+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7smp | ||||||
|---|---|---|---|---|---|---|---|
| Title | CryoEM structure of NKCC1 Bu-I | ||||||
 Components Components | Solute carrier family 12 member 2 | ||||||
 Keywords Keywords | MEMBRANE PROTEIN / Transport Protein / cotransporter / dimer / ion | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of cell volume / positive regulation of aspartate secretion / transepithelial ammonium transport / regulation of matrix metallopeptidase secretion / metal ion transmembrane transporter activity / cell body membrane / inorganic anion import across plasma membrane / inorganic cation import across plasma membrane / chloride:monoatomic cation symporter activity / sodium:potassium:chloride symporter activity ...positive regulation of cell volume / positive regulation of aspartate secretion / transepithelial ammonium transport / regulation of matrix metallopeptidase secretion / metal ion transmembrane transporter activity / cell body membrane / inorganic anion import across plasma membrane / inorganic cation import across plasma membrane / chloride:monoatomic cation symporter activity / sodium:potassium:chloride symporter activity / transepithelial chloride transport / potassium ion transmembrane transporter activity / Cation-coupled Chloride cotransporters / sodium ion homeostasis / intracellular chloride ion homeostasis / negative regulation of vascular wound healing / ammonium transmembrane transport / chloride ion homeostasis / cellular response to potassium ion / cell projection membrane / ammonium channel activity / intracellular potassium ion homeostasis / potassium ion homeostasis / sodium ion import across plasma membrane / T cell chemotaxis / cellular response to chemokine / intracellular sodium ion homeostasis / hyperosmotic response / cell volume homeostasis / gamma-aminobutyric acid signaling pathway / regulation of spontaneous synaptic transmission / maintenance of blood-brain barrier / potassium ion import across plasma membrane / transport across blood-brain barrier / lateral plasma membrane / sodium ion transmembrane transport / monoatomic ion transport / basal plasma membrane / chloride transmembrane transport / cell projection / cell periphery / Hsp90 protein binding / cytoplasmic vesicle membrane / extracellular vesicle / protein-folding chaperone binding / basolateral plasma membrane / cell body / neuron projection / apical plasma membrane / neuronal cell body / protein kinase binding / extracellular exosome / membrane / plasma membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.28 Å | ||||||
 Authors Authors | Moseng, M.A. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Sci Adv / Year: 2022 Journal: Sci Adv / Year: 2022Title: Inhibition mechanism of NKCC1 involves the carboxyl terminus and long-range conformational coupling. Authors: Mitchell A Moseng / Chih-Chia Su / Kerri Rios / Meng Cui / Meinan Lyu / Przemyslaw Glaza / Philip A Klenotic / Eric Delpire / Edward W Yu /  Abstract: The Na-K-2Cl cotransporter-1 (NKCC1) is an electroneutral Na-dependent transporter responsible for simultaneously translocating Na, K, and Cl ions into cells. In human tissue, NKCC1 plays a critical ...The Na-K-2Cl cotransporter-1 (NKCC1) is an electroneutral Na-dependent transporter responsible for simultaneously translocating Na, K, and Cl ions into cells. In human tissue, NKCC1 plays a critical role in regulating cytoplasmic volume, fluid intake, chloride homeostasis, and cell polarity. Here, we report four structures of human NKCC1 (hNKCC1), both in the absence and presence of loop diuretic (bumetanide or furosemide), using single-particle cryo-electron microscopy. These structures allow us to directly observe various novel conformations of the hNKCC1 dimer. They also reveal two drug-binding sites located at the transmembrane and cytosolic carboxyl-terminal domains, respectively. Together, our findings enable us to delineate an inhibition mechanism that involves a coupled movement between the cytosolic and transmembrane domains of hNKCC1. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7smp.cif.gz 7smp.cif.gz | 548.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7smp.ent.gz pdb7smp.ent.gz | 456.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7smp.json.gz 7smp.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  7smp_validation.pdf.gz 7smp_validation.pdf.gz | 1.8 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  7smp_full_validation.pdf.gz 7smp_full_validation.pdf.gz | 1.8 MB | Display | |
| Data in XML |  7smp_validation.xml.gz 7smp_validation.xml.gz | 64.2 KB | Display | |
| Data in CIF |  7smp_validation.cif.gz 7smp_validation.cif.gz | 94.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sm/7smp https://data.pdbj.org/pub/pdb/validation_reports/sm/7smp ftp://data.pdbj.org/pub/pdb/validation_reports/sm/7smp ftp://data.pdbj.org/pub/pdb/validation_reports/sm/7smp | HTTPS FTP |
-Related structure data
| Related structure data |  25204MC  7mxoC  7n3nC  7sflC  8steC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
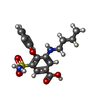
| #1: Protein | Mass: 103313.422 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SLC12A2, NKCC1 / Production host: Homo sapiens (human) / Gene: SLC12A2, NKCC1 / Production host:  Homo sapiens (human) / References: UniProt: P55011 Homo sapiens (human) / References: UniProt: P55011#2: Chemical | #3: Chemical | Has ligand of interest | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: NKCC1 Bu-I / Type: ORGANELLE OR CELLULAR COMPONENT / Entity ID: #1 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 5000 nm / Nominal defocus min: 1200 nm |
| Image recording | Electron dose: 40 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.19.2_4158: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING ONLY | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.28 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 69614 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller




 PDBj
PDBj