[English] 日本語
 Yorodumi
Yorodumi- EMDB-13952: Structure of the collided E. coli disome - VemP-stalled 70S ribosome -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-13952 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the collided E. coli disome - VemP-stalled 70S ribosome | |||||||||
 Map data Map data | disome map after focused refinement on stalled ribosome | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Ribosome rescue / disome / ribosome collision / stalling / no-go complex / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of cytoplasmic translational initiation / stringent response / transcription antitermination factor activity, RNA binding / ornithine decarboxylase inhibitor activity / misfolded RNA binding / Group I intron splicing / RNA folding / transcriptional attenuation / endoribonuclease inhibitor activity / positive regulation of ribosome biogenesis ...negative regulation of cytoplasmic translational initiation / stringent response / transcription antitermination factor activity, RNA binding / ornithine decarboxylase inhibitor activity / misfolded RNA binding / Group I intron splicing / RNA folding / transcriptional attenuation / endoribonuclease inhibitor activity / positive regulation of ribosome biogenesis / RNA-binding transcription regulator activity / translational termination / four-way junction DNA binding / negative regulation of cytoplasmic translation / DnaA-L2 complex / regulation of mRNA stability / translation repressor activity / negative regulation of translational initiation / negative regulation of DNA-templated DNA replication initiation / mRNA regulatory element binding translation repressor activity / positive regulation of RNA splicing / regulation of DNA-templated transcription elongation / transcription elongation factor complex / cytosolic ribosome assembly / response to reactive oxygen species / ribosome assembly / assembly of large subunit precursor of preribosome / transcription antitermination / DNA endonuclease activity / translational initiation / regulation of cell growth / DNA-templated transcription termination / response to radiation / maintenance of translational fidelity / mRNA 5'-UTR binding / regulation of translation / large ribosomal subunit / ribosomal small subunit assembly / transferase activity / ribosome biogenesis / ribosome binding / ribosomal small subunit biogenesis / 5S rRNA binding / small ribosomal subunit / ribosomal large subunit assembly / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / negative regulation of translation / rRNA binding / structural constituent of ribosome / ribosome / translation / response to antibiotic / negative regulation of DNA-templated transcription / hydrolase activity / mRNA binding / DNA binding / RNA binding / zinc ion binding / membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |   Vibrio alginolyticus (bacteria) Vibrio alginolyticus (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.97 Å | |||||||||
 Authors Authors | Kratzat H / Buschauer R | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2022 Journal: Nature / Year: 2022Title: Ribosome collisions induce mRNA cleavage and ribosome rescue in bacteria. Authors: Kazuki Saito / Hanna Kratzat / Annabelle Campbell / Robert Buschauer / A Maxwell Burroughs / Otto Berninghausen / L Aravind / Rachel Green / Roland Beckmann / Allen R Buskirk /   Abstract: Ribosome rescue pathways recycle stalled ribosomes and target problematic mRNAs and aborted proteins for degradation. In bacteria, it remains unclear how rescue pathways distinguish ribosomes stalled ...Ribosome rescue pathways recycle stalled ribosomes and target problematic mRNAs and aborted proteins for degradation. In bacteria, it remains unclear how rescue pathways distinguish ribosomes stalled in the middle of a transcript from actively translating ribosomes. Here, using a genetic screen in Escherichia coli, we discovered a new rescue factor that has endonuclease activity. SmrB cleaves mRNAs upstream of stalled ribosomes, allowing the ribosome rescue factor tmRNA (which acts on truncated mRNAs) to rescue upstream ribosomes. SmrB is recruited to ribosomes and is activated by collisions. Cryo-electron microscopy structures of collided disomes from E. coli and Bacillus subtilis show distinct and conserved arrangements of individual ribosomes and the composite SmrB-binding site. These findings reveal the underlying mechanisms by which ribosome collisions trigger ribosome rescue in bacteria. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13952.map.gz emd_13952.map.gz | 406.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13952-v30.xml emd-13952-v30.xml emd-13952.xml emd-13952.xml | 74 KB 74 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_13952.png emd_13952.png | 59 KB | ||
