+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-11791 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | MutS in Scanning state | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | DNA Mismatch Repair MutS / DNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationmismatched DNA binding / ATP-dependent DNA damage sensor activity / mismatch repair / damaged DNA binding / ATP binding / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.4 Å | |||||||||
 Authors Authors | Fernandez-Leiro R / Bhairosing-Kok D | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2021 Journal: Nat Struct Mol Biol / Year: 2021Title: The selection process of licensing a DNA mismatch for repair. Authors: Rafael Fernandez-Leiro / Doreth Bhairosing-Kok / Vladislav Kunetsky / Charlie Laffeber / Herrie H Winterwerp / Flora Groothuizen / Alexander Fish / Joyce H G Lebbink / Peter Friedhoff / ...Authors: Rafael Fernandez-Leiro / Doreth Bhairosing-Kok / Vladislav Kunetsky / Charlie Laffeber / Herrie H Winterwerp / Flora Groothuizen / Alexander Fish / Joyce H G Lebbink / Peter Friedhoff / Titia K Sixma / Meindert H Lamers /     Abstract: DNA mismatch repair detects and removes mismatches from DNA by a conserved mechanism, reducing the error rate of DNA replication by 100- to 1,000-fold. In this process, MutS homologs scan DNA, ...DNA mismatch repair detects and removes mismatches from DNA by a conserved mechanism, reducing the error rate of DNA replication by 100- to 1,000-fold. In this process, MutS homologs scan DNA, recognize mismatches and initiate repair. How the MutS homologs selectively license repair of a mismatch among millions of matched base pairs is not understood. Here we present four cryo-EM structures of Escherichia coli MutS that provide snapshots, from scanning homoduplex DNA to mismatch binding and MutL activation via an intermediate state. During scanning, the homoduplex DNA forms a steric block that prevents MutS from transitioning into the MutL-bound clamp state, which can only be overcome through kinking of the DNA at a mismatch. Structural asymmetry in all four structures indicates a division of labor between the two MutS monomers. Together, these structures reveal how a small conformational change from the homoduplex- to heteroduplex-bound MutS acts as a licensing step that triggers a dramatic conformational change that enables MutL binding and initiation of the repair cascade. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_11791.map.gz emd_11791.map.gz | 23.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-11791-v30.xml emd-11791-v30.xml emd-11791.xml emd-11791.xml | 22.9 KB 22.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_11791_fsc.xml emd_11791_fsc.xml | 8.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_11791.png emd_11791.png | 154.8 KB | ||
| Masks |  emd_11791_msk_1.map emd_11791_msk_1.map | 52.7 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-11791.cif.gz emd-11791.cif.gz | 7.6 KB | ||
| Others |  emd_11791_half_map_1.map.gz emd_11791_half_map_1.map.gz emd_11791_half_map_2.map.gz emd_11791_half_map_2.map.gz | 40.8 MB 40.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-11791 http://ftp.pdbj.org/pub/emdb/structures/EMD-11791 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11791 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11791 | HTTPS FTP |
-Related structure data
| Related structure data |  7ai5MC  7ai6C  7ai7C  7aibC  7aicC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_11791.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_11791.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.17 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_11791_msk_1.map emd_11791_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
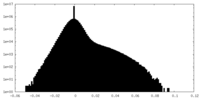
| Density Histograms |
-Half map: #2
| File | emd_11791_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
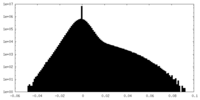
| Density Histograms |
-Half map: #1
| File | emd_11791_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : MutS loaded on matched DNA in the presence of ATP
