[English] 日本語
 Yorodumi
Yorodumi- EMDB-43279: Cryo-EM structure of short form insulin receptor (IR-A) with four... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. | |||||||||
 Map data Map data | Cryo-EM structure of short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Insulin receptor / IGF2 / RTK / SIGNALING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationembryonic placenta morphogenesis / negative regulation of muscle cell differentiation / positive regulation of skeletal muscle tissue growth / Signaling by Type 1 Insulin-like Growth Factor 1 Receptor (IGF1R) / regulation of muscle cell differentiation / IRS-related events triggered by IGF1R / regulation of female gonad development / positive regulation of meiotic cell cycle / positive regulation of organ growth / insulin-like growth factor II binding ...embryonic placenta morphogenesis / negative regulation of muscle cell differentiation / positive regulation of skeletal muscle tissue growth / Signaling by Type 1 Insulin-like Growth Factor 1 Receptor (IGF1R) / regulation of muscle cell differentiation / IRS-related events triggered by IGF1R / regulation of female gonad development / positive regulation of meiotic cell cycle / positive regulation of organ growth / insulin-like growth factor II binding / positive regulation of developmental growth / transmembrane receptor protein tyrosine kinase activator activity / genomic imprinting / male sex determination / insulin receptor complex / insulin-like growth factor I binding / insulin receptor activity / positive regulation of protein-containing complex disassembly / exocrine pancreas development / positive regulation of multicellular organism growth / positive regulation of activated T cell proliferation / positive regulation of vascular endothelial cell proliferation / dendritic spine maintenance / insulin binding / adrenal gland development / cargo receptor activity / PTB domain binding / Signaling by Insulin receptor / IRS activation / neuronal cell body membrane / positive regulation of respiratory burst / amyloid-beta clearance / positive regulation of cell division / regulation of embryonic development / insulin receptor substrate binding / positive regulation of receptor internalization / epidermis development / positive regulation of glycogen biosynthetic process / positive regulation of insulin receptor signaling pathway / Signal attenuation / protein kinase activator activity / embryonic placenta development / SHC-related events triggered by IGF1R / transport across blood-brain barrier / heart morphogenesis / phosphatidylinositol 3-kinase binding / Insulin receptor recycling / insulin-like growth factor receptor binding / striated muscle cell differentiation / neuron projection maintenance / positive regulation of mitotic nuclear division / insulin-like growth factor receptor signaling pathway / Insulin receptor signalling cascade / platelet alpha granule lumen / receptor-mediated endocytosis / dendrite membrane / positive regulation of glycolytic process / animal organ morphogenesis / positive regulation of D-glucose import across plasma membrane / insulin receptor binding / protein serine/threonine kinase activator activity / learning / growth factor activity / hormone activity / receptor protein-tyrosine kinase / caveola / receptor internalization / male gonad development / cellular response to growth factor stimulus / integrin binding / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / glucose metabolic process / cellular response to insulin stimulus / memory / positive regulation of nitric oxide biosynthetic process / osteoblast differentiation / insulin receptor signaling pathway / protein autophosphorylation / late endosome / Platelet degranulation / glucose homeostasis / amyloid-beta binding / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / protein tyrosine kinase activity / in utero embryonic development / positive regulation of canonical NF-kappaB signal transduction / positive regulation of MAPK cascade / lysosome / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / signaling receptor complex / endosome membrane / positive regulation of cell