| Filedesc metadata |  emd-13952.cif.gz emd-13952.cif.gz | 13.8 KB | ||
| Others |  emd_13952_additional_1.map.gz emd_13952_additional_1.map.gz emd_13952_additional_2.map.gz emd_13952_additional_2.map.gz | 21.2 MB 404.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13952 http://ftp.pdbj.org/pub/emdb/structures/EMD-13952 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13952 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13952 | HTTPS FTP |
-Related structure data
| Related structure data |  7qg8MC  7qghC  7qgnC  7qgrC  7qguC  7qh4C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_13952.map.gz / Format: CCP4 / Size: 824 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13952.map.gz / Format: CCP4 / Size: 824 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | disome map after focused refinement on stalled ribosome | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.09 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
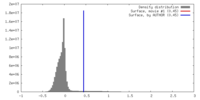
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: local resolution filtered map
| File | emd_13952_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | local resolution filtered map | ||||||||||||
| Projections & Slices |
| ||||||||||||
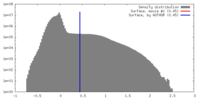
| Density Histograms |
-Additional map: disome map
| File | emd_13952_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | disome map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : stalled/leading ribosome of the collided disome structure
+Supramolecule #1: stalled/leading ribosome of the collided disome structure
+Macromolecule #1: A-site tRNA
+Macromolecule #3: P-site tRNA
+Macromolecule #4: 5S rRNA
+Macromolecule #34: 23S rRNA
+Macromolecule #37: 16S rRNA
+Macromolecule #2: VemP nascent chain
+Macromolecule #5: 50S ribosomal protein L2
+Macromolecule #6: 50S ribosomal protein L3
+Macromolecule #7: 50S ribosomal protein L4
+Macromolecule #8: 50S ribosomal protein L5
+Macromolecule #9: 50S ribosomal protein L6
+Macromolecule #10: 50S ribosomal protein L9
+Macromolecule #11: 50S ribosomal protein L11
+Macromolecule #12: 50S ribosomal protein L13
+Macromolecule #13: 50S ribosomal protein L14
+Macromolecule #14: 50S ribosomal protein L15
+Macromolecule #15: 50S ribosomal protein L16
+Macromolecule #16: 50S ribosomal protein L17
+Macromolecule #17: 50S ribosomal protein L18
+Macromolecule #18: 50S ribosomal protein L19
+Macromolecule #19: 50S ribosomal protein L20
+Macromolecule #20: 50S ribosomal protein L21
+Macromolecule #21: 50S ribosomal protein L22
+Macromolecule #22: 50S ribosomal protein L23
+Macromolecule #23: 50S ribosomal protein L24
+Macromolecule #24: 50S ribosomal protein L25
+Macromolecule #25: 50S ribosomal protein L27
+Macromolecule #26: 50S ribosomal protein L28
+Macromolecule #27: 50S ribosomal protein L29
+Macromolecule #28: 50S ribosomal protein L30
+Macromolecule #29: 50S ribosomal protein L32
+Macromolecule #30: 50S ribosomal protein L33
+Macromolecule #31: 50S ribosomal protein L34
+Macromolecule #32: 50S ribosomal protein L35
+Macromolecule #33: 50S ribosomal protein L36
+Macromolecule #35: 50S ribosomal protein L31
+Macromolecule #36: 50S ribosomal protein L1
+Macromolecule #38: 30S ribosomal protein S2
+Macromolecule #39: 30S ribosomal protein S3
+Macromolecule #40: 30S ribosomal protein S4
+Macromolecule #41: 30S ribosomal protein S5
+Macromolecule #42: 30S ribosomal protein S6, non-modified isoform
+Macromolecule #43: 30S ribosomal protein S7
+Macromolecule #44: 30S ribosomal protein S8
+Macromolecule #45: 30S ribosomal protein S9
+Macromolecule #46: 30S ribosomal protein S10
+Macromolecule #47: 30S ribosomal protein S11
+Macromolecule #48: 30S ribosomal protein S12
+Macromolecule #49: 30S ribosomal protein S13
+Macromolecule #50: 30S ribosomal protein S14
+Macromolecule #51: 30S ribosomal protein S15
+Macromolecule #52: 30S ribosomal protein S16
+Macromolecule #53: 30S ribosomal protein S17
+Macromolecule #54: 30S ribosomal protein S18
+Macromolecule #55: 30S ribosomal protein S19
+Macromolecule #56: 30S ribosomal protein S20
+Macromolecule #57: 30S ribosomal protein S21
+Macromolecule #58: MAGNESIUM ION
+Macromolecule #59: POTASSIUM ION
+Macromolecule #60: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 4.0 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.97 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 75081 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)