[English] 日本語
 Yorodumi
Yorodumi- EMDB-9220: Cryo-EM structures and dynamics of substrate-engaged human 26S pr... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9220 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structures and dynamics of substrate-engaged human 26S proteasome | ||||||||||||
 Map data Map data | The complete map of state EC2 of substrate-engaged human proteasome, low pass-filtered to 3 Angstrom without amplitude correction | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | Proteosome / HYDROLASE | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationthyrotropin-releasing hormone receptor binding / Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / nuclear proteasome complex / host-mediated perturbation of viral transcription / positive regulation of inclusion body assembly / integrator complex / proteasome accessory complex / meiosis I / purine ribonucleoside triphosphate binding ...thyrotropin-releasing hormone receptor binding / Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / nuclear proteasome complex / host-mediated perturbation of viral transcription / positive regulation of inclusion body assembly / integrator complex / proteasome accessory complex / meiosis I / purine ribonucleoside triphosphate binding / proteasome regulatory particle / cytosolic proteasome complex / positive regulation of proteasomal protein catabolic process / proteasome-activating activity / Antigen processing: Ub, ATP-independent proteasomal degradation / proteasome regulatory particle, lid subcomplex / proteasome regulatory particle, base subcomplex / sperm glycocalyx / protein K63-linked deubiquitination / negative regulation of programmed cell death / Regulation of ornithine decarboxylase (ODC) / metal-dependent deubiquitinase activity / perinuclear theca / Proteasome assembly / proteasome core complex / Cross-presentation of soluble exogenous antigens (endosomes) / Somitogenesis / K63-linked deubiquitinase activity / Homologous DNA Pairing and Strand Exchange / Defective homologous recombination repair (HRR) due to BRCA1 loss of function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA1 binding function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA2/RAD51/RAD51C binding function / Resolution of D-loop Structures through Synthesis-Dependent Strand Annealing (SDSA) / transcription factor binding / Resolution of D-loop Structures through Holliday Junction Intermediates / proteasome binding / Impaired BRCA2 binding to RAD51 / myofibril / regulation of protein catabolic process / proteasome storage granule / proteasomal ubiquitin-independent protein catabolic process / sperm head-tail coupling apparatus / Presynaptic phase of homologous DNA pairing and strand exchange / general transcription initiation factor binding / positive regulation of RNA polymerase II transcription preinitiation complex assembly / protein deubiquitination / blastocyst development / polyubiquitin modification-dependent protein binding / immune system process / NF-kappaB binding / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / endopeptidase activator activity / threonine-type endopeptidase activity / proteasome assembly / mRNA export from nucleus / proteasome core complex, alpha-subunit complex / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / enzyme regulator activity / ERAD pathway / regulation of proteasomal protein catabolic process / inclusion body / ciliary tip / : / proteasome complex / TBP-class protein binding / stem cell differentiation / Regulation of activated PAK-2p34 by proteasome mediated degradation / sarcomere / Autodegradation of Cdh1 by Cdh1:APC/C / APC/C:Cdc20 mediated degradation of Securin / ubiquitin binding / N-glycan trimming in the ER and Calnexin/Calreticulin cycle / Asymmetric localization of PCP proteins / Ubiquitin-dependent degradation of Cyclin D / SCF-beta-TrCP mediated degradation of Emi1 / NIK-->noncanonical NF-kB signaling / sperm end piece / TNFR2 non-canonical NF-kB pathway / AUF1 (hnRNP D0) binds and destabilizes mRNA / centriole / negative regulation of inflammatory response to antigenic stimulus / P-body / Assembly of the pre-replicative complex / Vpu mediated degradation of CD4 / Cdc20:Phospho-APC/C mediated degradation of Cyclin A / lipopolysaccharide binding / Degradation of DVL / Dectin-1 mediated noncanonical NF-kB signaling / Degradation of CRY and PER proteins / Degradation of AXIN / Hh mutants are degraded by ERAD / Activation of NF-kappaB in B cells / G2/M Checkpoints / Hedgehog ligand biogenesis / Degradation of GLI1 by the proteasome / Defective CFTR causes cystic fibrosis / Autodegradation of the E3 ubiquitin ligase COP1 / Regulation of RUNX3 expression and activity / Negative regulation of NOTCH4 signaling Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | ||||||||||||
 Authors Authors | Mao YD | ||||||||||||
| Funding support |  China, China,  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nature / Year: 2019 Journal: Nature / Year: 2019Title: Cryo-EM structures and dynamics of substrate-engaged human 26S proteasome. Authors: Yuanchen Dong / Shuwen Zhang / Zhaolong Wu / Xuemei Li / Wei Li Wang / Yanan Zhu / Svetla Stoilova-McPhie / Ying Lu / Daniel Finley / Youdong Mao /   Abstract: The proteasome is an ATP-dependent, 2.5-megadalton molecular machine that is responsible for selective protein degradation in eukaryotic cells. Here we present cryo-electron microscopy structures of ...The proteasome is an ATP-dependent, 2.5-megadalton molecular machine that is responsible for selective protein degradation in eukaryotic cells. Here we present cryo-electron microscopy structures of the substrate-engaged human proteasome in seven conformational states at 2.8-3.6 Å resolution, captured during breakdown of a polyubiquitylated protein. These structures illuminate a spatiotemporal continuum of dynamic substrate-proteasome interactions from ubiquitin recognition to substrate translocation, during which ATP hydrolysis sequentially navigates through all six ATPases. There are three principal modes of coordinated hydrolysis, featuring hydrolytic events in two oppositely positioned ATPases, in two adjacent ATPases and in one ATPase at a time. These hydrolytic modes regulate deubiquitylation, initiation of translocation and processive unfolding of substrates, respectively. Hydrolysis of ATP powers a hinge-like motion in each ATPase that regulates its substrate interaction. Synchronization of ATP binding, ADP release and ATP hydrolysis in three adjacent ATPases drives rigid-body rotations of substrate-bound ATPases that are propagated unidirectionally in the ATPase ring and unfold the substrate. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9220.map.gz emd_9220.map.gz | 748.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9220-v30.xml emd-9220-v30.xml emd-9220.xml emd-9220.xml | 62.6 KB 62.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9220.png emd_9220.png | 99.6 KB | ||
| Filedesc metadata |  emd-9220.cif.gz emd-9220.cif.gz | 14.6 KB | ||
| Others |  emd_9220_additional_1.map.gz emd_9220_additional_1.map.gz emd_9220_additional_2.map.gz emd_9220_additional_2.map.gz | 751.1 MB 731.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9220 http://ftp.pdbj.org/pub/emdb/structures/EMD-9220 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9220 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9220 | HTTPS FTP |
-Related structure data
| Related structure data |  6mshMC  9215C  9216C  9217C  9218C  9219C  9221C  9222C  9223C  9224C  9225C  9226C  9227C  9228C  9229C  6msbC  6msdC  6mseC  6msgC  6msjC  6mskC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10669 (Title: Cryo-EM dataset of the substrate-engaged human 26S proteasome EMPIAR-10669 (Title: Cryo-EM dataset of the substrate-engaged human 26S proteasomeData size: 13.9 TB Data #1: Drift-corrected frame-averaged super-counting mode micrographs and extracted particles of substrate-engaged human 26S proteasome [micrographs - single frame]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_9220.map.gz / Format: CCP4 / Size: 824 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9220.map.gz / Format: CCP4 / Size: 824 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The complete map of state EC2 of substrate-engaged human proteasome, low pass-filtered to 3 Angstrom without amplitude correction | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
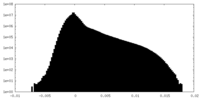
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.685 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
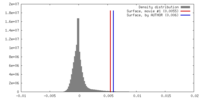
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: The complete map of state EC2 of substrate-engaged...
| File | emd_9220_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The complete map of state EC2 of substrate-engaged human proteasome, low pass-filtered to 3.5 Angstrom with amplitude correction with a B-factor of -30 | ||||||||||||
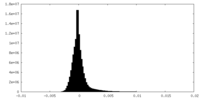
| Projections & Slices |
| ||||||||||||
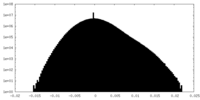
| Density Histograms |
-Additional map: Unfiltered, uncorrected raw EC2 map of complete holoenzyme
| File | emd_9220_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unfiltered, uncorrected raw EC2 map of complete holoenzyme | ||||||||||||
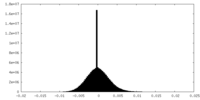
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Proteasome
+Supramolecule #1: Proteasome
+Macromolecule #1: 26S proteasome non-ATPase regulatory subunit 1
+Macromolecule #2: 26S proteasome non-ATPase regulatory subunit 3
+Macromolecule #3: 26S proteasome non-ATPase regulatory subunit 12
+Macromolecule #4: 26S proteasome non-ATPase regulatory subunit 11
+Macromolecule #5: 26S proteasome non-ATPase regulatory subunit 6
+Macromolecule #6: 26S proteasome non-ATPase regulatory subunit 7
+Macromolecule #7: 26S proteasome non-ATPase regulatory subunit 13
+Macromolecule #8: 26S proteasome non-ATPase regulatory subunit 4
+Macromolecule #9: 26S proteasome non-ATPase regulatory subunit 14
+Macromolecule #10: 26S proteasome non-ATPase regulatory subunit 8
+Macromolecule #11: 26S proteasome complex subunit SEM1
+Macromolecule #12: 26S proteasome non-ATPase regulatory subunit 2
+Macromolecule #13: 26S proteasome regulatory subunit 7
+Macromolecule #14: 26S proteasome regulatory subunit 4
+Macromolecule #15: 26S proteasome regulatory subunit 8
+Macromolecule #16: 26S proteasome regulatory subunit 6B
+Macromolecule #17: 26S proteasome regulatory subunit 10B
+Macromolecule #18: 26S proteasome regulatory subunit 6A
+Macromolecule #19: substrate
+Macromolecule #20: Proteasome subunit alpha type-6
+Macromolecule #21: Proteasome subunit alpha type-2
+Macromolecule #22: Proteasome subunit alpha type-4
+Macromolecule #23: Proteasome subunit alpha type-7
+Macromolecule #24: Proteasome subunit alpha type-5
+Macromolecule #25: Proteasome subunit alpha type-1
+Macromolecule #26: Proteasome subunit alpha type-3
+Macromolecule #27: Proteasome subunit beta type-6
+Macromolecule #28: Proteasome subunit beta type-7
+Macromolecule #29: Proteasome subunit beta type-3
+Macromolecule #30: Proteasome subunit beta type-2
+Macromolecule #31: Proteasome subunit beta type-5
+Macromolecule #32: Proteasome subunit beta type-1
+Macromolecule #33: Proteasome subunit beta type-4
+Macromolecule #34: ZINC ION
+Macromolecule #35: ADENOSINE-5'-TRIPHOSPHATE
+Macromolecule #36: ADENOSINE-5'-DIPHOSPHATE
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Details: unspecified |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 44.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.6 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 71651 |
| Initial angle assignment | Type: COMMON LINE |
| Final angle assignment | Type: PROJECTION MATCHING |
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)