migration / G protein-coupled receptor signaling pathway / receptor ligand activity / external side of plasma membrane / protein domain specific binding / axon / positive regulation of cell population proliferation / symbiont entry into host cell / regulation of DNA-templated transcription Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | An W / Hall C / Li J / Huang A / Wu J / Park J / Bai XC / Choi E | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: Activation of the insulin receptor by insulin-like growth factor 2. Authors: Weidong An / Catherine Hall / Jie Li / Albert Hung / Jiayi Wu / Junhee Park / Liwei Wang / Xiao-Chen Bai / Eunhee Choi /  Abstract: Insulin receptor (IR) controls growth and metabolism. Insulin-like growth factor 2 (IGF2) has different binding properties on two IR isoforms, mimicking insulin's function. However, the molecular ...Insulin receptor (IR) controls growth and metabolism. Insulin-like growth factor 2 (IGF2) has different binding properties on two IR isoforms, mimicking insulin's function. However, the molecular mechanism underlying IGF2-induced IR activation remains unclear. Here, we present cryo-EM structures of full-length human long isoform IR (IR-B) in both the inactive and IGF2-bound active states, and short isoform IR (IR-A) in the IGF2-bound active state. Under saturated IGF2 concentrations, both the IR-A and IR-B adopt predominantly asymmetric conformations with two or three IGF2s bound at site-1 and site-2, which differs from that insulin saturated IR forms an exclusively T-shaped symmetric conformation. IGF2 exhibits a relatively weak binding to IR site-2 compared to insulin, making it less potent in promoting full IR activation. Cell-based experiments validated the functional importance of IGF2 binding to two distinct binding sites in optimal IR signaling and trafficking. In the inactive state, the C-terminus of α-CT of IR-B contacts FnIII-2 domain of the same protomer, hindering its threading into the C-loop of IGF2, thus reducing the association rate of IGF2 with IR-B. Collectively, our studies demonstrate the activation mechanism of IR by IGF2 and reveal the molecular basis underlying the different affinity of IGF2 to IR-A and IR-B. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_43279.map.gz emd_43279.map.gz | 45 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-43279-v30.xml emd-43279-v30.xml emd-43279.xml emd-43279.xml | 19.1 KB 19.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_43279.png emd_43279.png | 36.8 KB | ||
| Filedesc metadata |  emd-43279.cif.gz emd-43279.cif.gz | 6.6 KB | ||
| Others |  emd_43279_half_map_1.map.gz emd_43279_half_map_1.map.gz emd_43279_half_map_2.map.gz emd_43279_half_map_2.map.gz | 57.5 MB 57.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-43279 http://ftp.pdbj.org/pub/emdb/structures/EMD-43279 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43279 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43279 | HTTPS FTP |
-Related structure data
| Related structure data |  8vjbMC  8u4bC  8u4cC  8u4eC  8vjcC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_43279.map.gz / Format: CCP4 / Size: 75.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_43279.map.gz / Format: CCP4 / Size: 75.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||
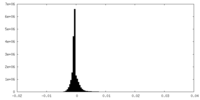
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Cryo-EM structure of short form insulin receptor (IR-A)...
| File | emd_43279_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. Half map 1. | ||||||||||||
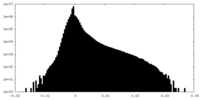
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Cryo-EM structure of short form insulin receptor (IR-A)...
| File | emd_43279_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. Half map 2. | ||||||||||||
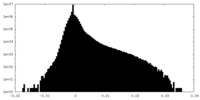
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Short form insulin receptor (IR-A) with four IGF2 bound, symmetri...
| Entire | Name: Short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. |
|---|---|
| Components |
|
-Supramolecule #1: Short form insulin receptor (IR-A) with four IGF2 bound, symmetri...
