+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | XptA2 wild type | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | TcA / Insecticide / Translocase / TOXIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationABC toxin, N-terminal domain / ABC toxin N-terminal region / TcA receptor binding domain / TcA receptor binding domain / Insecticidal toxin complex/plasmid virulence protein / Tc toxin complex TcA, C-terminal TcB-binding domain / Neuraminidase-like domain / Salmonella virulence plasmid 28.1kDa A protein / Tc toxin complex TcA C-terminal TcB-binding domain / Neuraminidase-like domain Similarity search - Domain/homology | |||||||||
| Biological species |  Xenorhabdus nematophila (bacteria) Xenorhabdus nematophila (bacteria) | |||||||||
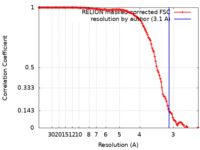
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Martin CL / Binshtein EM / Aller SG | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Int J Mol Sci / Year: 2023 Journal: Int J Mol Sci / Year: 2023Title: Structures of the Insecticidal Toxin Complex Subunit XptA2 Highlight Roles for Flexible Domains. Authors: Cole L Martin / David W Chester / Christopher D Radka / Lurong Pan / Zhengrong Yang / Rachel C Hart / Elad M Binshtein / Zhao Wang / Lisa Nagy / Lawrence J DeLucas / Stephen G Aller /  Abstract: The Toxin Complex (Tc) superfamily consists of toxin translocases that contribute to the targeting, delivery, and cytotoxicity of certain pathogenic Gram-negative bacteria. Membrane receptor ...The Toxin Complex (Tc) superfamily consists of toxin translocases that contribute to the targeting, delivery, and cytotoxicity of certain pathogenic Gram-negative bacteria. Membrane receptor targeting is driven by the A-subunit (TcA), which comprises IgG-like receptor binding domains (RBDs) at the surface. To better understand XptA2, an insect specific TcA secreted by the symbiont from the intestine of entomopathogenic nematodes, we determined structures by X-ray crystallography and cryo-EM. Contrary to a previous report, XptA2 is pentameric. RBD-B exhibits an indentation from crystal packing that indicates loose association with the shell and a hotspot for possible receptor binding or a trigger for conformational dynamics. A two-fragment XptA2 lacking an intact linker achieved the folded pre-pore state like wild type (wt), revealing no requirement of the linker for protein folding. The linker is disordered in all structures, and we propose it plays a role in dynamics downstream of the initial pre-pore state. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41503.map.gz emd_41503.map.gz | 148.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41503-v30.xml emd-41503-v30.xml emd-41503.xml emd-41503.xml | 17.3 KB 17.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_41503_fsc.xml emd_41503_fsc.xml | 12.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_41503.png emd_41503.png | 137.9 KB | ||
| Filedesc metadata |  emd-41503.cif.gz emd-41503.cif.gz | 7.3 KB | ||
| Others |  emd_41503_half_map_1.map.gz emd_41503_half_map_1.map.gz emd_41503_half_map_2.map.gz emd_41503_half_map_2.map.gz | 144.4 MB 144.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41503 http://ftp.pdbj.org/pub/emdb/structures/EMD-41503 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41503 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41503 | HTTPS FTP |
-Validation report
| Summary document |  emd_41503_validation.pdf.gz emd_41503_validation.pdf.gz | 929.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_41503_full_validation.pdf.gz emd_41503_full_validation.pdf.gz | 928.7 KB | Display | |
| Data in XML |  emd_41503_validation.xml.gz emd_41503_validation.xml.gz | 19.8 KB | Display | |
| Data in CIF |  emd_41503_validation.cif.gz emd_41503_validation.cif.gz | 25.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41503 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41503 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41503 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41503 | HTTPS FTP |
-Related structure data
| Related structure data |  8tqeMC  8tv0C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_41503.map.gz / Format: CCP4 / Size: 169.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41503.map.gz / Format: CCP4 / Size: 169.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.268 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_41503_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_41503_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Pentameric assembly of the XptA2 TcA
