[English] 日本語
 Yorodumi
Yorodumi- EMDB-11158: Cryo-EM structure of the nitrilase from Pseudomonas fluorescens E... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-11158 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the nitrilase from Pseudomonas fluorescens EBC191 at 3.3 Angstroms | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | bacterial nitrilase / arylacetonitrilase / hydrolase | |||||||||
| Function / homology |  Function and homology information Function and homology informationnitrilase activity / detoxification of nitrogen compound / nitrile hydratase activity Similarity search - Function | |||||||||
| Biological species |  Pseudomonas fluorescens (bacteria) Pseudomonas fluorescens (bacteria) | |||||||||
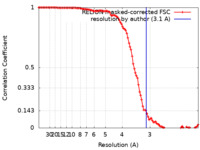
| Method | helical reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Eppinger E / Stolz A | |||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Cryo-EM structure of the nitrilase from Pseudomonas fluorescens EBC191 at 3.3 Angstroms Authors: Eppinger E / Stolz A / Sewell BT | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_11158.map.gz emd_11158.map.gz | 9.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-11158-v30.xml emd-11158-v30.xml emd-11158.xml emd-11158.xml | 10.8 KB 10.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_11158_fsc.xml emd_11158_fsc.xml | 10.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_11158.png emd_11158.png | 107.3 KB | ||
| Filedesc metadata |  emd-11158.cif.gz emd-11158.cif.gz | 5.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-11158 http://ftp.pdbj.org/pub/emdb/structures/EMD-11158 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11158 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11158 | HTTPS FTP |
-Related structure data
| Related structure data |  6zbyMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_11158.map.gz / Format: CCP4 / Size: 9.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_11158.map.gz / Format: CCP4 / Size: 9.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.048 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Active helical nitrilase homooligomer
| Entire | Name: Active helical nitrilase homooligomer |
|---|---|
| Components |
|
-Supramolecule #1: Active helical nitrilase homooligomer
| Supramolecule | Name: Active helical nitrilase homooligomer / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Pseudomonas fluorescens (bacteria) / Strain: EBC191 Pseudomonas fluorescens (bacteria) / Strain: EBC191 |
-Macromolecule #1: NitA
| Macromolecule | Name: NitA / type: protein_or_peptide / ID: 1 / Number of copies: 12 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Pseudomonas fluorescens (bacteria) Pseudomonas fluorescens (bacteria) |
| Molecular weight | Theoretical: 37.740746 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTVHKKQYKV AAVQAAPAFL DLEAGVAKAI GLIAQAAAEG ASLVAFPEAW LPGYPWWIWL DSPAGGMRFV QRNFDNALEV GSEPFERLC RAAAQHKIYV VLGFTERSGG TLYLAQAIID DCGRVVATRR KLKPTHVERS VYGEGDGSDL AVHDTTLGRL G ALCCAEHI ...String: MTVHKKQYKV AAVQAAPAFL DLEAGVAKAI GLIAQAAAEG ASLVAFPEAW LPGYPWWIWL DSPAGGMRFV QRNFDNALEV GSEPFERLC RAAAQHKIYV VLGFTERSGG TLYLAQAIID DCGRVVATRR KLKPTHVERS VYGEGDGSDL AVHDTTLGRL G ALCCAEHI QPLSKYAMYA QHEQVHIAAW PSFSVYRGAA FQLSAQANNA ASQVYALEGQ CFVLAPCATV SKEMLDELID SP AKAELLL EGGGFAMIYG PDGAPLCTPL AETEEGILYA DIDLGVIGVA KAAYDPVGHY SRPDVLRLLV NREPMTRVHY VQP QSLPET SVLAFGAGAD AIRSEENPEE QGDK UniProtKB: NitA |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 0.15 mg/mL |
|---|---|
| Buffer | pH: 7.8 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK I Details: The sample (2.5 ul) was applied to the grid and incubated for 30 seconds at 100% humidity before blotting and plunging.. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Number real images: 2929 / Average exposure time: 6.0 sec. / Average electron dose: 43.1 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL / Target criteria: Cross-correlation coefficient |
|---|---|
| Output model |  PDB-6zby: |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)