| Entire | Name: MutS loaded on matched DNA in the presence of ATP |
|---|---|
| Components |
|
-Supramolecule #1: MutS loaded on matched DNA in the presence of ATP
| Supramolecule | Name: MutS loaded on matched DNA in the presence of ATP / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Molecular weight | Theoretical: 190 KDa |
-Supramolecule #2: DNA mismatch repair protein MutS
| Supramolecule | Name: DNA mismatch repair protein MutS / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
-Supramolecule #3: DNA
| Supramolecule | Name: DNA / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2-#3 |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
-Macromolecule #1: DNA mismatch repair protein MutS
| Macromolecule | Name: DNA mismatch repair protein MutS / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 95.397898 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSAIENFDAH TPMMQQYLRL KAQHPEILLF YRMGDFYELF YDDAKRASQL LDISLTKRGA SAGEPIPMAG IPYHAVENYL AKLVNQGES VAICEQIGDP ATSKGPVERK VVRIVTPGTI SDEALLQERQ DNLLAAIWQD SKGFGYATLD ISSGRFRLSE P ADRETMAA ...String: MSAIENFDAH TPMMQQYLRL KAQHPEILLF YRMGDFYELF YDDAKRASQL LDISLTKRGA SAGEPIPMAG IPYHAVENYL AKLVNQGES VAICEQIGDP ATSKGPVERK VVRIVTPGTI SDEALLQERQ DNLLAAIWQD SKGFGYATLD ISSGRFRLSE P ADRETMAA ELQRTNPAEL LYAEDFAEMS LIEGRRGLRR RPLWEFEIDT ARQQLNLQFG TRDLVGFGVE NAPRGLCAAG CL LQYAKDT QRTTLPHIRS ITMEREQDSI IMDAATRRNL EITQNLAGGA ENTLASVLDC TVTPMGSRML KRWLHMPVRD TRV LLERQQ TIGALQDFTA GLQPVLRQVG DLERILARLA LRTARPRDLA RMRHAFQQLP ELRAQLETVD SAPVQALREK MGEF AELRD LLERAIIDTP PVLVRDGGVI ASGYNEELDE WRALADGATD YLERLEVRER ERTGLDTLKV GFNAVHGYYI QISRG QSHL APINYMRRQT LKNAERYIIP ELKEYEDKVL TSKGKALALE KQLYEELFDL LLPHLEALQQ SASALAELDV LVNLAE RAY TLNYTCPTFI DKPGIRITEG RHPVVEQVLN EPFIANPLNL SPQRRMLIIT GPNMGGKSTY MRQTALIALM AYIGSYV PA QKVEIGPIDR IFTRVGAADD LASGRSTFMV EMTETANILH NATEYSLVLM DEIGRGTSTY DGLSLAWACA ENLANKIK A LTLFATHYFE LTQLPEKMEG VANVHLDALE HGDTIAFMHS VQDGAASKSY GLAVAALAGV PKEVIKRARQ KLRELESIS PNAAATQVDG TQMSLLSVPE ETSPAVEALE NLDPRSLTPR QALEWIYRLK SLV UniProtKB: DNA mismatch repair protein MutS |
-Macromolecule #2: DNA (5'-D(P*CP*TP*AP*TP*AP*GP*GP*GP*CP*GP*AP*AP*TP*TP*GP*GP*GP*TP...
| Macromolecule | Name: DNA (5'-D(P*CP*TP*AP*TP*AP*GP*GP*GP*CP*GP*AP*AP*TP*TP*GP*GP*GP*TP*AP*CP*CP*G)-3') type: dna / ID: 2 Details: Plasmid DNA molecule (pRC1765), sequence in structure unidentified Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 6.832414 KDa |
| Sequence | String: (DC)(DT)(DA)(DT)(DA)(DG)(DG)(DG)(DC)(DG) (DA)(DA)(DT)(DT)(DG)(DG)(DG)(DT)(DA)(DC) (DC)(DG) |
-Macromolecule #3: DNA (5'-D(P*CP*GP*GP*TP*AP*CP*CP*CP*AP*AP*TP*TP*CP*GP*CP*CP*CP*TP...
| Macromolecule | Name: DNA (5'-D(P*CP*GP*GP*TP*AP*CP*CP*CP*AP*AP*TP*TP*CP*GP*CP*CP*CP*TP*AP*TP*AP*G)-3') type: dna / ID: 3 Details: Plasmid DNA molecule (pRC1765), sequence in structure unidentified Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 6.672318 KDa |
| Sequence | String: (DC)(DG)(DG)(DT)(DA)(DC)(DC)(DC)(DA)(DA) (DT)(DT)(DC)(DG)(DC)(DC)(DC)(DT)(DA)(DT) (DA)(DG) |
-Macromolecule #4: ADENOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 4 / Number of copies: 2 / Formula: ATP |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| ||||||||||||
| Grid | Model: Quantifoil / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. / Pretreatment - Atmosphere: AIR | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV / Details: blot for 3 seconds before plunging. | ||||||||||||
| Details | Protein sample was purified over a gel filtration column and mixed with DNA+ATP prior to grid preparation |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum LS / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 3838 pixel / Digitization - Dimensions - Height: 3710 pixel / Digitization - Frames/image: 1-40 / Number grids imaged: 1 / Number real images: 2351 / Average exposure time: 12.0 sec. / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus max: 4.0 µm / Calibrated defocus min: 0.5 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal magnification: 105000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)