[English] 日本語
 Yorodumi
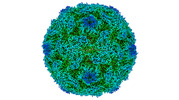
Yorodumi- PDB-8ayz: Poliovirus type 2 (strain MEF-1) virus-like particle in complex w... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8ayz | ||||||
|---|---|---|---|---|---|---|---|
| Title | Poliovirus type 2 (strain MEF-1) virus-like particle in complex with capsid binder compound 17 | ||||||
 Components Components | (Capsid protein, ...) x 3 | ||||||
 Keywords Keywords | VIRUS LIKE PARTICLE / Capsid protein / inhibitor / complex | ||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of RIG-I activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of MDA-5 activity / picornain 2A / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of MAVS activity / symbiont-mediated suppression of host mRNA export from nucleus / ribonucleoside triphosphate phosphatase activity / symbiont genome entry into host cell via pore formation in plasma membrane / picornain 3C / T=pseudo3 icosahedral viral capsid / host cell cytoplasmic vesicle membrane ...symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of RIG-I activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of MDA-5 activity / picornain 2A / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of MAVS activity / symbiont-mediated suppression of host mRNA export from nucleus / ribonucleoside triphosphate phosphatase activity / symbiont genome entry into host cell via pore formation in plasma membrane / picornain 3C / T=pseudo3 icosahedral viral capsid / host cell cytoplasmic vesicle membrane / nucleoside-triphosphate phosphatase / protein complex oligomerization / monoatomic ion channel activity / symbiont-mediated suppression of host gene expression / DNA replication / receptor-mediated endocytosis of virus by host cell / RNA helicase activity / induction by virus of host autophagy / RNA-directed RNA polymerase / viral RNA genome replication / cysteine-type endopeptidase activity / RNA-dependent RNA polymerase activity / virus-mediated perturbation of host defense response / DNA-templated transcription / host cell nucleus / virion attachment to host cell / structural molecule activity / proteolysis / RNA binding / ATP binding / membrane / metal ion binding Similarity search - Function | ||||||
| Biological species |  Human poliovirus 2 Human poliovirus 2 | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 1.88 Å | ||||||
 Authors Authors | Bahar, M.W. / Fry, E.E. / Stuart, D.I. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Commun Biol / Year: 2022 Journal: Commun Biol / Year: 2022Title: A conserved glutathione binding site in poliovirus is a target for antivirals and vaccine stabilisation. Authors: Mohammad W Bahar / Veronica Nasta / Helen Fox / Lee Sherry / Keith Grehan / Claudine Porta / Andrew J Macadam / Nicola J Stonehouse / David J Rowlands / Elizabeth E Fry / David I Stuart /   Abstract: Strategies to prevent the recurrence of poliovirus (PV) after eradication may utilise non-infectious, recombinant virus-like particle (VLP) vaccines. Despite clear advantages over inactivated or ...Strategies to prevent the recurrence of poliovirus (PV) after eradication may utilise non-infectious, recombinant virus-like particle (VLP) vaccines. Despite clear advantages over inactivated or attenuated virus vaccines, instability of VLPs can compromise their immunogenicity. Glutathione (GSH), an important cellular reducing agent, is a crucial co-factor for the morphogenesis of enteroviruses, including PV. We report cryo-EM structures of GSH bound to PV serotype 3 VLPs showing that it can enhance particle stability. GSH binds the positively charged pocket at the interprotomer interface shown recently to bind GSH in enterovirus F3 and putative antiviral benzene sulphonamide compounds in other enteroviruses. We show, using high-resolution cryo-EM, the binding of a benzene sulphonamide compound with a PV serotype 2 VLP, consistent with antiviral activity through over-stabilizing the interprotomer pocket, preventing the capsid rearrangements necessary for viral infection. Collectively, these results suggest GSH or an analogous tight-binding antiviral offers the potential for stabilizing VLP vaccines. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8ayz.cif.gz 8ayz.cif.gz | 156.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8ayz.ent.gz pdb8ayz.ent.gz | 124.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8ayz.json.gz 8ayz.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8ayz_validation.pdf.gz 8ayz_validation.pdf.gz | 1.4 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8ayz_full_validation.pdf.gz 8ayz_full_validation.pdf.gz | 1.4 MB | Display | |
| Data in XML |  8ayz_validation.xml.gz 8ayz_validation.xml.gz | 44.3 KB | Display | |
| Data in CIF |  8ayz_validation.cif.gz 8ayz_validation.cif.gz | 66.1 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ay/8ayz https://data.pdbj.org/pub/pdb/validation_reports/ay/8ayz ftp://data.pdbj.org/pub/pdb/validation_reports/ay/8ayz ftp://data.pdbj.org/pub/pdb/validation_reports/ay/8ayz | HTTPS FTP |
-Related structure data
| Related structure data | 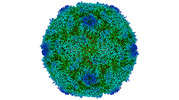 15727MC  8ayxC  8ayyC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 | x 60
|
- Components
Components
-Capsid protein, ... , 3 types, 3 molecules ABC
| #1: Protein | Mass: 33111.207 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Human poliovirus 2 / Strain: MEF-1 / Plasmid: MVA-T7-Polio2 pIRES / Details (production host): Vaccinia virus transfer vector / Cell line (production host): BHK-21 / Production host: Human poliovirus 2 / Strain: MEF-1 / Plasmid: MVA-T7-Polio2 pIRES / Details (production host): Vaccinia virus transfer vector / Cell line (production host): BHK-21 / Production host:  Mesocricetus auratus (golden hamster) / References: UniProt: P06210 Mesocricetus auratus (golden hamster) / References: UniProt: P06210 |
