[English] 日本語
 Yorodumi
Yorodumi- EMDB-5327: Cryo EM reconstruction of mammalian 80S ribosome in classic PRE s... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-5327 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo EM reconstruction of mammalian 80S ribosome in classic PRE state 2 | |||||||||
 Map data Map data | 80S mammalian ribosome | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | 80S ribosome / mammalian / elongation cycle / tRNA | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 9.0 Å | |||||||||
 Authors Authors | Budkevich T / Giesebrecht J / Altman R / Munro J / Mielke T / Nierhaus K / Blanchard S / Spahn C | |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2011 Journal: Mol Cell / Year: 2011Title: Structure and dynamics of the mammalian ribosomal pretranslocation complex. Authors: Tatyana Budkevich / Jan Giesebrecht / Roger B Altman / James B Munro / Thorsten Mielke / Knud H Nierhaus / Scott C Blanchard / Christian M T Spahn /  Abstract: Although the structural core of the ribosome is conserved in all kingdoms of life, eukaryotic ribosomes are significantly larger and more complex than their bacterial counterparts. The extent to ...Although the structural core of the ribosome is conserved in all kingdoms of life, eukaryotic ribosomes are significantly larger and more complex than their bacterial counterparts. The extent to which these differences influence the molecular mechanism of translation remains elusive. Multiparticle cryo-electron microscopy and single-molecule FRET investigations of the mammalian pretranslocation complex reveal spontaneous, large-scale conformational changes, including an intersubunit rotation of the ribosomal subunits. Through structurally related processes, tRNA substrates oscillate between classical and at least two distinct hybrid configurations facilitated by localized changes in their L-shaped fold. Hybrid states are favored within the mammalian complex. However, classical tRNA positions can be restored by tRNA binding to the E site or by the eukaryotic-specific antibiotic and translocation inhibitor cycloheximide. These findings reveal critical distinctions in the structural and energetic features of bacterial and mammalian ribosomes, providing a mechanistic basis for divergent translation regulation strategies and species-specific antibiotic action. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_5327.map.gz emd_5327.map.gz | 1.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-5327-v30.xml emd-5327-v30.xml emd-5327.xml emd-5327.xml | 10.1 KB 10.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_5327_1.jpg emd_5327_1.jpg | 99.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-5327 http://ftp.pdbj.org/pub/emdb/structures/EMD-5327 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5327 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5327 | HTTPS FTP |
-Related structure data
| Related structure data |  3j0oMC  5326C  5328C  5329C  3j0lC  3j0pC  3j0qC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_5327.map.gz / Format: CCP4 / Size: 21.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_5327.map.gz / Format: CCP4 / Size: 21.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 80S mammalian ribosome | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.52 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
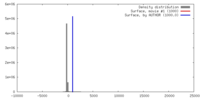
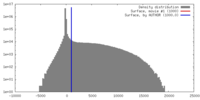
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : 80S-PRE-classic-2 complex from rabbit (Oryctolagus cuniculus) liver
| Entire | Name: 80S-PRE-classic-2 complex from rabbit (Oryctolagus cuniculus) liver |
|---|---|
| Components |
|
-Supramolecule #1000: 80S-PRE-classic-2 complex from rabbit (Oryctolagus cuniculus) liver
| Supramolecule | Name: 80S-PRE-classic-2 complex from rabbit (Oryctolagus cuniculus) liver type: sample / ID: 1000 / Details: Sample was re-associated from subunits / Oligomeric state: monomer / Number unique components: 5 |
|---|---|
| Molecular weight | Experimental: 4 MDa / Method: Sedimentation |
-Supramolecule #1: 80S ribosome
| Supramolecule | Name: 80S ribosome / type: complex / ID: 1 / Recombinant expression: No / Database: NCBI / Ribosome-details: ribosome-eukaryote: LSU 60S, SSU 40S |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 4 MDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Grid | Details: Quantifoil grids |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 77 K / Instrument: OTHER / Details: Vitrification instrument: VitroBot (FEI) Method: Flash-frozen in liquid ethane on carbon coated Quantifoil grids |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Temperature | Average: 77 K |
| Alignment procedure | Legacy - Astigmatism: Objective lens astigmatism was corrected at 200,000 times magnification |
| Date | Oct 17, 2006 |
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: PRIMESCAN / Digitization - Sampling interval: 4.7 µm / Number real images: 87 / Average electron dose: 20 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 65520 / Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Cs: 2 mm / Nominal defocus max: 4.0 µm / Nominal defocus min: 2.0 µm / Nominal magnification: 39000 |
| Sample stage | Specimen holder: Cartridge / Specimen holder model: GATAN HELIUM |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
- Image processing
Image processing
| CTF correction | Details: By defocus groups |
|---|---|
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 9.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: SPIDER Details: Several rounds of multireference 3D projection refinement Number images used: 81946 |
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)