| Entire | Name: Pentameric assembly of the XptA2 TcA |
|---|---|
| Components |
|
-Supramolecule #1: Pentameric assembly of the XptA2 TcA
| Supramolecule | Name: Pentameric assembly of the XptA2 TcA / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: This is a Protein toxin that is secreted in bacteria. I assume the best category to pick from is Organelle or Cellular Component |
|---|---|
| Source (natural) | Organism:  Xenorhabdus nematophila (bacteria) Xenorhabdus nematophila (bacteria) |
| Molecular weight | Theoretical: 1.4 MDa |
-Macromolecule #1: XptA2
| Macromolecule | Name: XptA2 / type: protein_or_peptide / ID: 1 / Number of copies: 5 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Xenorhabdus nematophila (bacteria) Xenorhabdus nematophila (bacteria) |
| Molecular weight | Theoretical: 284.392188 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MYSTAVLLNK ISPTRDGQTM TLADLQYLSF SELRKIFDDQ LSWGEARHLY HETIEQKKNN RLLEARIFTR ANPQLSGAIR LGIERDSVS RSYDEMFGAR SSSFVKPGSV ASMFSPAGYL TELYREAKDL HFSSSAYHLD NRRPDLADLT LSQSNMDTEI S TLTLSNEL ...String: MYSTAVLLNK ISPTRDGQTM TLADLQYLSF SELRKIFDDQ LSWGEARHLY HETIEQKKNN RLLEARIFTR ANPQLSGAIR LGIERDSVS RSYDEMFGAR SSSFVKPGSV ASMFSPAGYL TELYREAKDL HFSSSAYHLD NRRPDLADLT LSQSNMDTEI S TLTLSNEL LLEHITRKTG GDSDALMESL STYRQAIDTP YHQPYETIRQ VIMTHDSTLS ALSRNPEVMG QAEGASLLAI LA NISPELY NILTEEITEK NADALFAQNF SENITPENFA SQSWIAKYYG LELSEVQKYL GMLQNGYSDS TSAYVDNIST GLV VNNESK LEAYKITRVK TDDYDKNINY FDLMYEGNNQ FFIRANFKVS REFGATLRKN AGPSGIVGSL SGPLIANTNF KSNY LSNIS DSEYKNGVKI YAYRYTSSTS ATNQGGGIFT FESYPLTIFA LKLNKAIRLC LTSGLSPNEL QTIVRSDNAQ GIIND SVLT KVFYTLFYSH RYALSFDDAQ VLNGSVINQY ADDDSVSHFN RLFNTPPLKG KIFEADGNTV SIDPDEEQST FARSAL MRG LGVNSGELYQ LGKLAGVLDA QNTITLSVFV ISSLYRLTLL ARVHQLTVNE LCMLYGLSPF NGKTTASLSS GELPRLV IW LYQVTQWLTE AEITTEAIWL LCTPEFSGNI SPEISNLLNN LRPSISEDMA QSHNRELQAE ILAPFIAATL HLASPDMA R YILLWTDNLR PGGLDIAGFM TLVLKESLNA NETTQLVQFC HVMAQLSLSV QTLRLSEAEL SVLVISGFAV LGAKNQPAG QHNIDTLFSL YRFHQWINGL GNPGSDTLDM LRQQTLTADR LASVMGLDIS MVTQAMVSAG VNQLQCWQDI NTVLQWIDVA SALHTMPSV IRTLVNIRYV TALNKAESNL PSWDEWQTLA ENMEAGLSTQ QAQTLADYTA ERLSSVLCNW FLANIQPEGV S LHSRDDLY SYFLIDNQVS SAIKTTRLAE AIAGIQLYIN RALNRIEPNA RADVSTRQFF TDWTVNNRYS TWGGVSRLVY YP ENYIDPT QRIGQTRMMD ELLENISQSK LSRDTVEDAF KTYLTRFETV ADLKVVSAYH DNVNSNTGLT WFVGQTRENL PEY YWRNVD ISRMQAGELA ANAWKEWTKI DTAVNPYKDA IRPVIFRERL HLIWVEKEEV AKNGTDPVET YDRFTLKLAF LRHD GSWSA PWSYDITTQV EAVTDKKPDT ERLALAASGF QGEDTLLVFV YKTGKSYSDF GGSNKNVAGM TIYGDGSFKK MENTA LSRY SQLKNTFDII HTQGNDLVRK ASYRFAQDFE VPASLNMGSA IGDDSLTVME NGNIPQITSK YSSDNLAITL HNAAFT VRY DGSGNVIRNK QISAMKLTGV DGKSQYGNAF IIANTVKHYG GYSDLGGPIT VYNKTKNYIA SVQGHLMNAD YTRRLIL TP VENNYYARLF EFPFSPNTIL NTVFTVGSNK TSDFKKCSYA VDGNNSQGFQ IFSSYQSSGW LDIDTGINNT DIKITVMA G SKTHTFTASD HIASLPANSF DAMPYTFKPL EIDASSLAFT NNIAPLDIVF ETKAKDGRVL GKIKQTLSVK RVNYNPEDI LFLRETHSGA QYMQLGVYRI RLNTLLASQL VSRANTGIDT ILTMETQRLP EPPLGEGFFA NFVLPKYDPA EHGDERWFKI HIGNVGGNT GRQPYYSGML SDTSETSMTL FVPYAEGYYM HEGVRLGVGY QKITYDNTWE SAFFYFDETK QQFVLINDAD H DSGMTQQG IVKNIKKYKG FLNVSIATGY SAPMDFNSAS ALYYWELFYY TPMMCFQRLL QEKQFDEATQ WINYVYNPAG YI VNGEIAP WIWNCRPLEE TTSWNANPLD AIDPDAVAQN DPMHYKIATF MRLLDQLILR GDMAYRELTR DALNEAKMWY VRT LELLGD EPEDYGSQQW AAPSLSGAAS QTVQAAYQQD LTMLGRGGVS KNLRTANSLV GLFLPEYNPA LTDYWQTLRL RLFN LRHNL SIDGQPLSLA IYAEPTDPKA LLTSMVQASQ GGSAVLPGTL SLYRFPVMLE RTRNLVAQLT QFGTSLLSMA EHDDA DELT TLLLQQGMEL ATQSIRIQQR TVDEVDADIA VLAESRRSAQ NRLEKYQQLY DEDINHGEQR AMSLLDAAAG QSLAGQ VLS IAEGVADLVP NVFGLACGGS RWGAALRASA SVMSLSATAS QYSADKISRS EAYRRRRQEW EIQRDNADGE VKQMDAQ LE SLKIRREAAQ MQVEYQETQQ AHTQAQLELL QRKFTNKALY SWMRGKLSAI YYQFFDLTQS FCLMAQEALR RELTDNGV T FIRGGAWNGT TAGLMAGETL LLNLAEMEKV WLERDERALE VTRTVSLAQF YQALSSDNFN LTEKLTQFLR EGKGNVGAS GNELKLSNRQ IEASVRLSDL KIFSDYPESL GNTRQLKQVS VTLPALVGPY EDIRAVLNYG GSIVMPRGCS AIALSHGVND SGQFMLDFN DSRYLPFEGI SVNDSGSLTL SFPDATDRQK ALLESLSDII LHIRYTIRS UniProtKB: A component of insecticidal toxin complex (Tc) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.5 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Grid | Material: COPPER / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 75.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.2 mm / Nominal defocus max: 4.0 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)