[English] 日本語
 Yorodumi
Yorodumi- EMDB-33394: Structure of C-terminal truncated connexin43/Cx43/GJA1 gap juncti... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of C-terminal truncated connexin43/Cx43/GJA1 gap junction intercellular channel in POPE/CHS nanodiscs | |||||||||
 Map data Map data | sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Cx43 / Connexin / Gap junction channel / Gating mechanism / Membrane protein | |||||||||
| Function / homology |  Function and homology information Function and homology informationgap junction channel activity involved in cardiac conduction electrical coupling / negative regulation of gonadotropin secretion / microtubule-based transport / positive regulation of morphogenesis of an epithelium / positive regulation of mesodermal cell differentiation / cell communication by electrical coupling / negative regulation of trophoblast cell migration / SARS-CoV-2 targets PDZ proteins in cell-cell junction / gap junction channel activity involved in cell communication by electrical coupling / monoatomic ion transmembrane transporter activity ...gap junction channel activity involved in cardiac conduction electrical coupling / negative regulation of gonadotropin secretion / microtubule-based transport / positive regulation of morphogenesis of an epithelium / positive regulation of mesodermal cell differentiation / cell communication by electrical coupling / negative regulation of trophoblast cell migration / SARS-CoV-2 targets PDZ proteins in cell-cell junction / gap junction channel activity involved in cell communication by electrical coupling / monoatomic ion transmembrane transporter activity / gap junction hemi-channel activity / Oligomerization of connexins into connexons / Transport of connexins along the secretory pathway / gap junction assembly / Regulation of gap junction activity / Microtubule-dependent trafficking of connexons from Golgi to the plasma membrane / glutathione transmembrane transporter activity / atrial cardiac muscle cell action potential / cardiac conduction system development / cell-cell contact zone / Golgi-associated vesicle membrane / connexin complex / cell communication by electrical coupling involved in cardiac conduction / Formation of annular gap junctions / Gap junction degradation / gap junction / bone remodeling / export across plasma membrane / Gap junction assembly / gap junction channel activity / glutamate secretion / tight junction / Mechanical load activates signaling by PIEZO1 and integrins in osteocytes / positive regulation of stem cell proliferation / RHOJ GTPase cycle / RHOQ GTPase cycle / establishment of mitotic spindle orientation / maintenance of blood-brain barrier / efflux transmembrane transporter activity / intercalated disc / xenobiotic transport / alpha-tubulin binding / positive regulation of vascular associated smooth muscle cell proliferation / tubulin binding / negative regulation of cell growth / bone development / beta-catenin binding / cellular response to amyloid-beta / cell junction / intracellular protein localization / cell-cell signaling / positive regulation of cold-induced thermogenesis / heart development / spermatogenesis / monoatomic ion transmembrane transport / positive regulation of canonical NF-kappaB signal transduction / apical plasma membrane / membrane raft / Golgi membrane / focal adhesion / positive regulation of gene expression / endoplasmic reticulum membrane / Golgi apparatus / signal transduction / mitochondrion / nucleoplasm / nucleus / plasma membrane / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
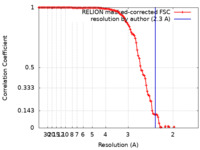
| Method | single particle reconstruction / cryo EM / Resolution: 2.3 Å | |||||||||
 Authors Authors | Lee HJ / Cha HJ / Jeong H / Lee SN / Lee CW / Woo JS | |||||||||
| Funding support |  Korea, Republic Of, 1 items Korea, Republic Of, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Conformational changes in the human Cx43/GJA1 gap junction channel visualized using cryo-EM. Authors: Hyuk-Joon Lee / Hyung Jin Cha / Hyeongseop Jeong / Seu-Na Lee / Chang-Won Lee / Minsoo Kim / Jejoong Yoo / Jae-Sung Woo /  Abstract: Connexin family proteins assemble into hexameric hemichannels in the cell membrane. The hemichannels dock together between two adjacent membranes to form gap junction intercellular channels (GJIChs). ...Connexin family proteins assemble into hexameric hemichannels in the cell membrane. The hemichannels dock together between two adjacent membranes to form gap junction intercellular channels (GJIChs). We report the cryo-electron microscopy structures of Cx43 GJICh, revealing the dynamic equilibrium state of various channel conformations in detergents and lipid nanodiscs. We identify three different N-terminal helix conformations of Cx43-gate-covering (GCN), pore-lining (PLN), and flexible intermediate (FIN)-that are randomly distributed in purified GJICh particles. The conformational equilibrium shifts to GCN by cholesteryl hemisuccinates and to PLN by C-terminal truncations and at varying pH. While GJIChs that mainly comprise GCN protomers are occluded by lipids, those containing conformationally heterogeneous protomers show markedly different pore sizes. We observe an α-to-π-helix transition in the first transmembrane helix, which creates a side opening to the membrane in the FIN and PLN conformations. This study provides basic structural information to understand the mechanisms of action and regulation of Cx43 GJICh. