[English] 日本語
 Yorodumi
Yorodumi- EMDB-42054: Structure of Semliki Forest virus VLP in complex with VLDLR LA2 -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of Semliki Forest virus VLP in complex with VLDLR LA2 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Semliki Forest Virus / Receptor / VIRUS LIKE PARTICLE | |||||||||
| Function / homology |  Function and homology information Function and homology informationreelin receptor activity / VLDL clearance / glycoprotein transport / ventral spinal cord development / very-low-density lipoprotein particle receptor activity / Reelin signalling pathway / very-low-density lipoprotein particle binding / reelin-mediated signaling pathway / low-density lipoprotein particle receptor activity / very-low-density lipoprotein particle clearance ...reelin receptor activity / VLDL clearance / glycoprotein transport / ventral spinal cord development / very-low-density lipoprotein particle receptor activity / Reelin signalling pathway / very-low-density lipoprotein particle binding / reelin-mediated signaling pathway / low-density lipoprotein particle receptor activity / very-low-density lipoprotein particle clearance / togavirin / very-low-density lipoprotein particle / positive regulation of dendrite development / T=4 icosahedral viral capsid / dendrite morphogenesis / cargo receptor activity / lipid transport / virion assembly / small molecule binding / apolipoprotein binding / clathrin-coated pit / VLDLR internalisation and degradation / cholesterol metabolic process / receptor-mediated endocytosis / memory / calcium-dependent protein binding / nervous system development / host cell endosome / symbiont-mediated suppression of host toll-like receptor signaling pathway / clathrin-dependent endocytosis of virus by host cell / receptor complex / host cell cytoplasm / symbiont entry into host cell / serine-type endopeptidase activity / lysosomal membrane / fusion of virus membrane with host endosome membrane / viral envelope / calcium ion binding / host cell nucleus / virion attachment to host cell / host cell plasma membrane / structural molecule activity / virion membrane / signal transduction / proteolysis / RNA binding / membrane / plasma membrane / cytoplasm Similarity search - Function | |||||||||
| Biological species |   Semliki Forest virus / Semliki Forest virus /  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.7 Å | |||||||||
 Authors Authors | Abraham J / Yang P / Li W / Fan X / Pan J | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Cryo-EM structure of SFV VLP-VLDLRAD2 complex at the 3-fold axes Authors: Abraham J / Yang P / Li W / Fan X / Pan J | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42054.map.gz emd_42054.map.gz | 272.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42054-v30.xml emd-42054-v30.xml emd-42054.xml emd-42054.xml | 20.5 KB 20.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_42054.png emd_42054.png | 61.9 KB | ||
| Filedesc metadata |  emd-42054.cif.gz emd-42054.cif.gz | 6.3 KB | ||
| Others |  emd_42054_additional_1.map.gz emd_42054_additional_1.map.gz emd_42054_half_map_1.map.gz emd_42054_half_map_1.map.gz emd_42054_half_map_2.map.gz emd_42054_half_map_2.map.gz | 275.7 MB 241.6 MB 241.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42054 http://ftp.pdbj.org/pub/emdb/structures/EMD-42054 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42054 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42054 | HTTPS FTP |
-Validation report
| Summary document |  emd_42054_validation.pdf.gz emd_42054_validation.pdf.gz | 1.1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_42054_full_validation.pdf.gz emd_42054_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  emd_42054_validation.xml.gz emd_42054_validation.xml.gz | 17 KB | Display | |
| Data in CIF |  emd_42054_validation.cif.gz emd_42054_validation.cif.gz | 20.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42054 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42054 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42054 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-42054 | HTTPS FTP |
-Related structure data
| Related structure data |  8ua8MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_42054.map.gz / Format: CCP4 / Size: 303.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42054.map.gz / Format: CCP4 / Size: 303.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.825 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_42054_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
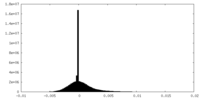
| Density Histograms |
-Half map: #2
| File | emd_42054_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
