+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | TRRAP module of the human TIP60 complex | |||||||||||||||
 Map data Map data | ||||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | Chromatin Modification / Gene Regulation | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationtranscription factor TFTC complex / Swr1 complex / protein antigen binding / regulation of double-strand break repair / SAGA complex / Formation of Senescence-Associated Heterochromatin Foci (SAHF) / DNA repair-dependent chromatin remodeling / NuA4 histone acetyltransferase complex / regulation of RNA splicing / positive regulation of double-strand break repair via homologous recombination ...transcription factor TFTC complex / Swr1 complex / protein antigen binding / regulation of double-strand break repair / SAGA complex / Formation of Senescence-Associated Heterochromatin Foci (SAHF) / DNA repair-dependent chromatin remodeling / NuA4 histone acetyltransferase complex / regulation of RNA splicing / positive regulation of double-strand break repair via homologous recombination / regulation of DNA repair / transcription coregulator activity / Formation of the beta-catenin:TCF transactivating complex / helicase activity / DNA Damage/Telomere Stress Induced Senescence / Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement / nucleosome / HATs acetylate histones / chromatin organization / regulation of apoptotic process / hydrolase activity / Ub-specific processing proteases / regulation of cell cycle / nuclear speck / DNA repair / chromatin binding / regulation of transcription by RNA polymerase II / regulation of DNA-templated transcription / positive regulation of DNA-templated transcription / Golgi apparatus / DNA binding / nucleoplasm / ATP binding / nucleus Similarity search - Function | |||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||||||||
 Authors Authors | Yang Z / Mameri A / Florez Ariza AJ / Cote J / Nogales E | |||||||||||||||
| Funding support |  United States, United States,  Canada, 4 items Canada, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Science / Year: 2024 Journal: Science / Year: 2024Title: Structural insights into the human NuA4/TIP60 acetyltransferase and chromatin remodeling complex. Authors: Zhenlin Yang / Amel Mameri / Claudia Cattoglio / Catherine Lachance / Alfredo Jose Florez Ariza / Jie Luo / Jonathan Humbert / Deepthi Sudarshan / Arul Banerjea / Maxime Galloy / Amélie ...Authors: Zhenlin Yang / Amel Mameri / Claudia Cattoglio / Catherine Lachance / Alfredo Jose Florez Ariza / Jie Luo / Jonathan Humbert / Deepthi Sudarshan / Arul Banerjea / Maxime Galloy / Amélie Fradet-Turcotte / Jean-Philippe Lambert / Jeff A Ranish / Jacques Côté / Eva Nogales /   Abstract: The human nucleosome acetyltransferase of histone H4 (NuA4)/Tat-interactive protein, 60 kilodalton (TIP60) coactivator complex, a fusion of the yeast switch/sucrose nonfermentable related 1 (SWR1) ...The human nucleosome acetyltransferase of histone H4 (NuA4)/Tat-interactive protein, 60 kilodalton (TIP60) coactivator complex, a fusion of the yeast switch/sucrose nonfermentable related 1 (SWR1) and NuA4 complexes, both incorporates the histone variant H2A.Z into nucleosomes and acetylates histones H4, H2A, and H2A.Z to regulate gene expression and maintain genome stability. Our cryo-electron microscopy studies show that, within the NuA4/TIP60 complex, the E1A binding protein P400 (EP400) subunit serves as a scaffold holding the different functional modules in specific positions, creating a distinct arrangement of the actin-related protein (ARP) module. EP400 interacts with the transformation/transcription domain-associated protein (TRRAP) subunit by using a footprint that overlaps with that of the Spt-Ada-Gcn5 acetyltransferase (SAGA) complex, preventing the formation of a hybrid complex. Loss of the TRRAP subunit leads to mislocalization of NuA4/TIP60, resulting in the redistribution of H2A.Z and its acetylation across the genome, emphasizing the dual functionality of NuA4/TIP60 as a single macromolecular assembly. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_45176.map.gz emd_45176.map.gz | 168.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-45176-v30.xml emd-45176-v30.xml emd-45176.xml emd-45176.xml | 22 KB 22 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_45176.png emd_45176.png | 115.7 KB | ||
| Filedesc metadata |  emd-45176.cif.gz emd-45176.cif.gz | 9.9 KB | ||
| Others |  emd_45176_half_map_1.map.gz emd_45176_half_map_1.map.gz emd_45176_half_map_2.map.gz emd_45176_half_map_2.map.gz | 165.3 MB 165.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-45176 http://ftp.pdbj.org/pub/emdb/structures/EMD-45176 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-45176 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-45176 | HTTPS FTP |
-Validation report
| Summary document |  emd_45176_validation.pdf.gz emd_45176_validation.pdf.gz | 1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_45176_full_validation.pdf.gz emd_45176_full_validation.pdf.gz | 1 MB | Display | |
| Data in XML |  emd_45176_validation.xml.gz emd_45176_validation.xml.gz | 14.9 KB | Display | |
