+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
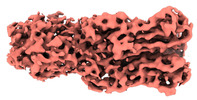
| Title | Cyanophage Pam3 fiber | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | fiber / VIRUS / VIRAL PROTEIN | |||||||||
| Biological species |  uncultured cyanophage (environmental samples) uncultured cyanophage (environmental samples) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.96 Å | |||||||||
 Authors Authors | Yang F / Jiang YL / Zhou CZ | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2023 Journal: Proc Natl Acad Sci U S A / Year: 2023Title: Fine structure and assembly pattern of a minimal myophage Pam3. Authors: Feng Yang / Yong-Liang Jiang / Jun-Tao Zhang / Jie Zhu / Kang Du / Rong-Cheng Yu / Zi-Lu Wei / Wen-Wen Kong / Ning Cui / Wei-Fang Li / Yuxing Chen / Qiong Li / Cong-Zhao Zhou /  Abstract: The myophage possesses a contractile tail that penetrates its host cell envelope. Except for investigations on the bacteriophage T4 with a rather complicated structure, the assembly pattern and tail ...The myophage possesses a contractile tail that penetrates its host cell envelope. Except for investigations on the bacteriophage T4 with a rather complicated structure, the assembly pattern and tail contraction mechanism of myophage remain largely unknown. Here, we present the fine structure of a freshwater cyanophage Pam3, which has an icosahedral capsid of ~680 Å in diameter, connected via a three-section neck to an 840-Å-long contractile tail, ending with a three-module baseplate composed of only six protein components. This simplified baseplate consists of a central hub-spike surrounded by six wedge heterotriplexes, to which twelve tail fibers are covalently attached via disulfide bonds in alternating upward and downward configurations. In vitro reduction assays revealed a putative redox-dependent mechanism of baseplate assembly and tail sheath contraction. These findings establish a minimal myophage that might become a user-friendly chassis phage in synthetic biology. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33799.map.gz emd_33799.map.gz | 404.9 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33799-v30.xml emd-33799-v30.xml emd-33799.xml emd-33799.xml | 13.9 KB 13.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_33799.png emd_33799.png | 100.2 KB | ||
| Filedesc metadata |  emd-33799.cif.gz emd-33799.cif.gz | 5.1 KB | ||
| Others |  emd_33799_half_map_1.map.gz emd_33799_half_map_1.map.gz emd_33799_half_map_2.map.gz emd_33799_half_map_2.map.gz | 23.3 MB 23.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33799 http://ftp.pdbj.org/pub/emdb/structures/EMD-33799 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33799 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33799 | HTTPS FTP |
-Related structure data
| Related structure data |  7yfwMC  7yfzC  8hdrC  8hdsC  8hdtC  8hdwC M: atomic model generated by this map C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_33799.map.gz / Format: CCP4 / Size: 2.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33799.map.gz / Format: CCP4 / Size: 2.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_33799_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_33799_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : uncultured cyanophage
| Entire | Name:  uncultured cyanophage (environmental samples) uncultured cyanophage (environmental samples) |
|---|---|
| Components |
|
-Supramolecule #1: uncultured cyanophage
| Supramolecule | Name: uncultured cyanophage / type: virus / ID: 1 / Parent: 0 / Macromolecule list: all / NCBI-ID: 215796 / Sci species name: uncultured cyanophage / Virus type: VIRION / Virus isolate: OTHER / Virus enveloped: No / Virus empty: No |
|---|---|
| Host (natural) | Organism:  Pseudanabaena mucicola (bacteria) Pseudanabaena mucicola (bacteria) |
-Macromolecule #1: Pam3 fiber proreins
| Macromolecule | Name: Pam3 fiber proreins / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  uncultured cyanophage (environmental samples) uncultured cyanophage (environmental samples) |
| Molecular weight | Theoretical: 27.323789 KDa |
| Sequence | String: MASINLPFSL SGSKRIPTSE ELADGYQCGP LDVELDNWLM WWLTGQVDGV IEGAGLTTDD TDLARLYKAI QSMTSGNLRT VVLTAASGN LPIPSDVSVL NWVRAVGGGG AGGNSNTGNS KASGGGGGAG FDRFNVAVTP GSNVPYTVGA AGAVNGLGAG Y NGGAGGST ...String: MASINLPFSL SGSKRIPTSE ELADGYQCGP LDVELDNWLM WWLTGQVDGV IEGAGLTTDD TDLARLYKAI QSMTSGNLRT VVLTAASGN LPIPSDVSVL NWVRAVGGGG AGGNSNTGNS KASGGGGGAG FDRFNVAVTP GSNVPYTVGA AGAVNGLGAG Y NGGAGGST AILGTTAGGG AGGLGVNNNA TAVQVNGGTT SGTTPEISYP GGLGTEGIVG TGGGSVLSQP TQRAFTNAGN NN PANSWGG GGPGGSDFGG AWQPGGVGKQ GIIIVQYFSR FAP |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 300 K |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.96 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 3.1) / Number images used: 45155 |
| Initial angle assignment | Type: RANDOM ASSIGNMENT |
| Final angle assignment | Type: RANDOM ASSIGNMENT |
 Movie
Movie Controller
Controller








 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)