[English] 日本語
 Yorodumi
Yorodumi- EMDB-9846: Cryo-EM structure of glutamate dehydrogenase from Thermococcus pr... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9846 | |||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of glutamate dehydrogenase from Thermococcus profundus | |||||||||||||||||||||||||||||||||||||||||||||
 Map data Map data | structure of glutamate dehydrogenase from Thermococcus profundus | |||||||||||||||||||||||||||||||||||||||||||||
 Sample Sample |
| |||||||||||||||||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationglutamate dehydrogenase [NAD(P)+] / glutamate dehydrogenase (NADP+) activity / glutamate dehydrogenase (NAD+) activity / amino acid metabolic process Similarity search - Function | |||||||||||||||||||||||||||||||||||||||||||||
| Biological species |   Thermococcus profundus (archaea) Thermococcus profundus (archaea) | |||||||||||||||||||||||||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.7 Å | |||||||||||||||||||||||||||||||||||||||||||||
 Authors Authors | Oide M / Kato T / Oroguchi T / Nakasako M | |||||||||||||||||||||||||||||||||||||||||||||
| Funding support |  Japan, 14 items Japan, 14 items
| |||||||||||||||||||||||||||||||||||||||||||||
 Citation Citation |  Journal: FEBS J / Year: 2020 Journal: FEBS J / Year: 2020Title: Energy landscape of domain motion in glutamate dehydrogenase deduced from cryo-electron microscopy. Authors: Mao Oide / Takayuki Kato / Tomotaka Oroguchi / Masayoshi Nakasako /  Abstract: Analysis of the conformational changes of protein is important to elucidate the mechanisms of protein motions correlating with their function. Here, we studied the spontaneous domain motion of ...Analysis of the conformational changes of protein is important to elucidate the mechanisms of protein motions correlating with their function. Here, we studied the spontaneous domain motion of unliganded glutamate dehydrogenase from Thermococcus profundus using cryo-electron microscopy and proposed a novel method to construct free-energy landscape of protein conformations. Each subunit of the homo-hexameric enzyme comprises nucleotide-binding domain (NAD domain) and hexamer-forming core domain. A large active-site cleft is situated between the two domains and varies from open to close according to the motion of a NAD domain. A three-dimensional map reconstructed from all cryo-electron microscopy images displayed disordered volumes of NAD domains, suggesting that NAD domains in the collected images adopted various conformations in domain motion. Focused classifications on NAD domain of subunits provided several maps of possible conformations in domain motion. To deduce what kinds of conformations appeared in EM images, we developed a novel analysis method that describe the EM maps as a linear combination of representative conformations appearing in a 200-ns molecular dynamics simulation as reference. The analysis enabled us to estimate the appearance frequencies of the representative conformations, which illustrated a free-energy landscape in domain motion. In the open/close domain motion, two free-energy basins hindered the direct transformation from open to closed state. Structure models constructed for representative EM maps in classifications demonstrated the correlation between the energy landscape and conformations in domain motion. Based on the results, the domain motion in glutamate dehydrogenase and the analysis method to visualize conformational changes and free-energy landscape were discussed. DATABASE: The EM maps of the four conformations were deposited to Electron Microscopy Data Bank (EMDB) as accession codes EMD-9845 (open), EMD-9846 (half-open1), EMD-9847 (half-open2), and EMD-9848 (closed), respectively. In addition, the structural models built for the four conformations were deposited to the Protein Data Bank (PDB) as accession codes 6JN9 (open), 6JNA (half-open1), 6JNC (half-open2), and 6JND (closed), respectively. | |||||||||||||||||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9846.map.gz emd_9846.map.gz | 40.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9846-v30.xml emd-9846-v30.xml emd-9846.xml emd-9846.xml | 11.2 KB 11.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9846.png emd_9846.png | 80.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9846 http://ftp.pdbj.org/pub/emdb/structures/EMD-9846 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9846 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9846 | HTTPS FTP |
-Validation report
| Summary document |  emd_9846_validation.pdf.gz emd_9846_validation.pdf.gz | 390.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_9846_full_validation.pdf.gz emd_9846_full_validation.pdf.gz | 389.8 KB | Display | |
| Data in XML |  emd_9846_validation.xml.gz emd_9846_validation.xml.gz | 5.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9846 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9846 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9846 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9846 | HTTPS FTP |
-Related structure data
| Related structure data |  6jnaMC  9845C  9847C  9848C  6jn9C  6jncC  6jndC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_9846.map.gz / Format: CCP4 / Size: 42.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9846.map.gz / Format: CCP4 / Size: 42.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | structure of glutamate dehydrogenase from Thermococcus profundus | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.87 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
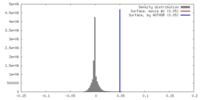
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Hexamer of glutamate dehydrogenase
| Entire | Name: Hexamer of glutamate dehydrogenase |
|---|---|
| Components |
|
-Supramolecule #1: Hexamer of glutamate dehydrogenase
| Supramolecule | Name: Hexamer of glutamate dehydrogenase / type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:   Thermococcus profundus (archaea) Thermococcus profundus (archaea) |
| Recombinant expression | Organism:  |
| Molecular weight | Theoretical: 280 KDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | JEOL CRYO ARM 200 |
|---|---|
| Image recording | Film or detector model: GATAN K2 BASE (4k x 4k) / Average electron dose: 10.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD |
- Image processing
Image processing
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.7 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 109568 |
|---|---|
| Initial angle assignment | Type: NOT APPLICABLE |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)