[English] 日本語
 Yorodumi
Yorodumi- PDB-8f2k: Structure of yeast F1-ATPase determined with 100 micromolar cruen... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8f2k | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of yeast F1-ATPase determined with 100 micromolar cruentaren A | ||||||||||||||||||
 Components Components | (ATP synthase subunit ...) x 3 | ||||||||||||||||||
 Keywords Keywords | HYDROLASE / F1-ATPase / ATP Synthase / cruentaren A / drug development | ||||||||||||||||||
| Function / homology |  Function and homology information Function and homology information: / : / : / : / proton motive force-driven mitochondrial ATP synthesis / proton-transporting ATPase activity, rotational mechanism / H+-transporting two-sector ATPase / proton-transporting ATP synthase activity, rotational mechanism / ADP binding / ATP hydrolysis activity / ATP binding Similarity search - Function | ||||||||||||||||||
| Biological species |  | ||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.9 Å | ||||||||||||||||||
 Authors Authors | Guo, H. / Rubinstein, J.L. | ||||||||||||||||||
| Funding support |  Canada, Canada,  United States, 5items United States, 5items
| ||||||||||||||||||
 Citation Citation |  Journal: Chemistry / Year: 2023 Journal: Chemistry / Year: 2023Title: CryoEM Structure with ATP Synthase Enables Late-Stage Diversification of Cruentaren A. Authors: Xiaozheng Dou / Hui Guo / Terin D'Amico / Leah Abdallah / Chitra Subramanian / Bhargav A Patel / Mark Cohen / John L Rubinstein / Brian S J Blagg /   Abstract: Cruentaren A is a natural product that exhibits potent antiproliferative activity against various cancer cell lines, yet its binding site within ATP synthase remained unknown, thus limiting the ...Cruentaren A is a natural product that exhibits potent antiproliferative activity against various cancer cell lines, yet its binding site within ATP synthase remained unknown, thus limiting the development of improved analogues as anticancer agents. Herein, we report the cryogenic electron microscopy (cryoEM) structure of cruentaren A bound to ATP synthase, which allowed the design of new inhibitors through semisynthetic modification. Examples of cruentaren A derivatives include a trans-alkene isomer, which was found to exhibit similar activity to cruentaren A against three cancer cell lines as well as several other analogues that retained potent inhibitory activity. Together, these studies provide a foundation for the generation of cruentaren A derivatives as potential therapeutics for the treatment of cancer. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8f2k.cif.gz 8f2k.cif.gz | 554.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8f2k.ent.gz pdb8f2k.ent.gz | 448.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8f2k.json.gz 8f2k.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/f2/8f2k https://data.pdbj.org/pub/pdb/validation_reports/f2/8f2k ftp://data.pdbj.org/pub/pdb/validation_reports/f2/8f2k ftp://data.pdbj.org/pub/pdb/validation_reports/f2/8f2k | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  28818MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-ATP synthase subunit ... , 3 types, 7 molecules ABCDEFG
| #1: Protein | Mass: 52376.539 Da / Num. of mol.: 3 / Source method: isolated from a natural source / Source: (natural)  #2: Protein | Mass: 50379.277 Da / Num. of mol.: 3 / Source method: isolated from a natural source / Source: (natural)  References: UniProt: A0A6A5PX46, H+-transporting two-sector ATPase #3: Protein | | Mass: 30399.875 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Non-polymers , 3 types, 12 molecules 

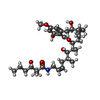


| #4: Chemical | ChemComp-ATP / #5: Chemical | ChemComp-MG / #6: Chemical | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Yeast F1-ATPase with 100 micromolar cruentaren A / Type: COMPLEX / Entity ID: #1-#3 / Source: NATURAL |
|---|---|
| Molecular weight | Value: 0.4 MDa / Experimental value: NO |
| Source (natural) | Organism:  |
| Buffer solution | pH: 7.4 |
| Specimen | Conc.: 15 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Grid material: COPPER/RHODIUM / Grid mesh size: 300 divisions/in. / Grid type: Homemade |
| Vitrification | Instrument: LEICA EM GP / Cryogen name: ETHANE-PROPANE / Humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 75000 X / Calibrated magnification: 133815 X / Nominal defocus max: 2000 nm / Nominal defocus min: 1000 nm / Cs: 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: ZEMLIN TABLEAU |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 8.4 sec. / Electron dose: 42 e/Å2 / Film or detector model: FEI FALCON IV (4k x 4k) / Num. of real images: 5814 |
| Image scans | Width: 4096 / Height: 4096 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.20.1_4487: / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 1352159 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.9 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 701444 / Algorithm: BACK PROJECTION / Num. of class averages: 3 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT / Space: RECIPROCAL | ||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 2HLD Accession code: 2HLD / Source name: PDB / Type: experimental model | ||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller



 PDBj
PDBj