| Data in CIF |  emd_45176_validation.cif.gz emd_45176_validation.cif.gz | 17.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-45176 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-45176 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-45176 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-45176 | HTTPS FTP |
-Related structure data
| Related structure data |  9c47MC  9c4bC  9c57C  9c62C  9c6nC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_45176.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_45176.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.05 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_45176_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
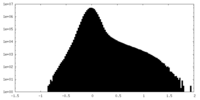
| Projections & Slices |
| ||||||||||||
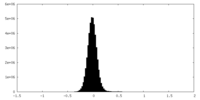
| Density Histograms |
-Half map: #1
| File | emd_45176_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
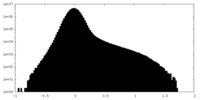
| Projections & Slices |
| ||||||||||||
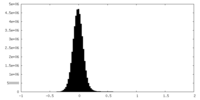
| Density Histograms |
- Sample components
Sample components
-Entire : TRRAP module of human TIP60 complex
| Entire | Name: TRRAP module of human TIP60 complex |
|---|---|
| Components |
|
-Supramolecule #1: TRRAP module of human TIP60 complex
| Supramolecule | Name: TRRAP module of human TIP60 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #2, #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Transformation/transcription domain-associated protein
| Macromolecule | Name: Transformation/transcription domain-associated protein type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 438.139531 KDa |
| Sequence | String: MAFVATQGAT VVDQTTLMKK YLQFVAALTD VNTPDETKLK MMQEVSENFE NVTSSPQYST FLEHIIPRFL TFLQDGEVQF LQEKPAQQL RKLVLEIIHR IPTNEHLRPH TKNVLSVMFR FLETENEENV LICLRIIIEL HKQFRPPITQ EIHHFLDFVK Q IYKELPKV ...String: MAFVATQGAT VVDQTTLMKK YLQFVAALTD VNTPDETKLK MMQEVSENFE NVTSSPQYST FLEHIIPRFL TFLQDGEVQF LQEKPAQQL RKLVLEIIHR IPTNEHLRPH TKNVLSVMFR FLETENEENV LICLRIIIEL HKQFRPPITQ EIHHFLDFVK Q IYKELPKV VNRYFENPQV IPENTVPPPE MVGMITTIAV KVNPEREDSE TRTHSIIPRG SLSLKVLAEL PIIVVLMYQL YK LNIHNVV AEFVPLIMNT IAIQVSAQAR QHKLYNKELY ADFIAAQIKT LSFLAYIIRI YQELVTKYSQ QMVKGMLQLL SNC PAETAH LRKELLIAAK HILTTELRNQ FIPCMDKLFD ESILIGSGYT ARETLRPLAY STLADLVHHV RQHLPLSDLS LAVQ LFAKN IDDESLPSSI QTMSCKLLLN LVDCIRSKSE QESGNGRDVL MRMLEVFVLK FHTIARYQLS AIFKKCKPQS ELGAV EAAL PGVPTAPAAP GPAPSPAPVP APPPPPPPPP PATPVTPAPV PPFEKQGEKD KEDKQTFQVT DCRSLVKTLV CGVKTI TWG ITSCKAPGEA QFIPNKQLQP KETQIYIKLV KYAMQALDIY QVQIAGNGQT YIRVANCQTV RMKEEKEVLE HFAGVFT MM NPLTFKEIFQ TTVPYMVERI SKNYALQIVA NSFLANPTTS