+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | PhiCPV4 bacteriophage Portal Protein | |||||||||||||||
 Map data Map data | ||||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | Bacteriophage / portal protein / cryoEM / VIRAL PROTEIN | |||||||||||||||
| Function / homology | Portal protein Gp10 / Portal protein Gp10 superfamily / Phage Connector (GP10) / Connector protein Function and homology information Function and homology information | |||||||||||||||
| Biological species |   Clostridium phage phiCPV4 (virus) Clostridium phage phiCPV4 (virus) | |||||||||||||||
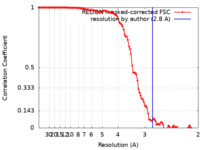
| Method | single particle reconstruction / cryo EM / Resolution: 2.8 Å | |||||||||||||||
 Authors Authors | Javed A / Villanueva H | |||||||||||||||
| Funding support |  United Kingdom, 4 items United Kingdom, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Viruses / Year: 2021 Journal: Viruses / Year: 2021Title: Cryo-EM Structures of Two Bacteriophage Portal Proteins Provide Insights for Antimicrobial Phage Engineering. Authors: Abid Javed / Hugo Villanueva / Shadikejiang Shataer / Sara Vasciaveo / Renos Savva / Elena V Orlova /  Abstract: Widespread antibiotic resistance has returned attention to bacteriophages as a means of managing bacterial pathogenesis. Synthetic biology approaches to engineer phages have demonstrated genomic ...Widespread antibiotic resistance has returned attention to bacteriophages as a means of managing bacterial pathogenesis. Synthetic biology approaches to engineer phages have demonstrated genomic editing to broaden natural host ranges, or to optimise microbicidal action. Gram positive pathogens cause serious pastoral animal and human infections that are especially lethal in newborns. Such pathogens are targeted by the obligate lytic phages of the and families. These phages have relatively small ~20 kb linear protein-capped genomes and their compact organisation, relatively few structural elements, and broad host range, are appealing from a phage-engineering standpoint. In this study, we focus on portal proteins, which are core elements for the assembly of such tailed phages. The structures of dodecameric portal complexes from phage GA1, which targets , and phage phiCPV4 that infects , were determined at resolutions of 3.3 Å and 2.9 Å, respectively. Both are found to closely resemble the related phi29 portal protein fold. However, the portal protein of phiCPV4 exhibits interesting differences in the clip domain. These structures provide new insights on structural diversity in portal proteins and will be essential for considerations in phage structural engineering. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13665.map.gz emd_13665.map.gz | 10.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13665-v30.xml emd-13665-v30.xml emd-13665.xml emd-13665.xml | 13.8 KB 13.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_13665_fsc.xml emd_13665_fsc.xml | 10.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_13665.png emd_13665.png | 98.2 KB | ||
| Filedesc metadata |  emd-13665.cif.gz emd-13665.cif.gz | 5.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13665 http://ftp.pdbj.org/pub/emdb/structures/EMD-13665 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13665 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13665 | HTTPS FTP |
-Related structure data
| Related structure data |  7pv4MC  7pv2C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_13665.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13665.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : PhiCPV4 phage portal protein
| Entire | Name: PhiCPV4 phage portal protein |
|---|---|
| Components |
|
-Supramolecule #1: PhiCPV4 phage portal protein
| Supramolecule | Name: PhiCPV4 phage portal protein / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all / Details: Dodecamer complex made up of 12 subunits. |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Connector protein
| Macromolecule | Name: Connector protein / type: protein_or_peptide / ID: 1 / Number of copies: 12 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Clostridium phage phiCPV4 (virus) Clostridium phage phiCPV4 (virus) |
| Molecular weight | Theoretical: 34.652238 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSKRKSYWNQ GDKILLKNDV FMVDRYYDYY SNMGLNRFRW KNLPPGMESR HIEQALFNEG QAVFFKNTDP NEPYGFLCLP CAPSNGQNI YGDPVDFNGI GVNKYFTNLS PLNAVRILDN DNGLAPVRHI AYYTYLMSQI EMTINMNLDQ QKFPIIIGAT Q KNKLSMEN ...String: MSKRKSYWNQ GDKILLKNDV FMVDRYYDYY SNMGLNRFRW KNLPPGMESR HIEQALFNEG QAVFFKNTDP NEPYGFLCLP CAPSNGQNI YGDPVDFNGI GVNKYFTNLS PLNAVRILDN DNGLAPVRHI AYYTYLMSQI EMTINMNLDQ QKFPIIIGAT Q KNKLSMEN LYEKYSSFEP NILVDEKLAQ ALQEGKGFDA LNTQAPYLLD KLADFKKTCE NELLTFLGIN NTNNDKRERL LT DEVNANN SQITFVLEMA YKNRLDACKR INEMFGLNLE VEKVVNLLEV DMKGDVKNEG INGE UniProtKB: Connector protein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.05 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: PELCO Ultrathin Carbon with Lacey Carbon / Material: COPPER / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 281 K / Instrument: FEI VITROBOT MARK IV Details: 3 ul of sample was applied to lacey carbon grids.. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Temperature | Min: 95.0 K / Max: 98.0 K |
| Specialist optics | Energy filter - Name: GIF Quantum LS / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 3838 pixel / Digitization - Dimensions - Height: 3710 pixel / Digitization - Frames/image: 1-12 / Number grids imaged: 1 / Number real images: 2903 / Average electron dose: 1.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.2 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL / Overall B value: 74.6 |
|---|---|
| Output model |  PDB-7pv4: |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)