[English] 日本語
 Yorodumi
Yorodumi- EMDB-16138: BAM-EspP complex structure with BamA-S425C/EspP-S1299C mutations ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | BAM-EspP complex structure with BamA-S425C/EspP-S1299C mutations in nanodisc | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | BAM / BamABCDE / EspP / Gram-negative bacteria / outer membrane protein / outer membrane barrel / BamA / BamB / BamC / BamD / BamE / MEMBRANE PROTEIN / Outer membrane protein insertion and release / TRANSPORT PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationBam protein complex / protein insertion into membrane / Gram-negative-bacterium-type cell outer membrane assembly / Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases / cell outer membrane / protein-macromolecule adaptor activity / periplasmic space / serine-type endopeptidase activity / response to antibiotic / cell surface ...Bam protein complex / protein insertion into membrane / Gram-negative-bacterium-type cell outer membrane assembly / Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases / cell outer membrane / protein-macromolecule adaptor activity / periplasmic space / serine-type endopeptidase activity / response to antibiotic / cell surface / proteolysis / extracellular region / identical protein binding / membrane Similarity search - Function | |||||||||
| Biological species |   | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Shen C / Chang S / Luo Q / Zhang Z / Xie T / Luo B / Lu G / Zhu X / Wei X / Dong C ...Shen C / Chang S / Luo Q / Zhang Z / Xie T / Luo B / Lu G / Zhu X / Wei X / Dong C / Zhou R / Zhang X / Tang X / Dong H | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: Structural basis of BAM-mediated outer membrane β-barrel protein assembly. Authors: Chongrong Shen / Shenghai Chang / Qinghua Luo / Kevin Chun Chan / Zhibo Zhang / Bingnan Luo / Teng Xie / Guangwen Lu / Xiaofeng Zhu / Xiawei Wei / Changjiang Dong / Ruhong Zhou / Xing Zhang ...Authors: Chongrong Shen / Shenghai Chang / Qinghua Luo / Kevin Chun Chan / Zhibo Zhang / Bingnan Luo / Teng Xie / Guangwen Lu / Xiaofeng Zhu / Xiawei Wei / Changjiang Dong / Ruhong Zhou / Xing Zhang / Xiaodi Tang / Haohao Dong /   Abstract: The outer membrane structure is common in Gram-negative bacteria, mitochondria and chloroplasts, and contains outer membrane β-barrel proteins (OMPs) that are essential interchange portals of ...The outer membrane structure is common in Gram-negative bacteria, mitochondria and chloroplasts, and contains outer membrane β-barrel proteins (OMPs) that are essential interchange portals of materials. All known OMPs share the antiparallel β-strand topology, implicating a common evolutionary origin and conserved folding mechanism. Models have been proposed for bacterial β-barrel assembly machinery (BAM) to initiate OMP folding; however, mechanisms by which BAM proceeds to complete OMP assembly remain unclear. Here we report intermediate structures of BAM assembling an OMP substrate, EspP, demonstrating sequential conformational dynamics of BAM during the late stages of OMP assembly, which is further supported by molecular dynamics simulations. Mutagenic in vitro and in vivo assembly assays reveal functional residues of BamA and EspP for barrel hybridization, closure and release. Our work provides novel insights into the common mechanism of OMP assembly. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_16138.map.gz emd_16138.map.gz | 32.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-16138-v30.xml emd-16138-v30.xml emd-16138.xml emd-16138.xml | 25.5 KB 25.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_16138.png emd_16138.png | 106.1 KB | ||
| Filedesc metadata |  emd-16138.cif.gz emd-16138.cif.gz | 7.2 KB | ||
| Others |  emd_16138_additional_1.map.gz emd_16138_additional_1.map.gz emd_16138_half_map_1.map.gz emd_16138_half_map_1.map.gz emd_16138_half_map_2.map.gz emd_16138_half_map_2.map.gz | 32.3 MB 59.4 MB 59.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-16138 http://ftp.pdbj.org/pub/emdb/structures/EMD-16138 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16138 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16138 | HTTPS FTP |