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33394.map.gz emd_33394.map.gz | 116.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33394-v30.xml emd-33394-v30.xml emd-33394.xml emd-33394.xml | 15.3 KB 15.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_33394_fsc.xml emd_33394_fsc.xml | 11.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_33394.png emd_33394.png | 41.7 KB | ||
| Filedesc metadata |  emd-33394.cif.gz emd-33394.cif.gz | 5.6 KB | ||
| Others |  emd_33394_half_map_1.map.gz emd_33394_half_map_1.map.gz emd_33394_half_map_2.map.gz emd_33394_half_map_2.map.gz | 96.4 MB 96.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33394 http://ftp.pdbj.org/pub/emdb/structures/EMD-33394 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33394 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33394 | HTTPS FTP |
-Related structure data
| Related structure data |  7xqfMC  7f92C  7f93C  7f94C  7xq9C  7xqbC  7xqdC  7xqgC  7xqhC  7xqiC  7xqjC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_33394.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33394.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.848 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: half map
| File | emd_33394_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map | ||||||||||||
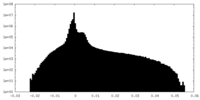
| Projections & Slices |
| ||||||||||||
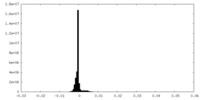
| Density Histograms |
-Half map: half map
| File | emd_33394_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map | ||||||||||||
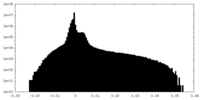
| Projections & Slices |
| ||||||||||||
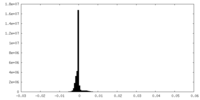
| Density Histograms |
- Sample components
Sample components
-Entire : Dodecameric complex of C-terminal deletion mutant of human Cx43/G...
| Entire | Name: Dodecameric complex of C-terminal deletion mutant of human Cx43/GJA1 (Cx43-M257) in lipid nanodiscs (POPE/CHS) |
|---|---|
| Components |
|
-Supramolecule #1: Dodecameric complex of C-terminal deletion mutant of human Cx43/G...
| Supramolecule | Name: Dodecameric complex of C-terminal deletion mutant of human Cx43/GJA1 (Cx43-M257) in lipid nanodiscs (POPE/CHS) type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 Details: Human Cx43-M257 gap junction channel with gate-covering NTH conformation |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Gap junction alpha-1 protein
| Macromolecule | Name: Gap junction alpha-1 protein / type: protein_or_peptide / ID: 1 / Number of copies: 12 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 29.396547 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MGDWSALGKL LDKVQAYSTA GGKVWLSVLF IFRILLLGTA VESAWGDEQS AFRCNTQQPG CENVCYDKSF PISHVRFWVL QIIFVSVPT LLYLAHVFYV MRKEEKLNKK EEELKVAQTD GVNVDMHLKQ IEIKKFKYGI EEHGKVKMRG GLLRTYIISI L FKSIFEVA ...String: MGDWSALGKL LDKVQAYSTA GGKVWLSVLF IFRILLLGTA VESAWGDEQS AFRCNTQQPG CENVCYDKSF PISHVRFWVL QIIFVSVPT LLYLAHVFYV MRKEEKLNKK EEELKVAQTD GVNVDMHLKQ IEIKKFKYGI EEHGKVKMRG GLLRTYIISI L FKSIFEVA FLLIQWYIYG FSLSAVYTCK RDPCPHQVDC FLSRPTEKTI FIIFMLVVSL VSLALNIIEL FYVFFKGVKD RV KGKSDPY HATSGALSPA UniProtKB: Gap junction alpha-1 protein |
-Macromolecule #2: TETRADECANE
| Macromolecule | Name: TETRADECANE / type: ligand / ID: 2 / Number of copies: 132 / Formula: C14 |
|---|---|
| Molecular weight | Theoretical: 198.388 Da |
| Chemical component information |  ChemComp-C14: |
-Macromolecule #3: PHOSPHATIDYLETHANOLAMINE
| Macromolecule | Name: PHOSPHATIDYLETHANOLAMINE / type: ligand / ID: 3 / Number of copies: 12 / Formula: PTY |
|---|---|
| Molecular weight | Theoretical: 734.039 Da |
| Chemical component information |  ChemComp-PTY: |
-Macromolecule #4: CHOLESTEROL HEMISUCCINATE
| Macromolecule | Name: CHOLESTEROL HEMISUCCINATE / type: ligand / ID: 4 / Number of copies: 24 / Formula: Y01 |
|---|---|
| Molecular weight | Theoretical: 486.726 Da |
| Chemical component information |  ChemComp-Y01: |
-Macromolecule #5: water
| Macromolecule | Name: water / type: ligand / ID: 5 / Number of copies: 492 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 72.42 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.4 µm / Nominal defocus min: 0.9 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller

















 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)