+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 7zbz | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
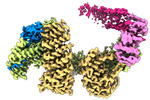
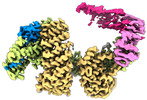
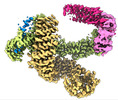
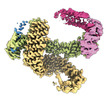
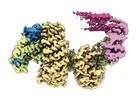
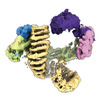
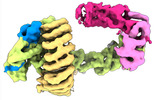
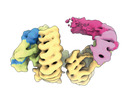
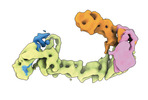
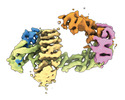
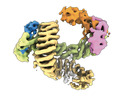
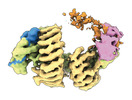
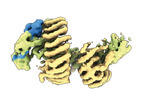
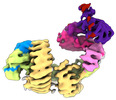
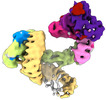
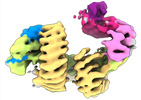
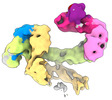
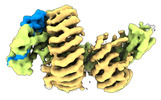
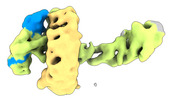
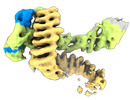
| タイトル | CAND1 delhairpin-SCF-SKP2 CAND1 partly engaged SCF partly rocked | |||||||||
 要素 要素 |
| |||||||||
 キーワード キーワード | LIGASE / cullin-RING E3 ubiquitin ligase / SCF / CAND1 / Assembly factor | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報SCF complex assembly / positive regulation of protein polyubiquitination / Parkin-FBXW7-Cul1 ubiquitin ligase complex / F-box domain binding / Aberrant regulation of mitotic exit in cancer due to RB1 defects / PcG protein complex / negative regulation of catalytic activity / cullin-RING ubiquitin ligase complex / Cul7-RING ubiquitin ligase complex / positive regulation of ubiquitin protein ligase activity ...SCF complex assembly / positive regulation of protein polyubiquitination / Parkin-FBXW7-Cul1 ubiquitin ligase complex / F-box domain binding / Aberrant regulation of mitotic exit in cancer due to RB1 defects / PcG protein complex / negative regulation of catalytic activity / cullin-RING ubiquitin ligase complex / Cul7-RING ubiquitin ligase complex / positive regulation of ubiquitin protein ligase activity / Loss of Function of FBXW7 in Cancer and NOTCH1 Signaling / maintenance of protein location in nucleus / positive regulation of protein autoubiquitination / protein neddylation / NEDD8 ligase activity / Cul5-RING ubiquitin ligase complex / positive regulation of intracellular estrogen receptor signaling pathway / ubiquitin-ubiquitin ligase activity / Cul4A-RING E3 ubiquitin ligase complex / SCF ubiquitin ligase complex / Cul2-RING ubiquitin ligase complex / Cul4B-RING E3 ubiquitin ligase complex / ubiquitin ligase complex scaffold activity / SCF-dependent proteasomal ubiquitin-dependent protein catabolic process / Cul3-RING ubiquitin ligase complex / Prolactin receptor signaling / protein monoubiquitination / cullin family protein binding / protein K63-linked ubiquitination / ubiquitin-like ligase-substrate adaptor activity / positive regulation of double-strand break repair via homologous recombination / positive regulation of RNA polymerase II transcription preinitiation complex assembly / protein K48-linked ubiquitination / Nuclear events stimulated by ALK signaling in cancer / ubiquitin ligase complex / TBP-class protein binding / Regulation of BACH1 activity / intrinsic apoptotic signaling pathway / MAP3K8 (TPL2)-dependent MAPK1/3 activation / SCF-beta-TrCP mediated degradation of Emi1 / NIK-->noncanonical NF-kB signaling / molecular function activator activity / Vpu mediated degradation of CD4 / Dectin-1 mediated noncanonical NF-kB signaling / Degradation of GLI1 by the proteasome / Activation of NF-kappaB in B cells / animal organ morphogenesis / Negative regulation of NOTCH4 signaling / Iron uptake and transport / GSK3B and BTRC:CUL1-mediated-degradation of NFE2L2 / Degradation of GLI2 by the