+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  |
|---|---|
 試料 試料 | Rab family protein CtRoco GDP
|
| 機能・相同性 |  機能・相同性情報 機能・相同性情報non-specific serine/threonine protein kinase / protein serine/threonine kinase activity / GTP binding / ATP binding / identical protein binding 類似検索 - 分子機能 |
| 生物種 |  Chlorobium tepidum (strain ATCC 49652 / DSM 12025 / NBRC 103806 / TLS) (バクテリア) Chlorobium tepidum (strain ATCC 49652 / DSM 12025 / NBRC 103806 / TLS) (バクテリア) |
 引用 引用 |  ジャーナル: Nat Commun / 年: 2017 ジャーナル: Nat Commun / 年: 2017タイトル: A homologue of the Parkinson's disease-associated protein LRRK2 undergoes a monomer-dimer transition during GTP turnover. 著者: Egon Deyaert / Lina Wauters / Giambattista Guaitoli / Albert Konijnenberg / Margaux Leemans / Susanne Terheyden / Arsen Petrovic / Rodrigo Gallardo / Laura M Nederveen-Schippers / Panagiotis ...著者: Egon Deyaert / Lina Wauters / Giambattista Guaitoli / Albert Konijnenberg / Margaux Leemans / Susanne Terheyden / Arsen Petrovic / Rodrigo Gallardo / Laura M Nederveen-Schippers / Panagiotis S Athanasopoulos / Henderikus Pots / Peter J M Van Haastert / Frank Sobott / Christian Johannes Gloeckner / Rouslan Efremov / Arjan Kortholt / Wim Versées /     要旨: Mutations in LRRK2 are a common cause of genetic Parkinson's disease (PD). LRRK2 is a multi-domain Roco protein, harbouring kinase and GTPase activity. In analogy with a bacterial homologue, LRRK2 ...Mutations in LRRK2 are a common cause of genetic Parkinson's disease (PD). LRRK2 is a multi-domain Roco protein, harbouring kinase and GTPase activity. In analogy with a bacterial homologue, LRRK2 was proposed to act as a GTPase activated by dimerization (GAD), while recent reports suggest LRRK2 to exist under a monomeric and dimeric form in vivo. It is however unknown how LRRK2 oligomerization is regulated. Here, we show that oligomerization of a homologous bacterial Roco protein depends on the nucleotide load. The protein is mainly dimeric in the nucleotide-free and GDP-bound states, while it forms monomers upon GTP binding, leading to a monomer-dimer cycle during GTP hydrolysis. An analogue of a PD-associated mutation stabilizes the dimer and decreases the GTPase activity. This work thus provides insights into the conformational cycle of Roco proteins and suggests a link between oligomerization and disease-associated mutations in LRRK2. |
 登録者 登録者 |
|
- 構造の表示
構造の表示
- ダウンロードとリンク
ダウンロードとリンク
-Data source
| SASBDBのページ |  SASDCA2 SASDCA2 |
|---|
-関連構造データ
| 関連構造データ | C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- 外部リンク
外部リンク
| 「今月の分子」の関連する項目 |
|---|
-モデル
- 試料
試料
 試料 試料 | 名称: Rab family protein CtRoco GDP / 試料濃度: 1.00-2.00 |
|---|---|
| バッファ | 名称: 20 mM HEPES 150 mM NaCl 5 mM MgCl2 5% Glycerol 1 mM DTT 200µM GDP pH: 7.5 |
| 要素 #601 | 名称: CtRoco / タイプ: protein / 記述: Rab family protein / 分子量: 127.147 / 分子数: 2 由来: Chlorobium tepidum (strain ATCC 49652 / DSM 12025 / NBRC 103806 / TLS) 参照: UniProt: Q8KC98 配列: MSYYHHHHHH DYDIPTTENL YFQGAMGSMS DLDVIRQIEQ ELGMQLEPVD KLKWYSKGYK LDKDQRVTAI GLYDCGSDTL DRIIQPLESL KSLSELSLSS NQITDISPLA SLNSLSMLWL DRNQITDIAP LASLNSLSML WLFGNKISDI APLESLKSLT ELQLSSNQIT ...配列: MSYYHHHHHH DYDIPTTENL YFQGAMGSMS DLDVIRQIEQ ELGMQLEPVD KLKWYSKGYK LDKDQRVTAI GLYDCGSDTL DRIIQPLESL KSLSELSLSS NQITDISPLA SLNSLSMLWL DRNQITDIAP LASLNSLSML WLFGNKISDI APLESLKSLT ELQLSSNQIT DIAPLASLKS LTELSLSGNN ISDIAPLESL KSLTELSLSS NQITDIAPLA SLKSLTELSL SSNQISDIAP LESLKSLTEL QLSRNQISDI APLESLKSLT ELQLSSNQIT DIAPLASLKS LTELQLSRNQ ISDIAPLESL NSLSKLWLNG NQITDIAPLA SLNSLTELEL SSNQITDIAP LASLKSLSTL WLSSNQISDI APLASLESLS ELSLSSNQIS DISPLASLNS LTGFDVRRNP IKRLPETITG FDMEILWNDF SSSGFITFFD NPLESPPPEI VKQGKEAVRQ YFQSIEEARS KGEALVHLQE IKVHLIGDGM AGKTSLLKQL IGETFDPKES QTHGLNVVTK QAPNIKGLEN DDELKECLFH FWDFGGQEIM HASHQFFMTR SSVYMLLLDS RTDSNKHYWL RHIEKYGGKS PVIVVMNKID ENPSYNIEQK KINERFPAIE NRFHRISCKN GDGVESIAKS LKSAVLHPDS IYGTPLAPSW IKVKEKLVEA TTAQRYLNRT EVEKICNDSG ITDPGERKTL LGYLNNLGIV LYFEALDLSE IYVLDPHWVT IGVYRIINSS KTKNGHLNTS ALGYILNEEQ IRCDEYDPAK NNKFTYTLLE QRYLLDIMKQ FELCYDEGKG LFIIPSNLPT QIDNEPEITE GEPLRFIMKY DYLPSTIIPR LMIAMQHQIL DRMQWRYGMV LKSQDHEGAL AKVVAETKDS TITIAIQGEP RCKREYLSII WYEIKKINAN FTNLDVKEFI PLPGHPDELV EYKELLGLEK MGRDEYVSGK LEKVFSVSKM LDSVISKEER NKERLMGDIN IKLENIGNPT IPIHQQVEVN VSQETVQHVE NLQGFFENLK ADILREAELE IDDPKERKRL ANELELAENA ITKMDAAVKS GKNKLKPDVK DRLGEFIDNL ANENSRLRKG IALVMNGAEK VQKLARYYNN VAPFFDLPSV PPVLLGKEKT |
-実験情報
| ビーム | 設備名称: SOLEIL SWING  / 地域: Saint-Aubin / 国: France / 地域: Saint-Aubin / 国: France  / 線源: X-ray synchrotron / 波長: 0.103 Å / 線源: X-ray synchrotron / 波長: 0.103 Å | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 検出器 | 名称: AVIEX / タイプ: CCD | |||||||||||||||||||||||||||||||||
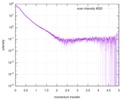
| スキャン |
| |||||||||||||||||||||||||||||||||
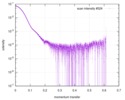
| 距離分布関数 P(R) |
| |||||||||||||||||||||||||||||||||
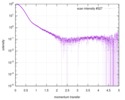
| 結果 |
|
 ムービー
ムービー コントローラー
コントローラー