048.png | 197.6 KB | ||
| Masks |  emd_22048_msk_1.map emd_22048_msk_1.map | 91.1 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-22048.cif.gz emd-22048.cif.gz | 7.7 KB | ||
| Others |  emd_22048_half_map_1.map.gz emd_22048_half_map_1.map.gz emd_22048_half_map_2.map.gz emd_22048_half_map_2.map.gz | 71.3 MB 71.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-22048 http://ftp.pdbj.org/pub/emdb/structures/EMD-22048 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22048 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22048 | HTTPS FTP |
-Validation report
| Summary document |  emd_22048_validation.pdf.gz emd_22048_validation.pdf.gz | 881.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_22048_full_validation.pdf.gz emd_22048_full_validation.pdf.gz | 881.3 KB | Display | |
| Data in XML |  emd_22048_validation.xml.gz emd_22048_validation.xml.gz | 17.1 KB | Display | |
| Data in CIF |  emd_22048_validation.cif.gz emd_22048_validation.cif.gz | 22.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-22048 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-22048 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-22048 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-22048 | HTTPS FTP |
-Related structure data
| Related structure data |  6x5bMC  6opnC  6opoC  6oppC  6opqC  6x5cC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_22048.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_22048.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.15 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
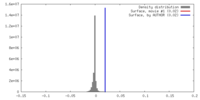
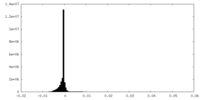
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_22048_msk_1.map emd_22048_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
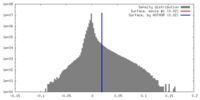
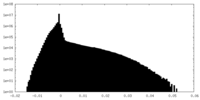
| Density Histograms |
-Half map: Half map 1
| File | emd_22048_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
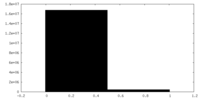
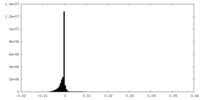
| Density Histograms |
-Half map: Half map 2
| File | emd_22048_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
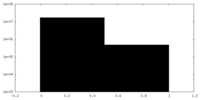
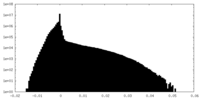
| Density Histograms |
- Sample components
Sample components
-Entire : B41 HIV-1 Env SOSIP in complex with soluble CD4, 17b Fab, and sma...
| Entire | Name: B41 HIV-1 Env SOSIP in complex with soluble CD4, 17b Fab, and small molecule GO52 |
|---|---|
| Components |
|
-Supramolecule #1: B41 HIV-1 Env SOSIP in complex with soluble CD4, 17b Fab, and sma...
| Supramolecule | Name: B41 HIV-1 Env SOSIP in complex with soluble CD4, 17b Fab, and small molecule GO52 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 Human immunodeficiency virus 1 |
| Molecular weight | Theoretical: 630 KDa |
-Macromolecule #1: Envelope glycoprotein gp120
| Macromolecule | Name: Envelope glycoprotein gp120 / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 / Strain: B41 Human immunodeficiency virus 1 / Strain: B41 |
| Molecular weight | Theoretical: 58.872902 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MDAMKRGLCC VLLLCGAVFV SPSQEIHARF RRGARAAKKW VTVYYGVPVW KEATTTLFCA SDAKAYDTEV HNVWATHACV PTDPNPQEI VLGNVTENFN MWKNNMVEQM HEDIISLWDQ SLKPCVKLTP LCVTLNCNNV NTNNTNNSTN ATISDWEKME T GEMKNCSF ...String: MDAMKRGLCC VLLLCGAVFV SPSQEIHARF RRGARAAKKW VTVYYGVPVW KEATTTLFCA SDAKAYDTEV HNVWATHACV PTDPNPQEI VLGNVTENFN MWKNNMVEQM HEDIISLWDQ SLKPCVKLTP LCVTLNCNNV NTNNTNNSTN ATISDWEKME T GEMKNCSF NVTTSIRDKI KKEYALFYKL DVVPLENKNN INNTNITNYR LINCNTSVIT QACPKVSFEP IPIHYCAPAG FA ILKCNSK TFNGSGPCTN VSTVQCTHGI RPVVSTQLLL NGSLAEEEIV IRSENITDNA KTIIVQLNEA VEINCTRPNN NTR KSIHIG PGRAFYATGD IIGNIRQAHC NISKARWNET LGQIVAKLEE QFPNKTIIFN HSSGGDPEIV THSFNCGGEF FYCN TTPLF NSTWNNTRTD DYPTGGEQNI TLQCRIKQII NMWQGVGKAM YAPPIRGQIR CSSNITGLLL TRDGGRDQNG TETFR PGGG NMRDNWRSEL YKYKVVKIEP LGIAPTACKR RVVQRRRRRR UniProtKB: Envelope glycoprotein gp160 |