| Supramolecule | Name: Short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Isoform Short of Insulin receptor
| Macromolecule | Name: Isoform Short of Insulin receptor / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO / EC number: receptor protein-tyrosine kinase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 155.329094 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MATGGRRGAA AAPLLVAVAA LLLGAAGHLY PGEVCPGMDI RNNLTRLHEL ENCSVIEGHL QILLMFKTRP EDFRDLSFPK LIMITDYLL LFRVYGLESL KDLFPNLTVI RGSRLFFNYA LVIFEMVHLK ELGLYNLMNI TRGSVRIEKN NELCYLATID W SRILDSVE ...String: MATGGRRGAA AAPLLVAVAA LLLGAAGHLY PGEVCPGMDI RNNLTRLHEL ENCSVIEGHL QILLMFKTRP EDFRDLSFPK LIMITDYLL LFRVYGLESL KDLFPNLTVI RGSRLFFNYA LVIFEMVHLK ELGLYNLMNI TRGSVRIEKN NELCYLATID W SRILDSVE DNYIVLNKDD NEECGDICPG TAKGKTNCPA TVINGQFVER CWTHSHCQKV CPTICKSHGC TAEGLCCHSE CL GNCSQPD DPTKCVACRN FYLDGRCVET CPPPYYHFQD WRCVNFSFCQ DLHHKCKNSR RQGCHQYVIH NNKCIPECPS GYT MNSSNL LCTPCLGPCP KVCHLLEGEK TIDSVTSAQE LRGCTVINGS LIINIRGGNN LAAELEANLG LIEEISGYLK IRRS YALVS LSFFRKLRLI RGETLEIGNY SFYALDNQNL RQLWDWSKHN LTITQGKLFF HYNPKLCLSE IHKMEEVSGT KGRQE RNDI ALKTNGDQAS CENELLKFSY IRTSFDKILL RWEPYWPPDF RDLLGFMLFY KEAPYQNVTE FDGQDACGSN SWTVVD IDP PLRSNDPKSQ NHPGWLMRGL KPWTQYAIFV KTLVTFSDER RTYGAKSDII YVQTDATNPS VPLDPISVSN SSSQIIL KW KPPSDPNGNI THYLVFWERQ AEDSELFELD YCLKGLKLPS RTWSPPFESE DSQKHNQSEY EDSAGECCSC PKTDSQIL K ELEESSFRKT FEDYLHNVVF VPRPSRKRRS LGDVGNVTVA VPTVAAFPNT SSTSVPTSPE EHRPFEKVVN KESLVISGL RHFTGYRIEL QACNQDTPEE RCSVAAYVSA RTMPEAKADD IVGPVTHEIF ENNVVHLMWQ EPKEPNGLIV LYEVSYRRYG DEELHLCVS RKHFALERGC RLRGLSPGNY SVRIRATSLA GNGSWTEPTY FYVTDYLDVP SNIAKIIIGP LIFVFLFSVV I GSIYLFLR KRQPDGPLGP LYASSNPEYL SASDVFPCSV YVPDEWEVSR EKITLLRELG QGSFGMVYEG NARDIIKGEA ET RVAVKTV NESASLRERI EFLNEASVMK GFTCHHVVRL LGVVSKGQPT LVVMELMAHG DLKSYLRSLR PEAENNPGRP PPT LQEMIQ MAAEIADGMA YLNAKKFVHR DLAARNCMVA HDFTVKIGDF GMTRDIYETD YYRKGGKGLL PVRWMAPESL KDGV FTTSS DMWSFGVVLW EITSLAEQPY QGLSNEQVLK FVMDGGYLDQ PDNCPERVTD LMRMCWQFNP KMRPTFLEIV NLLKD DLHP SFPEVSFFHS EENKAPESEE LEMEFEDMEN VPLDRSSHCQ REEAGGRDGG SSLGFKRSYE EHIPYTHMNG GKKNGR ILT LPRSNPS UniProtKB: Insulin receptor |
-Macromolecule #2: Insulin-like growth factor II
| Macromolecule | Name: Insulin-like growth factor II / type: protein_or_peptide / ID: 2 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 20.170398 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGIPMGKSML VLLTFLAFAS CCIAAYRPSE TLCGGELVDT LQFVCGDRGF YFSRPASRVS RRSRGIVEEC CFRSCDLALL ETYCATPAK SERDVSTPPT VLPDNFPRYP VGKFFQYDTW KQSTQRLRRG LPALLRARRG HVLAKELEAF REAKRHRPLI A LPTQDPAH GGAPPEMASN RK UniProtKB: Insulin-like growth factor 2 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.6 µm / Nominal defocus min: 1.6 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
|---|---|
| Output model |  PDB-8vjb: |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)