ALFATILVEY LLDRLPEMGS NVELSNLYLK LFKLVFGS V SLFAAENEQM LKPHLHKIVN SSMELAQTAK EPYNYFLLLR ALFRSIGGGS HDLLYQEFLP LLPNLLQGLN MLQSGLHKQ HMKDLFVELC LTVPVRLSSL LPYLPMLMDP LVSALNGSQT LVSQGLRTLE LCVDNLQPDF LYDHIQPVRA ELMQALWRTL RNPADSISH VAYRVLGKFG GSNRKMLKES QKLHYVVTEV QGPSITVEFS DCKASLQLPM EKAIETALDC LKSANTEPYY R RQAWEVIK CFLVAMMSLE DNKHALYQLL AHPNFTEKTI PNVIISHRYK AQDTPARKTF EQALTGAFMS AVIKDLRPSA LP FVASLIR HYTMVAVAQQ CGPFLLPCYQ VGSQPSTAMF HSEENGSKGM DPLVLIDAIA ICMAYEEKEL CKIGEVALAV IFD VASIIL GSKERACQLP LFSYIVERLC ACCYEQAWYA KLGGVVSIKF LMERLPLTWV LQNQQTFLKA LLFVMMDLTG EVSN GAVAM AKTTLEQLLM RCATPLKDEE RAEEIVAAQE KSFHHVTHDL VREVTSPNST VRKQAMHSLQ VLAQVTGKSV TVIME PHKE VLQDMVPPKK HLLRHQPANA QIGLMEGNTF CTTLQPRLFT MDLNVVEHKV FYTELLNLCE AEDSALTKLP CYKSLP SLV PLRIAALNAL AACNYLPQSR EKIIAALFKA LNSTNSELQE AGEACMRKFL EGATIEVDQI HTHMRPLLMM LGDYRSL TL NVVNRLTSVT RLFPNSFNDK FCDQMMQHLR KWMEVVVITH KGGQRSDGNE SISECGRCPL SPFCQFEEMK ICSAIINL F HLIPAAPQTL VKPLLEVVMK TERAMLIEAG SPFREPLIKF LTRHPSQTVE LFMMEATLND PQWSRMFMSF LKHKDARPL RDVLAANPNR FITLLLPGGA QTAVRPGSPS TSTMRLDLQF QAIKIISIIV KNDDSWLASQ HSLVSQLRRV WVSENFQERH RKENMAATN WKEPKLLAYC LLNYCKRNYG DIELLFQLLR AFTGRFLCNM TFLKEYMEEE IPKNYSIAQK RALFFRFVDF N DPNFGDEL KAKVLQHILN PAFLYSFEKG EGEQLLGPPN PEGDNPESIT SVFITKVLDP EKQADMLDSL RIYLLQYATL LV EHAPHHI HDNNKNRNSK LRRLMTFAWP CLLSKACVDP ACKYSGHLLL AHIIAKFAIH KKIVLQVFHS LLKAHAMEAR AIV RQAMAI LTPAVPARME DGHQMLTHWT RKIIVEEGHT VPQLVHILHL IVQHFKVYYP VRHHLVQHMV SAMQRLGFTP SVTI EQRRL AVDLSEVVIK WELQRIKDQQ PDSDMDPNSS GEGVNSVSSS IKRGLSVDSA QEVKRFRTAT GAISAVFGRS QSLPG ADSL LAKPIDKQHT DTVVNFLIRV ACQVNDNTNT AGSPGEVLSR RCVNLLKTAL RPDMWPKSEL KLQWFDKLLM TVEQPN QVN YGNICTGLEV LSFLLTVLQS PAILSSFKPL QRGIAACMTC GNTKVLRAVH SLLSRLMSIF PTEPSTSSVA SKYEELE CL YAAVGKVIYE GLTNYEKATN ANPSQLFGTL MILKSACSNN PSYIDRLISV FMRSLQKMVR EHLNPQAASG STEATSGT S ELVMLSLELV KTRLAVMSME MRKNFIQAIL TSLIEKSPDA KILRAVVKIV EEWVKNNSPM AANQTPTLRE KSILLVKMM TYIEKRFPED LELNAQFLDL VNYVYRDETL SGSELTAKLE PAFLSGLRCA QPLIRAKFFE VFDNSMKRRV YERLLYVTCS QNWEAMGNH FWIKQCIELL LAVCEKSTPI GTSCQGAMLP SITNVINLAD SHDRAAFAMV THVKQEPRER ENSESKEEDV E IDIELAPG DQTSTPKTKE LSEKDIGNQL HMLTNRHDKF LDTLREVKTG ALLSAFVQLC HISTTLAEKT WVQLFPRLWK IL SDRQQHA LAGEISPFLC SGSHQVQRDC QPSALNCFVE AMSQCVPPIP IRPCVLKYLG KTHNLWFRST LMLEHQAFEK GLS LQIKPK QTTEFYEQES ITPPQQEILD SLAELYSLLQ EEDMWAGLWQ KRCKYSETAT AIAYEQHGFF EQAQESYEKA MDKA KKEHE RSNASPAIFP EYQLWEDHWI RCSKELNQWE ALTEYGQSKG HINPYLVLEC AWRVSNWTAM KEALVQVEVS CPKEM AWKV NMYRGYLAIC HPEEQQLSFI ERLVEMASSL AIREWRRLPH VVSHVHTPLL QAAQQIIELQ EAAQINAGLQ PTNLGR NNS LHDMKTVVKT WRNRLPIVSD DLSHWSSIFM WRQHHYQGKP TWSGMHSSSI VTAYENSSQH