|---|---|
| #2: Protein | Mass: 37456.855 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: Sequence is given for the VP0 polypeptide. / Source: (gene. exp.)  Human poliovirus 2 / Strain: MEF-1 / Plasmid: MVA-T7-Polio2 pIRES / Details (production host): Vaccinia virus transfer vector / Cell line (production host): BHK-21 / Production host: Human poliovirus 2 / Strain: MEF-1 / Plasmid: MVA-T7-Polio2 pIRES / Details (production host): Vaccinia virus transfer vector / Cell line (production host): BHK-21 / Production host:  Mesocricetus auratus (golden hamster) / References: UniProt: P06210 Mesocricetus auratus (golden hamster) / References: UniProt: P06210 |
| #3: Protein | Mass: 26472.293 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Human poliovirus 2 / Strain: MEF-1 / Plasmid: MVA-T7-Polio2 pIRES / Details (production host): Vaccinia virus transfer vector / Cell line (production host): BHK-21 / Production host: Human poliovirus 2 / Strain: MEF-1 / Plasmid: MVA-T7-Polio2 pIRES / Details (production host): Vaccinia virus transfer vector / Cell line (production host): BHK-21 / Production host:  Mesocricetus auratus (golden hamster) / References: UniProt: P06210 Mesocricetus auratus (golden hamster) / References: UniProt: P06210 |
-Non-polymers , 3 types, 264 molecules 
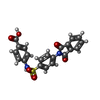



| #4: Chemical | ChemComp-SPH / |
|---|---|
| #5: Chemical | ChemComp-FHK / |
| #6: Water | ChemComp-HOH / |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Human poliovirus 2 / Type: VIRUS Details: Recombinantly expressed virus-like particle of PV2 (strain MEF-1) Entity ID: #1-#3 / Source: RECOMBINANT | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 5.81 MDa / Experimental value: NO | ||||||||||||
| Source (natural) | Organism:  Human poliovirus 2 / Strain: MEF-1 Human poliovirus 2 / Strain: MEF-1 | ||||||||||||
| Source (recombinant) | Organism:  Mesocricetus auratus (golden hamster) / Cell: BHK-21 / Plasmid: MVA-T7-Polio2 pIRES Mesocricetus auratus (golden hamster) / Cell: BHK-21 / Plasmid: MVA-T7-Polio2 pIRES | ||||||||||||
| Details of virus | Empty: YES / Enveloped: NO / Isolate: SEROTYPE / Type: VIRUS-LIKE PARTICLE | ||||||||||||
| Natural host | Organism: mammal environmental sample | ||||||||||||
| Virus shell | Name: Virus shell 1 / Diameter: 310 nm / Triangulation number (T number): 1 | ||||||||||||
| Buffer solution | pH: 7 Details: 1 x DPBS was made from tissue culture grade Dulbecco's Phosphate Buffered Saline (Sigma-Aldrich). 20 mM EDTA was prepared from fresh stocks of buffered EDTA. | ||||||||||||
| Buffer component |
| ||||||||||||
| Specimen | Conc.: 0.75 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES Details: Recombinant VLP of PV2 mixed with capsid inhibitor compound 17 at a molar ratio of 1:2500. | ||||||||||||
| Specimen support | Details: The specific type of grid used was Ultra-thin carbon support film, 3nm - on lacey carbon AGS187-4 from Agar Scientific. Grid material: COPPER / Grid mesh size: 400 divisions/in. / Grid type: EMS Lacey Carbon | ||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE-PROPANE / Humidity: 100 % / Chamber temperature: 277.15 K Details: Single blotting with 3.5ul of sample. Four second blot time and -17 blot force on the vitrobot. |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: TFS KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 105000 X / Calibrated magnification: 60314 X / Nominal defocus max: 2300 nm / Nominal defocus min: 800 nm / Cs: 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 1.7 sec. / Electron dose: 34.68 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 4750 Details: Images were collected in electron counting mode with a calibrated pixel size of 0.829 A/pixel. Super-resolution pixel size 0.4145 A/pixel. Movies were collected in 50 frames. |
| EM imaging optics | Energyfilter name: GIF Quantum LS / Energyfilter slit width: 20 eV |
| Image scans | Sampling size: 5 µm / Width: 5760 / Height: 4092 |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Image processing | Details: All frames were used for motion correction. | ||||||||||||||||||||||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 157002 Details: Particle selection was automated with ETHAN particle picking software. | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: I (icosahedral) | ||||||||||||||||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 1.88 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 51518 / Algorithm: BACK PROJECTION Details: Final reconstruction was Ewald sphere corrected and sharpened with Post-processing in RELION using an inverse B-factor of -15.0 Angstroms. B-factor was selected ad-hoc while testing a range ...Details: Final reconstruction was Ewald sphere corrected and sharpened with Post-processing in RELION using an inverse B-factor of -15.0 Angstroms. B-factor was selected ad-hoc while testing a range of values for map interpretability. Num. of class averages: 2 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT / Space: REAL / Target criteria: Correlation coefficient Details: Initial model was rigid body fitted using UCSF chimera and Coot. Global minimization and B-factor refinement was performed in real space using phenix_real.space.refine. | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 1EAH |
 Movie
Movie Controller
Controller



 PDBj
PDBj