proteasome / GLI3 is processed to GLI3R by the proteasome / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / APC/C:Cdh1 mediated degradation of Cdc20 and other APC/C:Cdh1 targeted proteins in late mitosis/early G1 / Degradation of beta-catenin by the destruction complex / NOTCH1 Intracellular Domain Regulates Transcription / CLEC7A (Dectin-1) signaling / SCF(Skp2)-mediated degradation of p27/p21 / beta-catenin binding / Constitutive Signaling by NOTCH1 PEST Domain Mutants / Constitutive Signaling by NOTCH1 HD+PEST Domain Mutants / FCERI mediated NF-kB activation / Interleukin-1 signaling / Orc1 removal from chromatin / G1/S transition of mitotic cell cycle / protein polyubiquitination / G2/M transition of mitotic cell cycle / Regulation of RUNX2 expression and activity / Cyclin D associated events in G1 / Regulation of PLK1 Activity at G2/M Transition / ubiquitin protein ligase activity / Circadian Clock / Antigen processing: Ubiquitination & Proteasome degradation / positive regulation of proteasomal ubiquitin-dependent protein catabolic process / Downstream TCR signaling / protein-macromolecule adaptor activity / Neddylation / mitotic cell cycle / ubiquitin-dependent protein catabolic process / regulation of apoptotic process / proteasome-mediated ubiquitin-dependent protein catabolic process / secretory granule lumen / positive regulation of canonical NF-kappaB signal transduction / defense response to virus / RNA polymerase II-specific DNA-binding transcription factor binding / ficolin-1-rich granule lumen / cell population proliferation / cell differentiation / Ub-specific processing proteases / regulation of cell cycle / protein ubiquitination / chromatin remodeling / protein domain specific binding / innate immune response / centrosome / ubiquitin protein ligase binding / Neutrophil degranulation / nucleolus / Golgi apparatus / zinc ion binding 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | 電子顕微鏡法 / 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.1 Å | |||||||||
 データ登録者 データ登録者 | Baek, K. / Schulman, B.A. | |||||||||
| 資金援助 |  ドイツ, European Union, 2件 ドイツ, European Union, 2件
| |||||||||
 引用 引用 |  ジャーナル: Cell / 年: 2023 ジャーナル: Cell / 年: 2023タイトル: Systemwide disassembly and assembly of SCF ubiquitin ligase complexes. 著者: Kheewoong Baek / Daniel C Scott / Lukas T Henneberg / Moeko T King / Matthias Mann / Brenda A Schulman /   要旨: Cells respond to environmental cues by remodeling their inventories of multiprotein complexes. Cellular repertoires of SCF (SKP1-CUL1-F box protein) ubiquitin ligase complexes, which mediate much ...Cells respond to environmental cues by remodeling their inventories of multiprotein complexes. Cellular repertoires of SCF (SKP1-CUL1-F box protein) ubiquitin ligase complexes, which mediate much protein degradation, require CAND1 to distribute the limiting CUL1 subunit across the family of ∼70 different F box proteins. Yet, how a single factor coordinately assembles numerous distinct multiprotein complexes remains unknown. We obtained cryo-EM structures of CAND1-bound SCF complexes in multiple states and correlated mutational effects on structures, biochemistry, and cellular assays. The data suggest that CAND1 clasps idling catalytic domains of an inactive SCF, rolls around, and allosterically rocks and destabilizes the SCF. New SCF production proceeds in reverse, through SKP1-F box allosterically destabilizing CAND1. The CAND1-SCF conformational ensemble recycles CUL1 from inactive complexes, fueling mixing and matching of SCF parts for E3 activation in response to substrate availability. Our data reveal biogenesis of a predominant family of E3 ligases, and the molecular basis for systemwide multiprotein complex assembly. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  7zbz.cif.gz 7zbz.cif.gz | 419.8 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb7zbz.ent.gz pdb7zbz.ent.gz | 328.7 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  7zbz.json.gz 7zbz.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| 文書・要旨 |  7zbz_validation.pdf.gz 7zbz_validation.pdf.gz | 1.1 MB | 表示 |  wwPDB検証レポート wwPDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  7zbz_full_validation.pdf.gz 7zbz_full_validation.pdf.gz | 1.1 MB | 表示 | |