DPSSNNAMLG VHASASA II QYGKIARKQG LVNVALDILS RIHTIPTVPI VDCFQKIRQQ VKCYLQLAGV MGKNECMQGL EVIESTNLKY FTKEMTAE F YALKGMFLAQ INKSEEANKA FSAAVQMHDV LVKAWAMWGD YLENIFVKER QLHLGVSAIT CYLHACRHQN ESKSRKYLA KVLWLLSFDD DKNTLADAVD KYCIGVPPIQ WLAWIPQLLT CLVGSEGKLL LNLISQVGRV YPQAVYFPIR TLYLTLKIEQ RERYKSDPG PIRATAPMWR CSRIMHMQRE LHPTLLSSLE GIVDQMVWFR ENWHEEVLRQ LQQGLAKCYS VAFEKSGAVS D AKITPHTL NFVKKLVSTF GVGLENVSNV STMFSSAASE SLARRAQATA QDPVFQKLKG QFTTDFDFSV PGSMKLHNLI SK LKKWIKI LEAKTKQLPK FFLIEEKCRF LSNFSAQTAE VEIPGEFLMP KPTHYYIKIA RFMPRVEIVQ KHNTAARRLY IRG HNGKIY PYLVMNDACL TESRREERVL QLLRLLNPCL EKRKETTKRH LFFTVPRVVA VSPQMRLVED NPSSLSLVEI YKQR CAKKG IEHDNPISRY YDRLATVQAR GTQASHQVLR DILKEVQSNM VPRSMLKEWA LHTFPNATDY WTFRKMFTIQ LALIG FAEF VLHLNRLNPE MLQIAQDTGK LNVAYFRFDI NDATGDLDAN RPVPFRLTPN ISEFLTTIGV SGPLTASMIA VARCFA QPN FKVDGILKTV LRDEIIAWHK KTQEDTSSPL SAAGQPENMD SQQLVSLVQK AVTAIMTRLH NLAQFEGGES KVNTLVA AA NSLDNLCRMD PAWHPWL UniProtKB: Transformation/transcription domain-associated protein |
-Macromolecule #2: E1A-binding protein p400
| Macromolecule | Name: E1A-binding protein p400 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO EC number: Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 343.867312 KDa |
| Sequence | String: MHHGTGPQNV QHQLQRSRAC PGSEGEEQPA HPNPPPSPAA PFAPSASPSA PQSPSYQIQQ LMNRSPATGQ NVNITLQSVG PVVGGNQQI TLAPLPLPSP TSPGFQFSAQ PRRFEHGSPS YIQVTSPLSQ QVQTQSPTQP SPGPGQALQN VRAGAPGPGL G LCSSSPTG ...String: MHHGTGPQNV QHQLQRSRAC PGSEGEEQPA HPNPPPSPAA PFAPSASPSA PQSPSYQIQQ LMNRSPATGQ NVNITLQSVG PVVGGNQQI TLAPLPLPSP TSPGFQFSAQ PRRFEHGSPS YIQVTSPLSQ QVQTQSPTQP SPGPGQALQN VRAGAPGPGL G LCSSSPTG GFVDASVLVR QISLSPSSGG HFVFQDGSGL TQIAQGAQVQ LQHPGTPITV RERRPSQPHT QSGGTIHHLG PQ SPAAAGG AGLQPLASPS HITTANLPPQ ISSIIQGQLV QQQQVLQGPP LPRPLGFERT PGVLLPGAGG AAGFGMTSPP PPT SPSRTA VPPGLSSLPL TSVGNTGMKK VPKKLEEIPP ASPEMAQMRK QCLDYHYQEM QALKEVFKEY LIELFFLQHF QGNM MDFLA FKKKHYAPLQ AYLRQNDLDI EEEEEEEEEE EEKSEVINDE VKVVTGKDGQ TGTPVAIATQ LPPKVSAAFS SQQQP FQQA LAGSLVAGAG STVETDLFKR QQAMPSTGMA EQSKRPRLEV GHQGVVFQHP GADAGVPLQQ LMPTAQGGMP PTPQAA QLA GQRQSQQQYD PSTGPPVQNA ASLHTPLPQL PGRLPPAGVP TAALSSALQF AQQPQVVEAQ TQLQIPVKTQ QPNVPIP AP PSSQLPIPPS QPAQLALHVP TPGKVQVQAS QLSSLPQMVA STRLPVDPAP PCPRPLPTSS TSSLAPVSGS GPGPSPAR S SPVNRPSSAT NKALSPVTSR TPGVVASAPT KPQSPAQNAT SSQDSSQDTL TEQITLENQV HQRIAELRKA GLWSQRRLP KLQEAPRPKS HWDYLLEEMQ WMATDFAQER RWKVAAAKKL VRTVVRHHEE KQLREERGKK EEQSRLRRIA ASTAREIECF WSNIEQVVE IKLRVELEEK RKKALNLQKV SRRGKELRPK GFDALQESSL DSGMSGRKRK ASISLTDDEV DDEEETIEEE E ANEGVVDH QTELSNLAKE AELPLLDLMK LYEGAFLPSS QWPRPKPDGE DTSGEEDADD CPGDRESRKD LVLIDSLFIM DQ FKAAERM NIGKPNAKDI