-Macromolecule #2: Envelope glycoprotein gp41
| Macromolecule | Name: Envelope glycoprotein gp41 / type: protein_or_peptide / ID: 2 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human immunodeficiency virus 1 / Strain: B41 Human immunodeficiency virus 1 / Strain: B41 |
| Molecular weight | Theoretical: 17.357824 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: AVGLGAFILG FLGAAGSTMG AASMALTVQA RLLLSGIVQQ QNNLLRAPEA QQHMLQLTVW GIKQLQARVL AVERYLRDQQ LLGIWGCSG KIICCTNVPW NDSWSNKTIN EIWDNMTWMQ WEKEIDNYTQ HIYTLLEVSQ IQQEKNEQEL LELD UniProtKB: Envelope glycoprotein gp160 |
-Macromolecule #3: 17b Fab light chain
| Macromolecule | Name: 17b Fab light chain / type: protein_or_peptide / ID: 3 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.425869 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: ELELTQSPAT LSVSPGERAT LSCRASESVS SDLAWYQQKP GQAPRLLIYG ASTRATGVPA RFSGSGSGAE FTLTISSLQS EDFAVYYCQ QYNNWPPRYT FGQGTRLEIK RTVAAPSVFI FPPSDEQLKS GTASVVCLLN NFYPREAKVQ WKVDNALQSG N SQESVTEQ ...String: ELELTQSPAT LSVSPGERAT LSCRASESVS SDLAWYQQKP GQAPRLLIYG ASTRATGVPA RFSGSGSGAE FTLTISSLQS EDFAVYYCQ QYNNWPPRYT FGQGTRLEIK RTVAAPSVFI FPPSDEQLKS GTASVVCLLN NFYPREAKVQ WKVDNALQSG N SQESVTEQ DSKDSTYSLS STLTLSKADY EKHKVYACEV THQGLSSPVT KSFNRG |
-Macromolecule #4: T-cell surface glycoprotein CD4
| Macromolecule | Name: T-cell surface glycoprotein CD4 / type: protein_or_peptide / ID: 4 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.039172 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: METDTLLLWV LLLWVPGSTG KKVVLGKKGD TVELTCTASQ KKSIQFHWKN SNQIKILGNQ GSFLTKGPSK LNDRADSRRS LWDQGNFPL IIKNLKIEDS DTYICEVEDQ KEEVQLLVFG LTANSDTHLL QGQSLTLTLE SPPGSSPSVQ CRSPRGKNIQ G GKTLSVSQ ...String: METDTLLLWV LLLWVPGSTG KKVVLGKKGD TVELTCTASQ KKSIQFHWKN SNQIKILGNQ GSFLTKGPSK LNDRADSRRS LWDQGNFPL IIKNLKIEDS DTYICEVEDQ KEEVQLLVFG LTANSDTHLL QGQSLTLTLE SPPGSSPSVQ CRSPRGKNIQ G GKTLSVSQ LELQDSGTWT CTVLQNQKKV EFKIDIVVLA GGSGHHHHHH UniProtKB: T-cell surface glycoprotein CD4 |
-Macromolecule #5: 17b Fab heavy chain
| Macromolecule | Name: 17b Fab heavy chain / type: protein_or_peptide / ID: 5 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 24.484455 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QVQLLESGAE VKKPGSSVKV SCKASGDTFI RYSFTWVRQA PGQGLEWMGR IITILDVAHY APHLQGRVTI TADKSTSTVY LELRNLRSD DTAVYFCAGV YEGEADEGEY DNNGFLKHWG QGTLVTVTSA STKGPSVFPL APSSKSTSGG TAALGCLVKD Y FPEPVTVS ...String: QVQLLESGAE VKKPGSSVKV SCKASGDTFI RYSFTWVRQA PGQGLEWMGR IITILDVAHY APHLQGRVTI TADKSTSTVY LELRNLRSD DTAVYFCAGV YEGEADEGEY DNNGFLKHWG QGTLVTVTSA STKGPSVFPL APSSKSTSGG TAALGCLVKD Y FPEPVTVS WNSGALTSGV HTFPAVLQSS GLYSLSSVVT VPSSSLGTQT YICNVNHKPS NTKVDKKVEP K |
-Macromolecule #8: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 8 / Number of copies: 27 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #9: N-(4'-methyl[1,1'-biphenyl]-4-yl)-1-oxa-7-azaspiro[3.5]nonane-7-c...
| Macromolecule | Name: N-(4'-methyl[1,1'-biphenyl]-4-yl)-1-oxa-7-azaspiro[3.5]nonane-7-carboxamide type: ligand / ID: 9 / Number of copies: 3 / Formula: UOV |
|---|---|
| Molecular weight | Theoretical: 336.427 Da |
| Chemical component information | 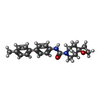 ChemComp-UOV: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.5 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
Details: Detergent diluted into sample shortly before application to grid | ||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 283 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number real images: 2654 / Average exposure time: 11.5 sec. / Average electron dose: 49.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 36000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)