+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8dhm | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human TMEM175 in complex with 4-aminopyridine | |||||||||
 Components Components | Endosomal/lysosomal potassium channel TMEM175 | |||||||||
 Keywords Keywords | TRANSPORT PROTEIN / potassium channel / ion channel | |||||||||
| Function / homology |  Function and homology information Function and homology informationlysosomal lumen pH elevation / phagosome-lysosome fusion / regulation of lysosomal lumen pH / potassium ion leak channel activity / proton channel activity / arachidonate binding / potassium channel activity / potassium ion transmembrane transport / proton transmembrane transport / neuron cellular homeostasis ...lysosomal lumen pH elevation / phagosome-lysosome fusion / regulation of lysosomal lumen pH / potassium ion leak channel activity / proton channel activity / arachidonate binding / potassium channel activity / potassium ion transmembrane transport / proton transmembrane transport / neuron cellular homeostasis / lysosome / endosome membrane / endosome / lysosomal membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.73 Å | |||||||||
 Authors Authors | Oh, S. / Hite, R.K. | |||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2022 Journal: Proc Natl Acad Sci U S A / Year: 2022Title: Mechanism of 4-aminopyridine inhibition of the lysosomal channel TMEM175. Authors: SeCheol Oh / Robyn Stix / Wenchang Zhou / José D Faraldo-Gómez / Richard K Hite /  Abstract: Transmembrane protein 175 (TMEM175) is an evolutionarily distinct lysosomal cation channel whose mutation is associated with the development of Parkinson's disease. Here, we present a cryoelectron ...Transmembrane protein 175 (TMEM175) is an evolutionarily distinct lysosomal cation channel whose mutation is associated with the development of Parkinson's disease. Here, we present a cryoelectron microscopy structure and molecular simulations of TMEM175 bound to 4-aminopyridine (4-AP), the only known small-molecule inhibitor of TMEM175 and a broad K channel inhibitor, as well as a drug approved by the Food and Drug Administration against multiple sclerosis. The structure shows that 4-AP, whose mode of action had not been previously visualized, binds near the center of the ion conduction pathway, in the open state of the channel. Molecular dynamics simulations reveal that this binding site is near the middle of the transmembrane potential gradient, providing a rationale for the voltage-dependent dissociation of 4-AP from TMEM175. Interestingly, bound 4-AP rapidly switches between three predominant binding poses, stabilized by alternate interaction patterns dictated by the twofold symmetry of the channel. Despite this highly dynamic binding mode, bound 4-AP prevents not only ion permeation but also water flow. Together, these studies provide a framework for the rational design of novel small-molecule inhibitors of TMEM175 that might reveal the role of this channel in human lysosomal physiology both in health and disease. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8dhm.cif.gz 8dhm.cif.gz | 251.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8dhm.ent.gz pdb8dhm.ent.gz | 203.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8dhm.json.gz 8dhm.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8dhm_validation.pdf.gz 8dhm_validation.pdf.gz | 1.2 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8dhm_full_validation.pdf.gz 8dhm_full_validation.pdf.gz | 1.2 MB | Display | |
| Data in XML |  8dhm_validation.xml.gz 8dhm_validation.xml.gz | 40.4 KB | Display | |
| Data in CIF |  8dhm_validation.cif.gz 8dhm_validation.cif.gz | 57.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dh/8dhm https://data.pdbj.org/pub/pdb/validation_reports/dh/8dhm ftp://data.pdbj.org/pub/pdb/validation_reports/dh/8dhm ftp://data.pdbj.org/pub/pdb/validation_reports/dh/8dhm | HTTPS FTP |
-Related structure data
| Related structure data |  27436MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
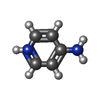
| #1: Protein | Mass: 55667.219 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TMEM175 / Production host: Homo sapiens (human) / Gene: TMEM175 / Production host:  Homo sapiens (human) / References: UniProt: Q9BSA9 Homo sapiens (human) / References: UniProt: Q9BSA9#2: Chemical | ChemComp-K / #3: Chemical | ChemComp-4AP / | #4: Water | ChemComp-HOH / | Has ligand of interest | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Human TMEM175 / Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Experimental value: NO | |||||||||||||||||||||||||||||||||||
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||||||||||||||||
| Source (recombinant) | Organism:  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||||||||||||||||
| Buffer solution | pH: 8 | |||||||||||||||||||||||||||||||||||
| Buffer component |
| |||||||||||||||||||||||||||||||||||
| Specimen | Conc.: 4 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | |||||||||||||||||||||||||||||||||||
| Specimen support | Grid material: GOLD / Grid mesh size: 400 divisions/in. / Grid type: Quantifoil R1.2/1.3 | |||||||||||||||||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 297 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2500 nm / Nominal defocus min: 1000 nm |
| Image recording | Electron dose: 61 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.19.2_4158: / Classification: refinement | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 4153614 | ||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C2 (2 fold cyclic) | ||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.73 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 261536 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: OTHER / Space: REAL / Details: Real Space Refinement | ||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 6WC9 Accession code: 6WC9 / Source name: PDB / Type: experimental model | ||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller



 PDBj
PDBj