-Validation report
| Summary document |  emd_16138_validation.pdf.gz emd_16138_validation.pdf.gz | 849.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_16138_full_validation.pdf.gz emd_16138_full_validation.pdf.gz | 849.3 KB | Display | |
| Data in XML |  emd_16138_validation.xml.gz emd_16138_validation.xml.gz | 12.1 KB | Display | |
| Data in CIF |  emd_16138_validation.cif.gz emd_16138_validation.cif.gz | 14.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16138 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16138 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16138 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16138 | HTTPS FTP |
-Related structure data
| Related structure data |  8bo2MC  7ye4C  7ye6C  8bnzC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_16138.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_16138.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.93 Å | ||||||||||||||||||||||||||||||||||||
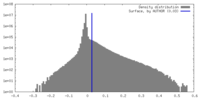
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
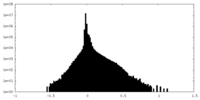
-Additional map: #1
| File | emd_16138_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
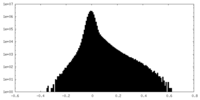
| Density Histograms |
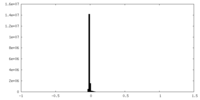
-Half map: #2
| File | emd_16138_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
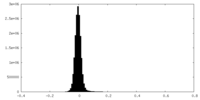
| Density Histograms |
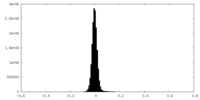
-Half map: #1
| File | emd_16138_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
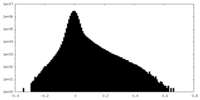
| Density Histograms |
- Sample components
Sample components
-Entire : BAM-EspP complex with BamA-G431C/EspP-N1293C mutations in nanodisc
| Entire | Name: BAM-EspP complex with BamA-G431C/EspP-N1293C mutations in nanodisc |
|---|---|
| Components |
|
-Supramolecule #1: BAM-EspP complex with BamA-G431C/EspP-N1293C mutations in nanodisc
| Supramolecule | Name: BAM-EspP complex with BamA-G431C/EspP-N1293C mutations in nanodisc type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #2: BAM
| Supramolecule | Name: BAM / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #3: EspP
| Supramolecule | Name: EspP / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #6 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Outer membrane protein assembly factor BamA
| Macromolecule | Name: Outer membrane protein assembly factor BamA / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 88.273555 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: FVVKDIHFEG LQRVAVGAAL LSMPVRTGDT VNDEDISNTI RALFATGNFE DVRVLRDGDT LLVQVKERPT IASITFSGNK SVKDDMLKQ NLEASGVRVG ESLDRTTIAD IEKGLEDFYY SVGKYSASVK AVVTPLPRNR VDLKLVFQEG VSAEIQQINI V GNHAFTTD ...String: FVVKDIHFEG LQRVAVGAAL LSMPVRTGDT VNDEDISNTI RALFATGNFE DVRVLRDGDT LLVQVKERPT IASITFSGNK SVKDDMLKQ NLEASGVRVG ESLDRTTIAD IEKGLEDFYY SVGKYSASVK AVVTPLPRNR VDLKLVFQEG VSAEIQQINI V GNHAFTTD ELISHFQLRD EVPWWNVVGD RKYQKQKLAG DLETLRSYYL DRGYARFNID STQVSLTPDK KGIYVTVNIT EG DQYKLSG VEVSGNLAGH SAEIEQLTKI EPGELYNGTK VTKMEDDIKK LLGRYGYAYP RVQSMPEIND ADKTVKLRVN VDA GNRFYV RKIRFEGNDT SKDAVLRREM RQMEGAWLGS DLVDQGKERL NRLGFFETVD TDTQRVPGSP DQVDVVYKVK ERNT GCFNF GIGYGTESGV SFQAGVQQDN WLGTGYAVGI NGTKNDYQTY AELSVTNPYF TVDGVSLGGR LFYNDFQADD ADLSD YTNK SYGTDVTLGF PINEYNSLRA GLGYVHNSLS NMQPQVAMWR YLYSMGEHPS TSDQDNSFKT DDFTFNYGWT YNKLDR GYF PTDGSRVNLT GKVTIPGSDN EYYKVTLDTA TYVPIDDDHK WVVLGRTRWG YGDGLGGKEM PFYENFYAGG SSTVRGF QS NTIGPKAVYF PHQASNYDPD YDYECATQDG AKDLCKSDDA VGGNAMAVAS LEFITPTPFI SDKYANSVRT SFFWDMGT V WDTNWDSSQY SGYPDYSDPS NIRMSAGIAL QWMSPLGPLV FSYAQPFKKY DGDKAEQFQF NIGKTW UniProtKB: Outer membrane protein assembly factor BamA |
-Macromolecule #2: Outer membrane protein assembly factor BamB
