[English] 日本語
 Yorodumi
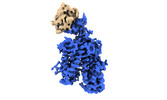
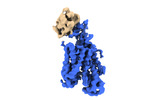
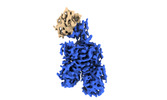
Yorodumi- PDB-8omu: Cryo-EM structure of rat SLC22A6 bound to alpha-ketoglutaric acid... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8omu | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of rat SLC22A6 bound to alpha-ketoglutaric acid in a low occupancy state | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords | METAL TRANSPORT / membrane protein / transporter | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationOrganic anion transport / renal tubular secretion / alpha-ketoglutarate transmembrane transporter activity / alpha-ketoglutarate transport / sodium-independent organic anion transmembrane transporter activity / sodium-independent organic anion transport / metanephric proximal tubule development / prostaglandin transport / prostaglandin transmembrane transporter activity / organic anion transport ...Organic anion transport / renal tubular secretion / alpha-ketoglutarate transmembrane transporter activity / alpha-ketoglutarate transport / sodium-independent organic anion transmembrane transporter activity / sodium-independent organic anion transport / metanephric proximal tubule development / prostaglandin transport / prostaglandin transmembrane transporter activity / organic anion transport / organic anion transmembrane transporter activity / solute:inorganic anion antiporter activity / monoatomic anion transport / antiporter activity / chloride ion binding / xenobiotic transmembrane transporter activity / transmembrane transporter activity / basal plasma membrane / caveola / response to organic cyclic compound / transmembrane transport / basolateral plasma membrane / protein-containing complex / identical protein binding / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |  synthetic construct (others) | ||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.43 Å | ||||||||||||
 Authors Authors | Parker, J.L. / Kato, T. / Newstead, S. | ||||||||||||
| Funding support |  United Kingdom, 3items United Kingdom, 3items
| ||||||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: Molecular basis for selective uptake and elimination of organic anions in the kidney by OAT1. Authors: Joanne L Parker / Takafumi Kato / Gabriel Kuteyi / Oleg Sitsel / Simon Newstead /   Abstract: In mammals, the kidney plays an essential role in maintaining blood homeostasis through the selective uptake, retention or elimination of toxins, drugs and metabolites. Organic anion transporters ...In mammals, the kidney plays an essential role in maintaining blood homeostasis through the selective uptake, retention or elimination of toxins, drugs and metabolites. Organic anion transporters (OATs) are responsible for the recognition of metabolites and toxins in the nephron and their eventual urinary excretion. Inhibition of OATs is used therapeutically to improve drug efficacy and reduce nephrotoxicity. The founding member of the renal organic anion transporter family, OAT1 (also known as SLC22A6), uses the export of α-ketoglutarate (α-KG), a key intermediate in the Krebs cycle, to drive selective transport and is allosterically regulated by intracellular chloride. However, the mechanisms linking metabolite cycling, drug transport and intracellular chloride remain obscure. Here, we present cryogenic-electron microscopy structures of OAT1 bound to α-KG, the antiviral tenofovir and clinical inhibitor probenecid, used in the treatment of Gout. Complementary in vivo cellular assays explain the molecular basis for α-KG driven drug elimination and the allosteric regulation of organic anion transport in the kidney by chloride. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8omu.cif.gz 8omu.cif.gz | 158.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8omu.ent.gz pdb8omu.ent.gz | 98.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8omu.json.gz 8omu.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8omu_validation.pdf.gz 8omu_validation.pdf.gz | 1.4 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8omu_full_validation.pdf.gz 8omu_full_validation.pdf.gz | 1.4 MB | Display | |
| Data in XML |  8omu_validation.xml.gz 8omu_validation.xml.gz | 35.4 KB | Display | |
| Data in CIF |  8omu_validation.cif.gz 8omu_validation.cif.gz | 50.2 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/om/8omu https://data.pdbj.org/pub/pdb/validation_reports/om/8omu ftp://data.pdbj.org/pub/pdb/validation_reports/om/8omu ftp://data.pdbj.org/pub/pdb/validation_reports/om/8omu | HTTPS FTP |
-Related structure data
| Related structure data |  16977MC  8bvrC  8bvsC  8bvtC  8bw7C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 60553.559 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|---|
| #2: Antibody | Mass: 16079.819 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.) synthetic construct (others) / Production host:  |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: OAT1 / Type: COMPLEX / Entity ID: all / Source: RECOMBINANT |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:  |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R1.2/1.3 |
| Vitrification | Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2000 nm / Nominal defocus min: 800 nm |
| Image recording | Electron dose: 42 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
| EM imaging optics | Energyfilter name: GIF Bioquantum |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.43 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 202820 / Symmetry type: POINT | ||||||||||||||||||||||||
| Atomic model building | Space: REAL | ||||||||||||||||||||||||
| Refinement | Cross valid method: NONE Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2 | ||||||||||||||||||||||||
| Displacement parameters | Biso mean: 68.38 Å2 | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller



 PDBj
PDBj




