+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | E. coli ExoVII(H238A) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Exonuclease / Endonuclease / DNA repair / DNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationexodeoxyribonuclease VII / exodeoxyribonuclease VII activity / exodeoxyribonuclease VII complex / DNA catabolic process / mismatch repair / DNA binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Liu C / Berger JM | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2024 Journal: Proc Natl Acad Sci U S A / Year: 2024Title: Structure of exonuclease VII. Authors: Chuan Liu / Glenn Hauk / Qianyun Yan / James M Berger /  Abstract: Exonuclease VII (ExoVII) is a ubiquitous bacterial nuclease. Encoded by the and genes, ExoVII participates in multiple nucleic acid-dependent pathways including the processing of multicopy single- ...Exonuclease VII (ExoVII) is a ubiquitous bacterial nuclease. Encoded by the and genes, ExoVII participates in multiple nucleic acid-dependent pathways including the processing of multicopy single-stranded DNA and the repair of covalent DNA-protein crosslinks (DPCs). Although many biochemical properties of ExoVII have been defined, little is known about its structure/function relationships. Here, we use cryoelectron microscopy (cryoEM) to determine that ExoVII comprises a highly elongated XseA·XseB holo-complex. Each XseA subunit dimerizes through a central extended α-helical segment decorated by six XseB subunits and a C-terminal, domain-swapped β-barrel element; two XseA·XseB subcomplexes further associate using N-terminal OB (oligonucleotide/oligosaccharide-binding) folds and catalytic domains to form a spindle-shaped, catenated octaicosamer. The catalytic domains of XseA, which adopt a nuclease fold related to 3-dehydroquinate dehydratases, are sequestered in the center of the complex and accessible only through large pores formed between XseA tetramers. The architectural organization of ExoVII, combined with biochemical studies, indicate that substrate selectivity is controlled by steric access to its nuclease elements and that tetramer dissociation results from substrate DNA binding. Despite a lack of sequence and fold homology, the physical organization of ExoVII is reminiscent of Mre11·Rad50/SbcCD ATP (adenosine triphosphate)-dependent nucleases used in the repair of double-stranded DNA breaks, including those formed by DPCs through aberrant topoisomerase activity, suggesting that there may have been convergent evolutionary pressure to contend with such damage events. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41704.map.gz emd_41704.map.gz | 496.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41704-v30.xml emd-41704-v30.xml emd-41704.xml emd-41704.xml | 13.3 KB 13.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_41704.png emd_41704.png | 40.9 KB | ||
| Filedesc metadata |  emd-41704.cif.gz emd-41704.cif.gz | 5.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41704 http://ftp.pdbj.org/pub/emdb/structures/EMD-41704 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41704 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41704 | HTTPS FTP |
-Related structure data
| Related structure data |  8txrMC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_41704.map.gz / Format: CCP4 / Size: 590.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41704.map.gz / Format: CCP4 / Size: 590.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1 Å | ||||||||||||||||||||||||||||||||||||
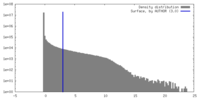
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : XseA4-XseB24 complex of ExoVII
| Entire | Name: XseA4-XseB24 complex of ExoVII |
|---|---|
| Components |
|
-Supramolecule #1: XseA4-XseB24 complex of ExoVII
| Supramolecule | Name: XseA4-XseB24 complex of ExoVII / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 420 KDa |
-Macromolecule #1: Exodeoxyribonuclease 7 large subunit
| Macromolecule | Name: Exodeoxyribonuclease 7 large subunit / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 51.840141 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MLPSQSPAIF TVSRLNQTVR LLLEHEMGQV WISGEISNFT QPASGHWYFT LKDDTAQVRC AMFRNSNRRV TFRPQHGQQV LVRANITLY EPRGDYQIIV ESMQPAGEGL LQQKYEQLKA KLQAEGLFDQ QYKKPLPSPA HCVGVITSKT GAALHDILHV L KRRDPSLP ...String: MLPSQSPAIF TVSRLNQTVR LLLEHEMGQV WISGEISNFT QPASGHWYFT LKDDTAQVRC AMFRNSNRRV TFRPQHGQQV LVRANITLY EPRGDYQIIV ESMQPAGEGL LQQKYEQLKA KLQAEGLFDQ QYKKPLPSPA HCVGVITSKT GAALHDILHV L KRRDPSLP VIIYPAAVQG DDAPGQIVRA IELANQRNEC DVLIVGRGGG SLEDLWSFND ERVARAIFTS RIPVVSAVGA ET DVTIADF VADLRAPTPS AAAEVVSRNQ QELLRQVQST RQRLEMAMDY YLANRTRRFT QIHHRLQQQH PQLRLARQQT MLE RLQKRM SFALENQLKR TGQQQQRLTQ RLNQQNPQPK IHRAQTRIQQ LEYRLAETLR AQLSATRERF GNAVTHLEAV SPLS TLARG YSVTTATDGN VLKKVKQVKA GEMLTTRLED GWIESEVKNI QPVKKSRKKV H UniProtKB: Exodeoxyribonuclease 7 large subunit |
-Macromolecule #2: Exodeoxyribonuclease 7 small subunit
| Macromolecule | Name: Exodeoxyribonuclease 7 small subunit / type: protein_or_peptide / ID: 2 / Number of copies: 16 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 8.959877 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MPKKNEAPAS FEKALSELEQ IVTRLESGDL PLEEALNEFE RGVQLARQGQ AKLQQAEQRV QILLSDNEDA SLTPFTPDNE UniProtKB: Exodeoxyribonuclease 7 small subunit |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.3 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.75 µm / Nominal defocus min: 0.75 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)