| XML形式データ |  7zbz_validation.xml.gz 7zbz_validation.xml.gz | 71.3 KB | 表示 | |
| CIF形式データ |  7zbz_validation.cif.gz 7zbz_validation.cif.gz | 108.3 KB | 表示 | |
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/zb/7zbz https://data.pdbj.org/pub/pdb/validation_reports/zb/7zbz ftp://data.pdbj.org/pub/pdb/validation_reports/zb/7zbz ftp://data.pdbj.org/pub/pdb/validation_reports/zb/7zbz | HTTPS FTP |
-関連構造データ
| 関連構造データ | 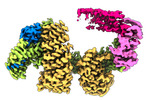 14597MC  7z8rC  7z8tC  7z8vC  7zbwC  8cdjC  8cdkC M: このデータのモデリングに利用したマップデータ C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
|
|---|---|
| 1 |
|
- 要素
要素
-タンパク質 , 3種, 3分子 CDR
| #1: タンパク質 | 分子量: 89800.367 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: CUL1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: CUL1 / 発現宿主:  Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: Q13616 Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: Q13616 |
|---|---|
| #2: タンパク質 | 分子量: 137314.156 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: CAND1, KIAA0829, TIP120, TIP120A / 発現宿主: Homo sapiens (ヒト) / 遺伝子: CAND1, KIAA0829, TIP120, TIP120A / 発現宿主:  |
| #3: タンパク質 | 分子量: 12089.677 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: RBX1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: RBX1 / 発現宿主:  Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: I2CVJ6 Trichoplusia ni (イラクサキンウワバ) / 参照: UniProt: I2CVJ6 |
-S-phase kinase-associated protein ... , 2種, 2分子 SF
| #4: タンパク質 | 分子量: 18679.965 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: SKP1, EMC19, OCP2, SKP1A, TCEB1L / 発現宿主: Homo sapiens (ヒト) / 遺伝子: SKP1, EMC19, OCP2, SKP1A, TCEB1L / 発現宿主:  |
|---|---|
| #5: タンパク質 | 分子量: 47961.910 Da / 分子数: 1 / 由来タイプ: 組換発現 詳細: SKP2 N-terminal region not visible in EM density. The LRR region of SKP2 was wholesale docked from previous structure with all sidechains removed. 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: SKP2, FBXL1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: SKP2, FBXL1 / 発現宿主:  |
-非ポリマー , 1種, 3分子 
| #6: 化合物 |
|---|
-詳細
| 研究の焦点であるリガンドがあるか | Y |
|---|
-実験情報
-実験
| 実験 | 手法: 電子顕微鏡法 |
|---|---|
| EM実験 | 試料の集合状態: PARTICLE / 3次元再構成法: 単粒子再構成法 |
- 試料調製
試料調製
| 構成要素 |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 由来(天然) |
| ||||||||||||||||||||||||
| 由来(組換発現) |
| ||||||||||||||||||||||||
| 緩衝液 | pH: 7.5 | ||||||||||||||||||||||||
| 試料 | 濃度: 2 mg/ml / 包埋: NO / シャドウイング: NO / 染色: NO / 凍結: YES | ||||||||||||||||||||||||
| 急速凍結 | 凍結剤: ETHANE |
- 電子顕微鏡撮影
電子顕微鏡撮影
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
|---|---|
| 顕微鏡 | モデル: FEI TITAN KRIOS |
| 電子銃 | 電子線源:  FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM |
| 電子レンズ | モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2000 nm / 最小 デフォーカス(公称値): 500 nm |
| 撮影 | 電子線照射量: 68 e/Å2 フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) |
- 解析
解析
| ソフトウェア | 名称: PHENIX / バージョン: 1.19.2_4158: / 分類: 精密化 | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF補正 | タイプ: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3次元再構成 | 解像度: 3.1 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 粒子像の数: 196974 / 対称性のタイプ: POINT | ||||||||||||||||||||||||
| 拘束条件 |
|
 ムービー
ムービー コントローラー
コントローラー




























 PDBj
PDBj