| Macromolecule | Name: Outer membrane protein assembly factor BamB / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 41.918945 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MQLRKLLLPG LLSVTLLSGC SLFNSEEDVV KMSPLPTVEN QFTPTTAWST SVGSGIGNFY SNLHPALADN VVYAADRAGL VKALNADDG KEIWSVSLAE KDGWFSKEPA LLSGGVTVSG GHVYIGSEKA QVYALNTSDG TVAWQTKVAG EALSRPVVSD G LVLIHTSN ...String: MQLRKLLLPG LLSVTLLSGC SLFNSEEDVV KMSPLPTVEN QFTPTTAWST SVGSGIGNFY SNLHPALADN VVYAADRAGL VKALNADDG KEIWSVSLAE KDGWFSKEPA LLSGGVTVSG GHVYIGSEKA QVYALNTSDG TVAWQTKVAG EALSRPVVSD G LVLIHTSN GQLQALNEAD GAVKWTVNLD MPSLSLRGES APTTAFGAAV VGGDNGRVSA VLMEQGQMIW QQRISQATGS TE IDRLSDV DTTPVVVNGV VFALAYNGNL TALDLRSGQI MWKRELGSVN DFIVDGNRIY LVDQNDRVMA LTIDGGVTLW TQS DLLHRL LTSPVLYNGN LVVGDSEGYL HWINVEDGRF VAQQKVDSSG FQTEPVAADG KLLIQAKDGT VYSITR UniProtKB: Outer membrane protein assembly factor BamB |
-Macromolecule #3: Outer membrane protein assembly factor BamC
| Macromolecule | Name: Outer membrane protein assembly factor BamC / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 36.875277 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MAYSVQKSRL AKVAGVSLVL LLAACSSDSR YKRQVSGDEA YLEAAPLAEL HAPAGMILPV TSGDYAIPVT NGSGAVGKAL DIRPPAQPL ALVSGARTQF TGDTASLLVE NGRGNTLWPQ VVSVLQAKNY TITQRDDAGQ TLTTDWVQWN RLDEDEQYRG R YQISVKPQ ...String: MAYSVQKSRL AKVAGVSLVL LLAACSSDSR YKRQVSGDEA YLEAAPLAEL HAPAGMILPV TSGDYAIPVT NGSGAVGKAL DIRPPAQPL ALVSGARTQF TGDTASLLVE NGRGNTLWPQ VVSVLQAKNY TITQRDDAGQ TLTTDWVQWN RLDEDEQYRG R YQISVKPQ GYQQAVTVKL LNLEQAGKPV ADAASMQRYS TEMMNVISAG LDKSATDAAN AAQNRASTTM DVQSAADDTG LP MLVVRGP FNVVWQRLPA ALEKVGMKVT DSTRSQGNMA VTYKPLSDSD WQELGASDPG LASGDYKLQV GDLDNRSSLQ FID PKGHTL TQSQNDALVA VFQAAFSK UniProtKB: Outer membrane protein assembly factor BamC |
-Macromolecule #4: Outer membrane protein assembly factor BamD
| Macromolecule | Name: Outer membrane protein assembly factor BamD / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 27.85835 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTRMKYLVAA ATLSLFLAGC SGSKEEVPDN PPNEIYATAQ QKLQDGNWRQ AITQLEALDN RYPFGPYSQQ VQLDLIYAYY KNADLPLAQ AAIDRFIRLN PTHPNIDYVM YMRGLTNMAL DDSALQGFFG VDRSDRDPQH ARAAFSDFSK LVRGYPNSQY T TDATKRLV ...String: MTRMKYLVAA ATLSLFLAGC SGSKEEVPDN PPNEIYATAQ QKLQDGNWRQ AITQLEALDN RYPFGPYSQQ VQLDLIYAYY KNADLPLAQ AAIDRFIRLN PTHPNIDYVM YMRGLTNMAL DDSALQGFFG VDRSDRDPQH ARAAFSDFSK LVRGYPNSQY T TDATKRLV FLKDRLAKYE YSVAEYYTER GAWVAVVNRV EGMLRDYPDT QATRDALPLM ENAYRQMQMN AQAEKVAKII AA NSSNT UniProtKB: Outer membrane protein assembly factor BamD |
-Macromolecule #5: Outer membrane protein assembly factor BamE
| Macromolecule | Name: Outer membrane protein assembly factor BamE / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 13.530256 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MRCKTLTAAA AVLLMLTAGC STLERVVYRP DINQGNYLTA NDVSKIRVGM TQQQVAYALG TPLMSDPFGT NTWFYVFRQQ PGHEGVTQQ TLTLTFNSSG VLTNIDNKPA LSGNGGHHHH HHHH UniProtKB: Outer membrane protein assembly factor BamE |
-Macromolecule #6: Serine protease EspP
| Macromolecule | Name: Serine protease EspP / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO EC number: Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 38.754836 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: NIELVSAPKD TNENVFKASK QTIGFSDVTP VITTRETDDK ITWSLTGYNT VANKEATRNA AALFSVDYKA FLNEVNNLNK RMGDLRDIN GEAGAWARIM SGTGSASGGF SDNYTHVQVG VDKKHELDGL DLFTGFTVTH TDSSASADVF SGKTKSVGAG L YASAMFDS ...String: NIELVSAPKD TNENVFKASK QTIGFSDVTP VITTRETDDK ITWSLTGYNT VANKEATRNA AALFSVDYKA FLNEVNNLNK RMGDLRDIN GEAGAWARIM SGTGSASGGF SDNYTHVQVG VDKKHELDGL DLFTGFTVTH TDSSASADVF SGKTKSVGAG L YASAMFDS GAYIDLIGKY VHHDNEYTAT FAGLGTRDYS THSWYAGAEA GYRYHVTEDA WIEPQAELVY GSVSGKQFAW KD QGMHLSM KDKDYNPLIG RTGVDVGKSF SGKDWKVTAR AGLGYQFDLL ANGETVLRDA SGEKRIKGEK DSRMLMSVGL NAE IRDNVR FGLEFEKSAF GKYNVDNAVN ANFRYCF UniProtKB: Serine protease EspP |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.0 µm / Calibrated defocus min: 1.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 3.1 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 203494 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)