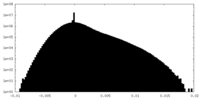
| Density Histograms |
-Half map: #1
| File | emd_42054_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Semliki Forest virus
| Entire | Name:   Semliki Forest virus Semliki Forest virus |
|---|---|
| Components |
|
-Supramolecule #1: Semliki Forest virus
| Supramolecule | Name: Semliki Forest virus / type: virus / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 / NCBI-ID: 11033 / Sci species name: Semliki Forest virus / Virus type: VIRUS-LIKE PARTICLE / Virus isolate: STRAIN / Virus enveloped: Yes / Virus empty: No |
|---|
-Macromolecule #1: Glycoprotein E1
| Macromolecule | Name: Glycoprotein E1 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Semliki Forest virus Semliki Forest virus |
| Molecular weight | Theoretical: 47.489766 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: YEHSTVMPNV VGFPYKAHIE RPGYSPLTLQ MQVVETSLEP TLNLEYITCE YKTVVPSPYV KCCGASECST KEKPDYQCKV YTGVYPFMW GGAYCFCDSE NTQLSEAYVD RSDVCRHDHA SAYKAHTASL KAKVRVMYGN VNQTVDVYVN GDHAVTIGGT Q FIFGPLSS ...String: YEHSTVMPNV VGFPYKAHIE RPGYSPLTLQ MQVVETSLEP TLNLEYITCE YKTVVPSPYV KCCGASECST KEKPDYQCKV YTGVYPFMW GGAYCFCDSE NTQLSEAYVD RSDVCRHDHA SAYKAHTASL KAKVRVMYGN VNQTVDVYVN GDHAVTIGGT Q FIFGPLSS AWTPFDNKIV VYKDEVFNQD FPPYGSGQPG RFGDIQSRTV ESNDLYANTA LKLARPSPGM VHVPYTQTPS GF KYWLKEK GTALNTKAPF GCQIKTNPVR AMNCAVGNIP VSMNLPDSAF TRIVEAPTII DLTCTVATCT HSSDFGGVLT LTY KTDKNG DCSVHSHSNV ATLQEATAKV KTAGKVTLHF STASASPSFV VSLCSARATC SASCEPPKDH IVPYAASHSN VVFP DMSGT ALSWVQKISG GLGAFAIGAI LVLVVVTCIG LRR UniProtKB: Structural polyprotein |
-Macromolecule #2: Glycoprotein E2
| Macromolecule | Name: Glycoprotein E2 / type: protein_or_peptide / ID: 2 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Semliki Forest virus Semliki Forest virus |
| Molecular weight | Theoretical: 46.330719 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: FNVYKATRPY IAYCADCGAG HSCHSPVAIE AVRSEATDGM LKIQFSAQIG IDKSDNHDYT KIRYADGHAI ENAVRSSLKV ATSGDCFVH GTMGHFILAK CPPGEFLQVS IQDTRNAVRA CRIQYHHDPQ PVGREKFTIR PHYGKEIPCT TYQQTTAKTV E EIDMHMPP ...String: FNVYKATRPY IAYCADCGAG HSCHSPVAIE AVRSEATDGM LKIQFSAQIG IDKSDNHDYT KIRYADGHAI ENAVRSSLKV ATSGDCFVH GTMGHFILAK CPPGEFLQVS IQDTRNAVRA CRIQYHHDPQ PVGREKFTIR PHYGKEIPCT TYQQTTAKTV E EIDMHMPP DTPDRTLLSQ QSGNVKITVG GKKVKYNCTC GTGNVGTTNS DMTINTCLIE QCHVSVTDHK KWQFNSPFVP RA DEPARKG KVHIPFPLDN ITCRVPMARE PTVIHGKREV TLHLHPDHPT LFSYRTLGED PQYHEEWVTA AVERTIPVPV DGM EYHWGN NDPVRLWSQL TTEGKPHGWP HQIVQYYYGL YPAATVSAVV GMSLLALISI FASCYMLVAA RSKCLTPYAL TPGA AVPWT LGILCCAPRA HA UniProtKB: UNIPROTKB: A0A0E3T652 |
-Macromolecule #3: Assembly protein E3
| Macromolecule | Name: Assembly protein E3 / type: protein_or_peptide / ID: 3 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Semliki Forest virus Semliki Forest virus |
| Molecular weight | Theoretical: 6.020911 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: TAMCVLANAT FPCFQPPCVP CCYENNAEAT LRMLEDNVDR PGYYDLLQAA LTCR UniProtKB: Structural polyprotein |
-Macromolecule #4: Capsid protein
| Macromolecule | Name: Capsid protein / type: protein_or_peptide / ID: 4 / Number of copies: 4 / Enantiomer: LEVO / EC number: togavirin |
|---|---|
| Source (natural) | Organism:   Semliki Forest virus Semliki Forest virus |
| Molecular weight | Theoretical: 16.723904 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: IENDCIFEVK HEGKVTGYAC LVGDKVMKPA HVKGVIDNAD LAKLAFKKSS KYDLECAQIP VHMRSDASKY THEKPEGHYN WHHGAVQYS GGRFTIPTGA GKPGDSGRPI FDNKGRVVAI VLGGANEGSR TALSVVTWNK DMVTRVTPEG SEEW UniProtKB: Structural polyprotein |
-Macromolecule #5: Very low-density lipoprotein receptor
| Macromolecule | Name: Very low-density lipoprotein receptor / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 4.028225 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: CAESDFVCNN GQCVPSRWKC DGDPDCEDGS DESPEQC UniProtKB: Very low-density lipoprotein receptor |
-Macromolecule #8: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 8 / Number of copies: 2 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #9: CALCIUM ION
| Macromolecule | Name: CALCIUM ION / type: ligand / ID: 9 / Number of copies: 1 / Formula: CA |
|---|---|
| Molecular weight | Theoretical: 40.078 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.2 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 54.9 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: EMDB MAP EMDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.7 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 439486 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION |
| Final angle assignment | Type: ANGULAR RECONSTITUTION |
 Movie
Movie Controller
Controller













 Z
Z Y
Y X
X