ADVTAVAEAI LPKGSARVTT SVKFNAPSLL YGALRDYQKI GLDWLAKLYR KNLNGILADE AGL GKTVQI IAFFAHLACN EGNWGPHLVV VRSCNILKWE LELKRWCPGL KILSYIGSHR ELKAKRQEWA EPNSFHVCIT SYTQ FFRGL TAFTRVRWKC LVIDEMQRVK GMTERHWEAV FTLQSQQRLL LIDSPLHNTF LELWTMVHFL VPGISRPYLS SPLRA PSEE SQDYYHKVVI RLHRVTQPFI LRRTKRDVEK QLTKKYEHVL KCRLSNRQKA LYEDVILQPG TQEALKSGHF VNVLSI LVR LQRICNHPGL VEPRHPGSSY VAGPLEYPSA SLILKALERD FWKEADLSMF DLIGLENKIT RHEAELLSKK KIPRKLM EE ISTSAAPAAR PAAAKLKASR LFQPVQYGQK PEGRTVAFPS THPPRTAAPT TASAAPQGPL RGRPPIATFS ANPEAKAA A APFQTSQASA SAPRHQPASA SSTAASPAHP AKLRAQTTAQ ASTPGQPPPQ PQAPSHAAGQ SALPQRLVLP SQAQARLPS GEVVKIAQLA SITGPQSRVA QPETPVTLQF QGSKFTLSHS QLRQLTAGQP LQLQGSVLQI VSAPGQPYLR APGPVVMQTV SQAGAVHGA LGSKPPAGGP SPAPLTPQVG VPGRVAVNAL AVGEPGTASK PASPIGGPTQ EEKTRLLKER LDQIYLVNER R CSQAPVYG RDLLRICALP SHGRVQWRGS LDGRRGKEAG PAHSYTSSSE SPSELMLTLC RCGESLQDVI DRVAFVIPPV VA APPSLRV PRPPPLYSHR MRILRQGLRE HAAPYFQQLR QTTAPRLLQF PELRLVQFDS GKLEALAILL QKLKSEGRRV LIL SQMILM LDILEMFLNF HYLTYVRIDE NASSEQRQEL MRSFNRDRRI FCAILSTHSR TTGINLVEAD TVVFYDNDLN PVMD AKAQE WCDRIGRCKD IHIYRLVSGN SIEEKLLKNG TKDLIREVAA QGNDYSMAFL TQRTIQELFE VYSPMDDAGF PVKAE EFVV LSQEPSVTET IAPKIARPFI EALKSIEYLE EDAQKSAQEG VLGPHTDALS SDSENMPCDE EPSQLEELAD FMEQLT PIE KYALNYLELF HTSIEQEKER NSEDAVMTAV RAWEFWNLKT LQEREARLRL EQEEAELLTY TREDAYSMEY VYEDVDG QT EVMPLWTPPT PPQDDSDIYL DSVMCLMYEA TPIPEAKLPP VYVRKERKRH KTDPSAAGRK KKQRHGEAVV PPRSLFDR A TPGLLKIRRE GKEQKKNILL KQQVPFAKPL PTFAKPTAEP GQDNPEWLIS EDWALLQAVK QLLELPLNLT IVSPAHTPN WDLVSDVVNS CSRIYRSSKQ CRNRYENVII PREEGKSKNN RPLRTSQIYA QDENATHTQL YTSHFDLMKM TAGKRSPPIK PLLGMNPFQ KNPKHASVLA ESGINYDKPL PPIQVASLRA ERIAKEKKAL ADQQKAQQPA VAQPPPPQPQ PPPPPQQPPP P LPQPQAAG SQPPAGPPAV QPQPQPQPQT QPQPVQAPAK AQPAITTGGS AAVLAGTIKT SVTGTSMPTG AVSGNVIVNT IA GVPAATF QSINKRLASP VAPGALTTPG GSAPAQVVHT QPPPRAVGSP ATATPDLVSM ATTQGVRAVT SVTASAVVTT NLT PVQTPA RSLVPQVSQA TGVQLPGKTI TPAHFQLLRQ QQQQQQQQQQ QQQQQQQQQQ QQQQQQQQTT TTSQVQVPQI QGQA QSPAQ IKAVGKLTPE HLIKMQKQKL QMPPQPPPPQ AQSAPPQPTA QVQVQTSQPP QQQSPQLTTV TAPRPGALLT GTTVA NLQV ARLTRVPTSQ LQAQGQMQTQ APQPAQVALA KPPVVSVPAA VVSSPGVTTL PMNVAGISVA IGQPQKAAGQ TVVAQP VHM QQLLKLKQQA VQQQKAIQPQ AAQGPAAVQQ KITAQQITTP GAQQKVAYAA QPALKTQFLT TPISQAQKLA GAQQVQT QI QVAKLPQVVQ QQTPVASIQQ VASASQQASP QTVALTQATA AGQQVQMIPA VTATAQVVQQ KLIQQQVVTT ASAPLQTP G APNPAQVPAS SDSPSQQPKL QMRVPAVRLK TPTKPPCQ UniProtKB: E1A-binding protein p400 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: 4D-STEM / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.4 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 23442 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)